Abstract
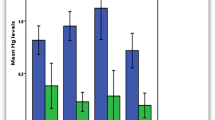
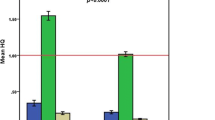
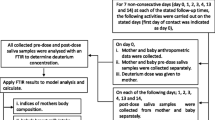
The objective of this work was to assess exposure to mercury (Hg) and its induction of oxidative stress in 155 healthy lactating Saudi mothers and their infants. Samples of breast milk and blood were collected from the mothers, while urine was taken from both infants and mothers. Both urinary 8-hydroxy-2′-deoxyguanosine (8-OHdG) and malondialdehyde (MDA) were measured in mothers and infants as biomarkers of oxidative stress. The mean concentration of Hg in breast milk was 1.19 μg/L (range 0.012–6.44 μg/L) with only one mother having Hg >4 μg/L, the upper limit established by the US Agency for Toxic Substance and Disease Registry. However, 57.4 % had Hg ≥1 μg/L, the background level for Hg in human milk. The mean urinary Hg corrected for creatinine (Hg-C) in mothers and infants was 1.47 and 7.90 μg/g creatinine, respectively, with a significant correlation between the two (p < 0.001). Urinary Hg levels over 5 μg/g creatinine (the background level in an unexposed population) were found in 3.3 % of mothers and 50.1 % of infants. None of the mothers had total blood Hg above the US Environmental Protection Agency’s maximum reference dose of 5.8 μg/L. No correlation was noted between urinary Hg in infants and Hg in breast milk (p > 0.05). Hg in breast milk, though, was associated with Hg in blood (p < 0.001), suggesting the efficient transfer of Hg from blood to milk. Hg in the breast milk of mothers and in the urine of infants affected the excretion of urinary MDA and 8-OHdG, respectively, in a dose-related manner. These findings reveal for the first time lactational exposure to Hg-induced oxidative stress in breast-fed infants, which may play a role in pathogenesis, particularly during neurodevelopment. This will also contribute to the debate over the benefits of breast milk versus the adverse effects of exposure to pollutants. Nevertheless, breastfeeding should not be discouraged, but efforts should be made to identify and eliminate the source of Hg exposure in the population.

Similar content being viewed by others
References
World Health Organization, WHO (1990) Methylmercury. In: Environmental health criteria 101. WHO, Geneva
World Health Organization, WHO (2003) CICAD 50. Elemental mercury and inorganic mercury compounds: human health aspects. IPCS, World Health Organization, Geneva, Switzerland
Chan TY (2011) Inorganic mercury poisoning associated with skin-lightening cosmetic products. Clin Toxicol (Phila) 49:886–891
Clarkson TW, Magos L (2006) The toxicology of mercury and its chemical compounds. Crit Rev Toxicol 36:609–662
Dorea JG (2004) Mercury and lead during breast-feeding. Br J Nutr 92:21–40
Stein J, Schettler T, Wallinga D, Valenti M (2002) In harm’s way: toxic threats to child development. J Dev Behav Pediatr 23(1 Suppl):S13–S22
Daston G, Faustman E, Ginsberg G, Fenner-Crisp P, Olin S, Sonawane B, Bruckner J, Breslin W, McLaughlin TJ (2004) A framework for assessing risks to children from exposure to environmental agents. Environ Health Perspect 112:238–256
Valko M, Morris H, Cronin MT (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12:1161–1208
Ercal N, Gurer-Orhan H, Aykin-Burns N (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1:529–539
Gobe G, Crane D (2010) Mitochondria, reactive oxygen species and cadmium toxicity in the kidney. Toxicol Lett 198:49–55
Roberts RA, Smith RA, Safe S, Szabo C, Tjalkens RB, Robertson FM (2010) Toxicological and pathophysiological roles of reactive oxygen and nitrogen species. Toxicology 276:85–94
Hwang ES, Kim GH (2007) Biomarkers for oxidative stress status of DNA, lipids, and proteins in vitro and in vivo cancer research. Toxicology 229:1–10
Valavanidis A, Vlachogianni T, Fiotakis C (2009) 8-hydroxy-2′-deoxyguanosine (8-OHdG): a critical biomarker of oxidative stress and carcinogenesis. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 27:120–139
Del Rio D, Stewart AJ, Pellegrini N (2005) A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr Metab Cardiovasc Dis 15:316–228
