Abstract
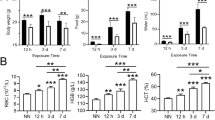
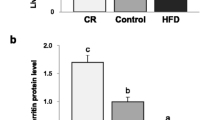
The liver is contributed to maintaining body iron homeostasis and controlling of body adaptation to fasting. Although previous studies implied a negative relationship between iron and ghrelin in both mice and humans, it remains to be explored whether fasting or ghrelin has a functional effect on iron homeostasis in the liver. In this study, we examined the roles of fasting and ghrelin in modulating the protein expression of Fpn1, transferrin receptor 1 (TfR1), and ferritin light chain (Ft-L), as well as the mRNA expression of ghrelin, hepcidin, ghrelin O-acyltransferase (GOAT), and growth hormone secretagogue receptor 1 alpha (GHSR1α) in mouse liver and cultured hepatocytes. Our in vivo results suggested that fasting significantly upregulated the mRNA expression of ghrelin, GOAT, and GHSR1α, as well as the protein levels of ghrelin, Fpn1, and Ft-L, but not TfR1, in mouse liver. Interestingly, mRNA expression of hepcidin did not change significantly after fasting. Meanwhile, in cultured hepatocytes, ghrelin significantly increased the protein expression of Fpn1 but not Ft-L and TfR1 and significantly enhanced ERK phosphorylation. Furthermore, the pretreatment of cultured hepatocytes with either a pERK inhibitor or a GHSR1α antagonist abolished the effects of ghrelin on Fpn1 expression and ERK phosphorylation. Our findings confirmed that fasting increases iron export in the liver by upregulating Fpn1 expression through the ghrelin/GHSR1α/MAPK signaling pathway.







Similar content being viewed by others
Data Availability
All original data are available upon request.
Abbreviations
- Fpn1:
-
Ferroportin 1
- TfR1:
-
Transferrin receptor 1
- Ft-L:
-
Ferritin-L
- GOAT:
-
Ghrelin O-acyltransferase
- GHSR1α:
-
Growth hormone secretagogue receptor 1α
- MAPK:
-
Mitogen-activated protein kinase
- ERK:
-
Extracellular-regulated protein kinase
References
Sikorska K, Bernat A, Wroblewska A (2016) Molecular pathogenesis and clinical consequences of iron overload in liver cirrhosis. Hepatobiliary Pancreat Dis Int 15(5):461–479
Rommelaere S, Millet V, Vu Manh TP, Gensollen T, Andreoletti P, Cherkaoui-Malki M, Bourges C, Escaliere B, Du X, Xia Y, Imbert J, Beutler B, Kanai Y, Malissen B, Malissen M, Tailleux A, Staels B, Galland F, Naquet P (2014) Sox17 regulates liver lipid metabolism and adaptation to fasting. PLoS One 9(8):e104925. https://doi.org/10.1371/journal.pone.0104925
de Vries EM, Eggels L, van Beeren HC, Ackermans MT, Kalsbeek A, Fliers E, Boelen A (2014) Fasting-induced changes in hepatic thyroid hormone metabolism in male rats are independent of autonomic nervous input to the liver. Endocrinology 155(12):5033–5041. https://doi.org/10.1210/en.2014-1608
Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K (1999) Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402(6762):656–660. https://doi.org/10.1038/45230
Goldstone AP, Prechtl CG, Scholtz S, Miras AD, Chhina N, Durighel G, Deliran SS, Beckmann C, Ghatei MA, Ashby DR, Waldman AD, Gaylinn BD, Thorner MO, Frost GS, Bloom SR, Bell JD (2014) Ghrelin mimics fasting to enhance human hedonic, orbitofrontal cortex, and hippocampal responses to food. Am J Clin Nutr 99(6):1319–1330. https://doi.org/10.3945/ajcn.113.075291
