Abstract
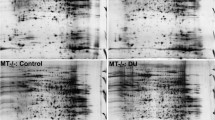
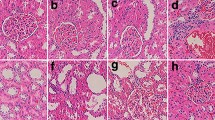
Cadmium (Cd) is a highly toxic metal and kidney is its main target. However, the molecular effects and associated potential impacts of Cd-accumulated kidney have not been well investigated. In this study, mouse was used as a model to investigate the Cd-induced proteomic profile change in kidney, and a total of 34 differentially expressed proteins were detected by two-dimensional gel electrophoresis (2-DE) and further identified by matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS). Through Gene Ontology analysis and KEGG pathway annotation, it showed that Cd-regulated kidney metabolism and promoted renal damage and cell migration. By validation of Western blotting and RT-qPCR, metastasis-related proteins LIM and SH3 domain protein 1 (LASP1) and phosphoenolpyruvate carboxykinase/cytosolic [GTP] (PEPCK1) were confirmed to be upregulated; Acyl-CoA synthetase medium-chain family member 3 (ACSM3) was downregulated. Furthermore, carcinoma development-related proteins initiation factor 4A (eIF4A) and pyridoxine-5′-phosphate oxidase (PNPO) were upregulated, and pyridoxal kinase (PK) was downregulated. The downregulation of Na(+)/H(+) exchange regulatory cofactor (NHERF3) might promote renal damage which associated with decrease of transferrin (TRF) in kidney. Taken together, our results revealed proteomic profile of Cd-induced nephrotoxicity and provided data for further insights into the mechanisms of Cd toxicity.







Similar content being viewed by others
Abbreviations
- Cd:
-
Cadmium
- 2-DE:
-
Two-dimensional electrophoresis
- MS:
-
Mass spectrometry
- ACSM3:
-
Acyl-CoA synthetase medium-chain family member 3
- TRF:
-
Transferrin
- PEPCK1:
-
Phosphoenolpyruvate carboxykinase 1, cytosolic
- PNPO:
-
Pyridoxine 5′-phosphate oxidase
- eIF4A:
-
Eukaryotic translation initiation factor 4A
- PDZK1:
-
PDZ domain containing 1
- LASP1:
-
LIM and SH3 protein 1
- PK:
-
Pyridoxal kinase
- MMP-2:
-
Matrix metalloproteinase 2
- MMP-9:
-
Matrix metalloproteinase 9
References
Company R, Antunez O, Cosson RP, Serafim A, Shillito B, Cajaraville M, Bebianno MJ, Torreblanca A (2019) Protein expression profiles in Bathymodiolus azoricus exposed to cadmium. Ecotoxicol Environ Saf 171:621–630. https://doi.org/10.1016/j.ecoenv.2019.01.031
Hu X, Chandler JD, Park S, Liu K, Fernandes J, Orr M, Smith MR, Ma C, Kang SM, Uppal K, Jones DP, Go YM (2019) Low-dose cadmium disrupts mitochondrial citric acid cycle and lipid metabolism in mouse lung. Free Radic Biol Med 131:209–217. https://doi.org/10.1016/j.freeradbiomed.2018.12.005
Yang H, Shu Y (2015) Cadmium transporters in the kidney and cadmium-induced nephrotoxicity. Int J Mol Sci 16(1):1484–1494. https://doi.org/10.3390/ijms16011484
Zhang H, Reynolds M (2019) Cadmium exposure in living organisms: a short review. Sci Total Environ 678:761–767. https://doi.org/10.1016/j.scitotenv.2019.04.395
Chen J, He W, Zhu X, Yang S, Yu T, Ma W (2019) Epidemiological study of kidney health in an area with high levels of soil cadmium and selenium: does selenium protect against cadmium-induced kidney injury? Sci Total Environ 698:134106. https://doi.org/10.1016/j.scitotenv.2019.134106
Ferraro PM, Costanzi S, Naticchia A, Sturniolo A, Gambaro G (2010) Low level exposure to cadmium increases the risk of chronic kidney disease: analysis of the NHANES 1999-2006. BMC Public Health 10:304. https://doi.org/10.1186/1471-2458-10-304
Jarup L, Akesson A (2009) Current status of cadmium as an environmental health problem. Toxicol Appl Pharmacol 238(3):201–208. https://doi.org/10.1016/j.taap.2009.04.020