Crespo-López ME, Macêdo GL, Pereira SI, Arrifano GP, Picanço-Diniz DL, do Nascimento JL, Herculano AM (2009) Mercury and human genotoxicity: critical considerations and possible molecular mechanisms. Pharmacol Res 60:212–220
Ceccatelli S, Daré E, Moors M (2010) Methylmercury-induced neurotoxicity and apoptosis. Chem Biol Interact 188:301–308
Houston MC (2011) Role of mercury toxicity in hypertension, cardiovascular disease, and stroke. J Clin Hypertens (Greenwich) 13:621–627
Al-Saleh I, Al anoud Al-Sedairi A, Elkhatib R (2012) Effect of mercury (Hg) dental amalgam fillings on renal and oxidative stress biomarkers in children. Sci Total Environ 431:188–196
Asikainen TM, Raivio KO, Saksela M, Kinnula VL (1998) Expression and developmental profile of antioxidant enzymes in human lung and liver. Am J Respir Cell Mol Biol 19:942–949
Auten RL, Davis JM (2009) Oxygen toxicity and reactive oxygen species: the devil is in the details. Pediatr Res 66:121–127
Jensen AA (1991) Levels and trends of environmental chemicals in human milk. In: Jensen AA, Slorach SA (eds) Chemical contaminants in human milk. CRC, Boca Raton, pp 45–198
Al-Saleh I, Al-Doush I (1997) Mercury content in skin-lightening creams and potential hazards to the health of Saudi Women. J Toxicol Environ Health 51:123–130
Food and Drug Administration, FDA (1992) FDA’s cosmetics handbook. U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration
Al-Saleh I, Shinwari N, Mashhour A, Mohamed G, Rabah A (2011) Heavy metals (lead, cadmium and mercury) in maternal, cord blood and placenta of healthy women. Int J Hyg Environ Health 214:79–101
Wilhelm M, Ewers U, Schulz C (2004) Revised and new reference values for some trace elements in blood and urine for human biomonitoring in environmental medicine. Int J Hyg Environ Health 207:69–73
Sly PD, Flack F (2008) Susceptibility of children to environmental pollutants. Ann NY Acad Sci 1140:163–183
Castoldi AF, Johansson C, Onishchenko N, Coccini T, Roda E, Vahter M, Ceccatelli S, Manzo L (2008) Human developmental neurotoxicity of methylmercury: Impact of variables and risk modifiers. Reg Toxicol Pharmacol 51:201–214
Karagas MR, Choi AL, Oken E, Horvat M, Schoeny R, Kamai E, Cowell W, Grandjean P, Korrick S (2012) Evidence on the human health effects of low-level methylmercury exposure. Environ Health Perspect 120:799–806
Myers GJ, Thurston SW, Pearson AT, Davidson PW, Cox C, Shamlaye CF, Cernichiari E, Clarkson TW (2009) Postnatal exposure to methyl mercury from fish consumption: a review and new data from the Seychelles Child Development Study. Neurotoxicology 30:338–349
Al-Saleh I, Al-Sedairi A (2011) Mercury (Hg) burden in children: the impact of dental amalgam. Sci Total Environ 409:3003–3015
Fukunaga K, Yoshida M, Nakazona N (1998) A simple, rapid, highly sensitive and reproducible quantitation method for plasma malonialdehyde by high-performance liquid chromatography. Biomed Chromatogr 12:300–303
Agency for Toxic Substances and Disease Registry, ATSDR (1999) Report no.: CAS# 7439-97-6. http://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=115&tid=24. Accessed 20 Jan 2013
Al-Saleh I, Shinwari N, Mashhour A (2003) Heavy metals concentrations in the breast milk of Saudi women. Biol Trace Elem Res 96:21–38
da Cunha LR, da Costa TH, Caldas ED (2013) Mercury concentration in breast milk and infant exposure assessment during the first 90 days of lactation in a midwestern region of Brazil. Biol Trace Elem Res 151:30–37
Orün E, Yalçin SS, Aykut O, Orhan G, Koç-Morgil G, Yurdakök K, Uzun R (2012) Mercury exposure via breast-milk in infants from a suburban area of Ankara, Turkey. Turk J Pediatr 54:136–143
Goudarzi MA, Parsaei P, Nayebpour F, Rahimi E (2012) Determination of mercury, cadmium and lead in human milk in Iran. Toxicol Ind Health. doi:10.1177/0748233712445047. PubMed PMID: 22534496