Patterson M, Bloom SR, Gardiner JV (2011) Ghrelin and appetite control in humans--potential application in the treatment of obesity. Peptides 32(11):2290–2294. https://doi.org/10.1016/j.peptides.2011.07.021
Davies JS, Kotokorpi P, Eccles SR, Barnes SK, Tokarczuk PF, Allen SK, Whitworth HS, Guschina IA, Evans BA, Mode A, Zigman JM, Wells T (2009) Ghrelin induces abdominal obesity via GHS-R-dependent lipid retention. Mol Endocrinol 23(6):914–924. https://doi.org/10.1210/me.2008-0432
Lv Y, Liang T, Wang G, Li Z (2018) Ghrelin, a gastrointestinal hormone, regulates energy balance and lipid metabolism. Biosci Rep 38(5). https://doi.org/10.1042/BSR20181061
Castaneda TR, Tong J, Datta R, Culler M, Tschop MH (2010) Ghrelin in the regulation of body weight and metabolism. Front Neuroendocrinol 31(1):44–60. https://doi.org/10.1016/j.yfrne.2009.10.008
Wortley KE, Anderson KD, Garcia K, Murray JD, Malinova L, Liu R, Moncrieffe M, Thabet K, Cox HJ, Yancopoulos GD, Wiegand SJ, Sleeman MW (2004) Genetic deletion of ghrelin does not decrease food intake but influences metabolic fuel preference. Proc Natl Acad Sci U S A 101(21):8227–8232. https://doi.org/10.1073/pnas.0402763101
Kojima M, Hamamoto A, Sato T (2016) Ghrelin O-acyltransferase (GOAT), a specific enzyme that modifies ghrelin with a medium-chain fatty acid. J Biochem 160(4):189–194. https://doi.org/10.1093/jb/mvw046
Taylor MS, Hwang Y, Hsiao PY, Boeke JD, Cole PA (2012) Ghrelin O-acyltransferase assays and inhibition. Methods Enzymol 514:205–228. https://doi.org/10.1016/B978-0-12-381272-8.00013-1
Kang K, Schmahl J, Lee JM, Garcia K, Patil K, Chen A, Keene M, Murphy A, Sleeman MW (2012) Mouse ghrelin-O-acyltransferase (GOAT) plays a critical role in bile acid reabsorption. FASEB J 26(1):259–271. https://doi.org/10.1096/fj.11-191460
Cummings DE, Frayo RS, Marmonier C, Aubert R, Chapelot D (2004) Plasma ghrelin levels and hunger scores in humans initiating meals voluntarily without time- and food-related cues. Am J Phys Endocrinol Metab 287(2):E297–E304. https://doi.org/10.1152/ajpendo.00582.2003
Kirsz K, Zieba DA (2011) Ghrelin-mediated appetite regulation in the central nervous system. Peptides 32(11):2256–2264. https://doi.org/10.1016/j.peptides.2011.04.010
Hosoda H, Kojima M, Kangawa K (2002) Ghrelin and the regulation of food intake and energy balance. Mol Interv 2(8):494–503. https://doi.org/10.1124/mi.2.8.494
Akarsu S, Ustundag B, Gurgoze MK, Sen Y, Aygun AD (2007) Plasma ghrelin levels in various stages of development of iron deficiency anemia. J Pediatr Hematol Oncol 29(6):384–387. https://doi.org/10.1097/MPH.0b013e3180645170
Luke B (2005) Nutrition and multiple gestation. Semin Perinatol 29(5):349–354. https://doi.org/10.1053/j.semperi.2005.08.004
Natalucci G, Riedl S, Gleiss A, Zidek T, Frisch H (2005) Spontaneous 24-h ghrelin secretion pattern in fasting subjects: maintenance of a meal-related pattern. Eur J Endocrinol 152(6):845–850. https://doi.org/10.1530/eje.1.01919