Bernard A (2004) Renal dysfunction induced by cadmium: biomarkers of critical effects. Biometals 17(5):519–523. https://doi.org/10.1023/b:biom.0000045731.75602.b9
Haynes CM, Titus EA, Cooper AA (2004) Degradation of misfolded proteins prevents ER-derived oxidative stress and cell death. Mol Cell 15(5):767–776. https://doi.org/10.1016/j.molcel.2004.08.025
Yokouchi M, Hiramatsu N, Hayakawa K, Kasai A, Takano Y, Yao J, Kitamura M (2007) Atypical, bidirectional regulation of cadmium-induced apoptosis via distinct signaling of unfolded protein response. Cell Death Differ 14(8):1467–1474. https://doi.org/10.1038/sj.cdd.4402154
Yuan HM, Liu WC, Jin Y, Lu YT (2013) Role of ROS and auxin in plant response to metal-mediated stress. Plant Signal Behav 8(7):e24671. https://doi.org/10.4161/psb.24671
Gu J, Ren Z, Zhao J, Peprah FA, Xie Y, Cheng D, Wang Y, Liu H, Chu Wong CK, Zhou Y, Shi H (2020) Calcimimetic compound NPS R-467 protects against chronic cadmium-induced mouse kidney injury by restoring autophagy process. Ecotoxicol Environ Saf 189:110052. https://doi.org/10.1016/j.ecoenv.2019.110052
Il'yasova D, Schwartz GG (2005) Cadmium and renal cancer. Toxicol Appl Pharmacol 207(2):179–186. https://doi.org/10.1016/j.taap.2004.12.005
Song J, Luo H, Yin X, Huang G, Luo S, Lin du R, Yuan DB, Zhang W, Zhu J (2015) Association between cadmium exposure and renal cancer risk: a meta-analysis of observational studies. Sci Rep 5: 17976. https://doi.org/10.1038/srep17976
Ge J, Zhang C, Sun YC, Zhang Q, Lv MW, Guo K, Li JL (2019) Cadmium exposure triggers mitochondrial dysfunction and oxidative stress in chicken (Gallus gallus) kidney via mitochondrial UPR inhibition and Nrf2-mediated antioxidant defense activation. Sci Total Environ 689:1160–1171. https://doi.org/10.1016/j.scitotenv.2019.06.405
Das S, Dewanjee S, Dua TK, Joardar S, Chakraborty P, Bhowmick S, Saha A, Bhattacharjee S, De Feo V (2019) Carnosic acid attenuates cadmium induced nephrotoxicity by inhibiting oxidative stress, promoting Nrf2/HO-1 signalling and impairing TGF-beta1/Smad/collagen IV signalling. Molecules 24(22):4176. https://doi.org/10.3390/molecules24224176
Naaby-Hansen S, Waterfield MD, Cramer R (2001) Proteomics--post-genomic cartography to understand gene function. Trends Pharmacol Sci 22(7):376–384. https://doi.org/10.1016/s0165-6147(00)01663-1
Wen M, Jin Y, Zhang H, Sun X, Kuai Y, Tan W (2019) Proteomic analysis of rat cerebral cortex in the subacute to long-term phases of focal cerebral ischemia-reperfusion injury. J Proteome Res 18(8):3099–3118. https://doi.org/10.1021/acs.jproteome.9b00220
Engstrom Y, Loseva O, Theopold U (2004) Proteomics of the Drosophila immune response. Trends Biotechnol 22(11):600–605. https://doi.org/10.1016/j.tibtech.2004.09.002
Gorg A, Weiss W, Dunn MJ (2004) Current two-dimensional electrophoresis technology for proteomics. Proteomics 4(12):3665–3685. https://doi.org/10.1002/pmic.200401031
Bagheri R, Bashir H, Ahmad J, Iqbal M, Qureshi MI (2015) Spinach (Spinacia oleracea L.) modulates its proteome differentially in response to salinity, cadmium and their combination stress. Plant Physiol Biochem 97:235–245. https://doi.org/10.1016/j.plaphy.2015.10.012
Chora S, Starita-Geribaldi M, Guigonis JM, Samson M, Romeo M, Bebianno MJ (2009) Effect of cadmium in the clam Ruditapes decussatus assessed by proteomic analysis. Aquat Toxicol 94(4):300–308. https://doi.org/10.1016/j.aquatox.2009.07.014