García-Esquinas E, Pérez-Gómez B, Fernández MA, Pérez-Meixeira AM, Gil E, de Paz C, Iriso A, Sanz JC, Astray J, Cisneros M, de Santos A, Asensio A, García-Sagredo JM, García JF, Vioque J, Pollán M, López-Abente G, González MJ, Martínez M, Bohigas PA, Pastor R, Aragonés N (2011) Mercury, lead and cadmium in human milk in relation to diet, lifestyle habits and sociodemographic variables in Madrid (Spain). Chemosphere 85:268–276
Jensen AA (1983) Chemical contaminants in human milk. Residue Rev 89:1–127
Whitehead RG, Paul AA (2000) Long-term adequacy of exclusive breast-feeding: how scientific research has led to revised opinions. Proc Nutr Soc 59:17–23
United States Environmental Protection Agency, US EPA (1997) Mercury study report to congress. Volume v: health effects of mercury and mercury compounds. Office of Air Quality Planning and Standards and Office of Research and Development, Washington, DC. http://www.epa.gov/ttn/atw/112nmerc/volume5.pdf. Accessed 20 Jan 2013
Boischio AA, Henshel D (2000) Fish consumption, fish lore, and mercury pollution-risk communication for the Madeira River people. Environ Res 84:108–126
United States Environmental Protection Agency, US EPA (2007) Mercury. Human exposure. http://www.epa.gov/mercury/exposure.htm. Accessed 20 Jan 2013
McKelvey W, Jeffery N, Clark N, Kass D, Parsons PJ (2011) Population-based inorganic mercury biomonitoring and the identification of skin care products as a source of exposure in New York City. Environ Health Perspect 119:203–209
Watson GE, Evans K, Thurston SW, van Wijngaarden E, Wallace JM, McSorley EM, Bonham MP, Mulhern MS, McAfee AJ, Davidson PW, Shamlaye CF, Strain JJ, Love T, Zareba G, Myers GJ (2012) Prenatal exposure to dental amalgam in the Seychelles Child Development Nutrition Study: associations with neurodevelopmental outcomes at 9 and 30 months. Neurotoxicology 33:1511–1517
Valent F, Horvat M, Sofianou-Katsoulis A, Spiric Z, Mazej D, Little D, Prasouli A, Mariuz M, Tamburlini G, Nakou S, Barbone F (2013) Neurodevelopmental effects of low-level prenatal mercury exposure from maternal fish consumption in a Mediterranean cohort: study rationale and design. J Epidemiol 23:146–152
Sundberg J, Jönsson S, Karlsson MO, Hallén IP, Oskarsson A (1998) Kinetics of methylmercury and inorganic mercury in lactating and nonlactating mice. Toxicol Appl Pharmacol 151:319–329
Björnberg KA, Vahter M, Berglund B, Niklasson B, Blennow M, Sandborgh-Englund G (2005) Transport of methylmercury and inorganic mercury to the fetus and breast-fed infant. Environ Health Perspect 113:1381–1385
Vahter M, Akesson A, Lind B, Björs U, Schütz A, Berglund M (2000) Longitudinal study of methylmercury and inorganic mercury in blood and urine of pregnant and lactating women, as well as in umbilical cord blood. Environ Res 84:186–194
Wong YT, Ruan R, Tay FE (2006) Relationship between levels of oxidative DNA damage, lipid peroxidation and mitochondrial membrane potential in young and old F344 rats. Free Radic Res 40:393–402
Canakci CF, Cicek Y, Yildirim A, Sezer U, Canakci V (2009) Increased levels of 8-hydroxydeoxyguanosine and malondialdehyde and its relationship with antioxidant enzymes in saliva of periodontitis patients. Eur J Dent 3:100–106
Marnett LJ (1999) Lipid peroxidation—DNA damage by malondialdehyde. Mutat Res 424:83–95
Park JW, Floyd RA (1992) Lipid peroxidation products mediate the formation of 8-hydroxydeoxyguanosine in DNA. Free Radic Biol Med 12:245–250
Mead MN (2008) Contaminants in human milk: weighing the risks against the benefits of breastfeeding. Environ Health Perspect 116:A427–A434
Section on Breastfeeding (2012) Breastfeeding and the use of human milk. Pediatrics 129:29(3):e827–841
Acknowledgments
The investigators thank King Abdulaziz City for Science and Technology (ARP-29-23) for funding this study. We would like to thank all the women who participated in this study and the staff of primary health-care units in Riyadh.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Saleh, I., Abduljabbar, M., Al-Rouqi, R. et al. Mercury (Hg) Exposure in Breast-Fed Infants and Their Mothers and the Evidence of Oxidative Stress. Biol Trace Elem Res 153, 145–154 (2013). https://doi.org/10.1007/s12011-013-9687-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-013-9687-7