Kucuk N, Orbak Z, Karakelloglu C, Akcay F (2019) The effect of therapy on plasma ghrelin and leptin levels, and appetite in children with iron deficiency anemia. J Pediatr Endocrinol Metab 32(3):275–280. https://doi.org/10.1515/jpem-2018-0352
Isguven P, Arslanoglu I, Erol M, Yildiz M, Adal E, Erguven M (2007) Serum levels of ghrelin, leptin, IGF-I, IGFBP-3, insulin, thyroid hormones and cortisol in prepubertal children with iron deficiency. Endocr J 54(6):985–990
Isguven P, Arslanoglu I, Erol M, Yildiz M, Adal E, Erguven M (2007) Serum levels of ghrelin, leptin, thyroid hormones and cortisol iron deficiency IGF-I, IGFBP-3, insulin, in prepubertal children with. Endocr J 54(6):985–990. https://doi.org/10.1507/endocrj.K07-031
Dogan A, Alioglu B, Dindar N, Dallar Y (2013) Increased serum hepcidin and ghrelin levels in children treated for iron deficiency anemia. J Clin Lab Anal 27(1):81–85. https://doi.org/10.1002/jcla.21566
Luo QQ, Zhou G, Huang SN, Mu MD, Chen YJ, Qian ZM (2018) Ghrelin is negatively correlated with iron in the serum in human and mice. Ann Nutr Metab 72(1):37–42. https://doi.org/10.1159/000484698
Rao J, Zhang C, Wang P, Lu L, Qian X, Qin J, Pan X, Li G, Wang X, Zhang F (2015) C/EBP homologous protein (CHOP) contributes to hepatocyte death via the promotion of ERO1alpha signalling in acute liver failure. Biochem J 466(2):369–378. https://doi.org/10.1042/BJ20140412
Luo QQ, Zhou YF, Chen MY, Liu L, Ma J, Zhang MW, Zhang FL, Ke Y, Qian ZM (2018) Fasting up-regulates ferroportin 1 expression via a ghrelin/GHSR/MAPK signaling pathway. J Cell Physiol 233(1):30–37. https://doi.org/10.1002/jcp.25931
Luo QQ, Wang D, Yu MY, Zhu L (2011) Effect of hypoxia on the expression of iron regulatory proteins 1 and the mechanisms involved. IUBMB Life 63(2):120–128. https://doi.org/10.1002/iub.419
Page AJ, Slattery JA, Milte C, Laker R, O’Donnell T, Dorian C, Brierley SM, Blackshaw LA (2007) Ghrelin selectively reduces mechanosensitivity of upper gastrointestinal vagal afferents. Am J Physiol Gastrointest Liver Physiol 292(5):G1376–G1384. https://doi.org/10.1152/ajpgi.00536.2006
Chan CB, Cheng CH (2004) Identification and functional characterization of two alternatively spliced growth hormone secretagogue receptor transcripts from the pituitary of black seabream Acanthopagrus schlegeli. Mol Cell Endocrinol 214(1–2):81–95. https://doi.org/10.1016/j.mce.2003.11.020
Wang JH, Li HZ, Shao XX, Nie WH, Liu YL, Xu ZG, Guo ZY (2019) Identifying the binding mechanism of LEAP2 to receptor GHSR1a. FEBS J 286(7):1332–1345. https://doi.org/10.1111/febs.14763
Xue Q, Bai B, Ji B, Chen X, Wang C, Wang P, Yang C, Zhang R, Jiang Y, Pan Y, Cheng B, Chen J (2018) Ghrelin through GHSR1a and OX1R heterodimers reveals a Galphas-cAMP-cAMP response element binding protein signaling pathway in vitro. Front Mol Neurosci 11:245. https://doi.org/10.3389/fnmol.2018.00245
Schrottke S, Kaiser A, Vortmeier G, Els-Heindl S, Worm D, Bosse M, Schmidt P, Scheidt HA, Beck-Sickinger AG, Huster D (2017) Expression, functional characterization, and solid-state NMR investigation of the G protein-coupled GHS receptor in bilayer membranes. Sci Rep 7:46128. https://doi.org/10.1038/srep46128