Gutsch A, Zouaghi S, Renaut J, Cuypers A, Hausman JF, Sergeant K (2018) Changes in the proteome of medicago sativa leaves in response to long-term cadmium exposure using a cell-wall targeted approach. Int J Mol Sci 19(9):2498–2514. https://doi.org/10.3390/ijms19092498
McDonagh B, Tyther R, Sheehan D (2006) Redox proteomics in the mussel, Mytilus edulis. Mar Environ Res 62(Suppl):S101–S104. https://doi.org/10.1016/j.marenvres.2006.04.001
Zhu JY, Huang HQ, Bao XD, Lin QM, Cai Z (2006) Acute toxicity profile of cadmium revealed by proteomics in brain tissue of Paralichthys olivaceus: potential role of transferrin in cadmium toxicity. Aquat Toxicol 78(2):127–135. https://doi.org/10.1016/j.aquatox.2006.02.010
Li XH, Wu XF, Yue WF, Liu JM, Li GL, Miao YG (2006) Proteomic analysis of the silkworm (Bombyx mori L.) hemolymph during developmental stage. J Proteome Res 5(10):2809–2814. https://doi.org/10.1021/pr0603093
Casanova AG, Vicente-Vicente L, Hernández-Sánchez MT, Prieto M, Rihuete MI, Ramis LM, Del Barco E, Cruz JJ, Ortiz A, Cruz-González I et al (2020) Urinary transferrin pre-emptively identifies the risk of renal damage posed by subclinical tubular alterations. Biomed Pharmacother 121:109684. https://doi.org/10.1016/j.biopha.2019.109684
Maeda H, Sogawa K, Sakaguchi K, Abe S, Sagizaka W, Mochizuki S, Horie W, Watanabe T, Shibata Y, Satoh M et al (2015) Urinary albumin and transferrin as early diagnostic markers of chronic kidney disease. J Vet Med Sci 77(8):937–943. https://doi.org/10.1292/jvms.14-0427
Goudarzi M, Koga T, Khozoie C, Mak TD, Kang BH, Fornace AJ Jr, Peters JM (2013) PPARβ/δ modulates ethanol-induced hepatic effects by decreasing pyridoxal kinase activity. Toxicology 311(3):87–98. https://doi.org/10.1016/j.tox.2013.07.002
Nasr Z, Robert F, Porco JA Jr, Muller WJ, Pelletier J (2013) eIF4F suppression in breast cancer affects maintenance and progression. Oncogene 32(7):861–871. https://doi.org/10.1038/onc.2012.105
Ren W, Guan W, Zhang J, Wang F, Xu G (2019) Pyridoxine 5′-phosphate oxidase is correlated with human breast invasive ductal carcinoma development. Aging (Albany N Y) 11(7):2151–2176. https://doi.org/10.18632/aging.101908
Zheng J, Wang L, Peng Z, Yang Y, Feng D, He J (2017) Low level of PDZ domain containing 1 (PDZK1) predicts poor clinical outcome in patients with clear cell renal cell carcinoma. EBioMedicine 15:62–72. https://doi.org/10.1016/j.ebiom.2016.12.003
Li Y, Zhang M, Dorfman RG, Pan Y, Tang D, Xu L, Zhao Z, Zhou Q, Zhou L, Wang Y, Yin Y, Shen S, Kong B, Friess H, Zhao S, Wang L, Zou X (2018) SIRT2 promotes the migration and invasion of gastric cancer through RAS/ERK/JNK/MMP-9 pathway by increasing PEPCK1-related metabolism. Neoplasia 20(7):745–756. https://doi.org/10.1016/j.neo.2018.03.008
Li Z, Chen Y, Wang X, Zhang H, Zhang Y, Gao Y, Weng M, Wang L, Liang H, Li M, Zhang F, Zhao S, Liu S, Cao Y, Shu Y, Bao R, Zhou J, Liu X, Yan Y, Zhen L, Dong Q, Liu Y (2016) LASP-1 induces proliferation, metastasis and cell cycle arrest at the G2/M phase in gallbladder cancer by down-regulating S100P via the PI3K/AKT pathway. Cancer Lett 372(2):239–250. https://doi.org/10.1016/j.canlet.2016.01.008
Ruan HY, Yang C, Tao XM, He J, Wang T, Wang H, Wang C, Jin GZ, Jin HJ, Qin WX (2017) Downregulation of ACSM3 promotes metastasis and predicts poor prognosis in hepatocellular carcinoma. Am J Cancer Res 7(3):543–553
Satoh M, Koyama H, Kaji T, Kito H, Tohyama C (2002) Perspectives on cadmium toxicity research. Tohoku J Exp Med 196(1):23–32. https://doi.org/10.1620/tjem.196.23
Järup L, Berglund M, Elinder CG, Nordberg G, Vahter M (1998) Health effects of cadmium exposure--a review of the literature and a risk estimate. Scand J Work Environ Health 24(Suppl 1):1–51
Zalups RK, Ahmad S (2003) Molecular handling of cadmium in transporting epithelia. Toxicol Appl Pharmacol 186(3):163–188. https://doi.org/10.1016/s0041-008x(02)00021-2
Borne Y, Fagerberg B, Sallsten G, Hedblad B, Persson M, Melander O, Nilsson J, Orho-Melander M, Barregard L, Engstrom G (2019) Biomarkers of blood cadmium and incidence of cardiovascular events in non-smokers: results from a population-based proteomics study. Clin Proteomics 16:21. https://doi.org/10.1186/s12014-019-9231-7
Satarug S, Vesey DA, Nishijo M, Ruangyuttikarn W, Gobe GC (2019) The inverse association of glomerular function and urinary beta2-MG excretion and its implications for cadmium health risk assessment. Environ Res 173:40–47. https://doi.org/10.1016/j.envres.2019.03.026
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68:167–182. https://doi.org/10.1093/bmb/ldg032
Orr SE, Bridges CC (2017) Chronic kidney disease and exposure to nephrotoxic metals. Int J Mol Sci 18(5):1039–1073. https://doi.org/10.3390/ijms18051039
Monsinjon T, Knigge T (2007) Proteomic applications in ecotoxicology. Proteomics 7(16):2997–3009. https://doi.org/10.1002/pmic.200700101
Shigeta S, Toyoshima M, Kitatani K, Ishibashi M, Usui T, Yaegashi N (2016) Transferrin facilitates the formation of DNA double-strand breaks via transferrin receptor 1: the possible involvement of transferrin in carcinogenesis of high-grade serous ovarian cancer. Oncogene 35(27):3577–3586. https://doi.org/10.1038/onc.2015.425
Howard CM, Bearss N, Subramaniyan B, Tilley A, Sridharan S, Villa N, Fraser CS, Raman D (2019) The CXCR4-LASP1-eIF4F axis promotes translation of oncogenic proteins in triple-negative breast cancer cells. Front Oncol 9:284. https://doi.org/10.3389/fonc.2019.00284
Wolfe AL, Singh K, Zhong Y, Drewe P, Rajasekhar VK, Sanghvi VR, Mavrakis KJ, Jiang M, Roderick JE, Van der Meulen J et al (2014) RNA G-quadruplexes cause eIF4A-dependent oncogene translation in cancer. Nature 513(7516):65–70. https://doi.org/10.1038/nature13485
Tao T, Yang X, Zheng J, Feng D, Qin Q, Shi X, Wang Q, Zhao C, Peng Z, Liu H, Jiang WG, He J (2017) PDZK1 inhibits the development and progression of renal cell carcinoma by suppression of SHP-1 phosphorylation. Oncogene 36(44):6119–6131. https://doi.org/10.1038/onc.2017.199
Avula LR, Chen T, Kovbasnjuk O, Donowitz M (2018) Both NHERF3 and NHERF2 are necessary for multiple aspects of acute regulation of NHE3 by elevated Ca(2+), cGMP, and lysophosphatidic acid. Am J Physiol Gastrointest Liver Physiol 314(1):G81–G90. https://doi.org/10.1152/ajpgi.00140.2017
Cunningham R, Biswas R, Steplock D, Shenolikar S, Weinman E (2010) Role of NHERF and scaffolding proteins in proximal tubule transport. Urol Res 38(4):257–262. https://doi.org/10.1007/s00240-010-0294-1
Weinman EJ, Boddeti A, Cunningham R, Akom M, Wang F, Wang Y, Liu J, Steplock D, Shenolikar S, Wade JB (2003) NHERF-1 is required for renal adaptation to a low-phosphate diet. Am J Physiol Ren Physiol 285(6):F1225–F1232. https://doi.org/10.1152/ajprenal.00215.2003
Gu J, Dai S, Liu Y, Liu H, Zhang Y, Ji X, Yu F, Zhou Y, Chen L, Tse WKF, Wong CKC, Chen B, Shi H (2018) Activation of Ca(2+)-sensing receptor as a protective pathway to reduce cadmium-induced cytotoxicity in renal proximal tubular cells. Sci Rep 8(1):1092. https://doi.org/10.1038/s41598-018-19327-9
Wang L, Cao J, Chen D, Liu X, Lu H, Liu Z (2009) Role of oxidative stress, apoptosis, and intracellular homeostasis in primary cultures of rat proximal tubular cells exposed to cadmium. Biol Trace Elem Res 127(1):53–68. https://doi.org/10.1007/s12011-008-8223-7