Nakata S, Yoshino Y, Okita M, Kawabe K, Yamazaki K, Ozaki Y, Mori Y, Ochi S, Iga JI, Ueno SI (2019) Differential expression of the ghrelin-related mRNAs GHS-R1a, GHS-R1b, and MBOAT4 in Japanese patients with schizophrenia. Psychiatry Res 272:334–339. https://doi.org/10.1016/j.psychres.2018.12.135
Gnanapavan S, Kola B, Bustin SA, Morris DG, McGee P, Fairclough P, Bhattacharya S, Carpenter R, Grossman AB, Korbonits M (2002) The tissue distribution of the mRNA of ghrelin and subtypes of its receptor, GHS-R, in humans. J Clin Endocrinol Metab 87(6):2988. https://doi.org/10.1210/jcem.87.6.8739
Anderson ER, Shah YM (2013) Iron homeostasis in the liver. Compr Physiol 3(1):315–330. https://doi.org/10.1002/cphy.c120016
Harrison PM, Arosio P (1996) The ferritins: molecular properties, iron storage function and cellular regulation. Biochim Biophys Acta 1275(3):161–203
Nemeth E, Tuttle MS, Powelson J, Vaughn MB, Donovan A, Ward DM, Ganz T, Kaplan J (2004) Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306(5704):2090–2093. https://doi.org/10.1126/science.1104742
Wallace DF, McDonald CJ, Ostini L, Iser D, Tuckfield A, Subramaniam VN (2017) The dynamics of hepcidin-ferroportin internalization and consequences of a novel ferroportin disease mutation. Am J Hematol 92(10):1052–1061. https://doi.org/10.1002/ajh.24844
Waseem T, Duxbury M, Ashley SW, Robinson MK (2014) Ghrelin promotes intestinal epithelial cell proliferation through PI3K/Akt pathway and EGFR trans-activation both converging to ERK 1/2 phosphorylation. Peptides 52:113–121. https://doi.org/10.1016/j.peptides.2013.11.021
Mazzocchi G, Neri G, Rucinski M, Rebuffat P, Spinazzi R, Malendowicz LK, Nussdorfer GG (2004) Ghrelin enhances the growth of cultured human adrenal zona glomerulosa cells by exerting MAPK-mediated proliferogenic and antiapoptotic effects. Peptides 25(8):1269–1277. https://doi.org/10.1016/j.peptides.2004.05.011
Funding
This study was funded by the Natural Science Foundation of China (Grants 81873924, 81471257, and 31300973); the Open Cooperation Program from Key Laboratory of Extreme Environmental Medicine, Ministry of Education (KL2019GY011); the Natural Science Foundation of Jiangsu Province in China (Grant BK20161283); and also sponsored by the Nantong Science and Technology Project (MS12018030, MS12018048).
Author information
Authors and Affiliations
Contributions
G.H.W. and L.Z. conceived, organized, and supervised the work; Q.Q.L., J.N.H., and G.Y. performed the experiments; X.Y.Y., Z.P.C., D.W., and Y.P.L. contributed to the analysis of data; Q.Q.L. and G.H.W. prepared, wrote, and revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interests
The authors declare that they have no conflicts of interest.
Ethics Approval and Consent to Participate
All animal handling procedures were conducted in accordance with guidelines approved by the Laboratory Animal Ethics Committee of Nantong University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luo, Q., Hu, J., Yang, G. et al. Fasting Increases Iron Export by Modulating Ferroportin 1 Expression Through the Ghrelin/GHSR1α/MAPK Pathway in the Liver. Biol Trace Elem Res 199, 267–277 (2021). https://doi.org/10.1007/s12011-020-02114-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02114-x