Wang L, Lin SQ, He YL, Liu G, Wang ZY (2013) Protective effects of quercetin on cadmium-induced cytotoxicity in primary cultures of rat proximal tubular cells. Biomed Environ Sci 26(4):258–267. https://doi.org/10.3967/0895-3988.2013.04.004
Johri N, Jacquillet G, Unwin R (2010) Heavy metal poisoning: the effects of cadmium on the kidney. BioMetals 23(5):783–792. https://doi.org/10.1007/s10534-010-9328-y
Wallin M, Sallsten G, Fabricius-Lagging E, Öhrn C, Lundh T, Barregard L (2013) Kidney cadmium levels and associations with urinary calcium and bone mineral density: a cross-sectional study in Sweden. Environ Health 12:22. https://doi.org/10.1186/1476-069x-12-22
Xu MY, Wang P, Sun YJ, Wu YJ (2019) Disruption of kidney metabolism in rats after subchronic combined exposure to low-dose cadmium and chlorpyrifos. Chem Res Toxicol 32(1):122–129. https://doi.org/10.1021/acs.chemrestox.8b00219
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, Campbell LL, Polyak K, Brisken C, Yang J, Weinberg RA (2008) The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133(4):704–715. https://doi.org/10.1016/j.cell.2008.03.027
Wei Z, Shan Z, Shaikh ZA (2018) Epithelial-mesenchymal transition in breast epithelial cells treated with cadmium and the role of snail. Toxicol Appl Pharmacol 344:46–55. https://doi.org/10.1016/j.taap.2018.02.022
Shan Z, Wei Z, Shaikh ZA (2018) Suppression of ferroportin expression by cadmium stimulates proliferation, EMT, and migration in triple-negative breast cancer cells. Toxicol Appl Pharmacol 356:36–43. https://doi.org/10.1016/j.taap.2018.07.017
Luo H, Li Z, Ge H, Mei D, Zhao L, Jiang L, Geng C, Li Q, Yao X, Cao J (2017) HMGA2 upregulation mediates Cd-induced migration and invasion in A549 cells and in lung tissues of mice. Chem Biol Interact 277:1–7. https://doi.org/10.1016/j.cbi.2017.08.012
Mitruţ R, Stepan AE, Mărgăritescu C, Andreiana BC, Kesse AM, Simionescu CE, Militaru C (2019) Immunoexpression of MMP-8, MMP-9 and TIMP-2 in dilated cardiomyopathy. Romanian J Morphol Embryol 60(1):119–124
Lian S, Xia Y, Khoi PN, Ung TT, Yoon HJ, Kim NH, Kim KK, Jung YD (2015) Cadmium induces matrix metalloproteinase-9 expression via ROS-dependent EGFR, NF-small ka, CyrillicB, and AP-1 pathways in human endothelial cells. Toxicology 338:104–116. https://doi.org/10.1016/j.tox.2015.10.008
Zhang JF, Wang P, Yan YJ, Li Y, Guan MW, Yu JJ, Wang XD (2017) IL33 enhances glioma cell migration and invasion by upregulation of MMP2 and MMP9 via the ST2-NF-kappaB pathway. Oncol Rep 38(4):2033–2042. https://doi.org/10.3892/or.2017.5926
Acknowledgments
We thank the animal house staff for their care of the animals, and Ms. Ye Pan for technical assistance with the 2D gel analysis.
Funding
This work was supported by the National Natural Science Foundation of China (31600952 and 31271272) and the Start-Up Research Funding of Jiangsu University for Distinguished Scholars (5501330001).
Author information
Authors and Affiliations
Contributions
XS, YW, TJ, and XY performed the research; JG and HS designed the research study and contributed essential reagents or tools; XS, YW, TJ, XY, ZR, HL, YZ, JG, and HS analyzed and interpreted the data; XS, YW, TJ, XY, AT, JG, and HS wrote the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, X., Wang, Y., Jiang, T. et al. Nephrotoxicity Profile of Cadmium Revealed by Proteomics in Mouse Kidney. Biol Trace Elem Res 199, 1929–1940 (2021). https://doi.org/10.1007/s12011-020-02312-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02312-7