Abstract
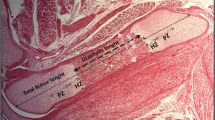
Zinc (Zn) deficiency during pregnancy may result in a variety of defects in the offspring. We evaluated the influence of marginal Zn deficiency during pregnancy on neonatal bone status. Nine-week-old male Sprague-Dawley rats were divided into two groups and fed AIN-93G-based experimental diets containing 35 mg Zn/kg (Zn adequately supplied, N) or 7 mg Zn/kg (low level of Zn, L) from 14-day preconception to 20 days of gestation, that is, 1 day before normal delivery. Neonates were delivered by cesarean section. Litter size and neonate weight were not different between the two groups. However, in the L-diet-fed dam group, bone matrix formation in isolated neonatal calvaria culture was clearly impaired and was not recovered by the addition of Zn into the culture media. Additionally, serum concentration of osteocalcin, as a bone formation parameter, was lower in neonates from the L-diet-fed dam group. Impaired bone mineralization was observed with a significantly lower content of phosphorus in neonate femurs from L-diet-fed dams compared with those from N-diet-fed dams. Moreover, Zn content in the femur and calvaria of neonates from the L-diet group was lower than that of the N-diet-fed group. In the marginally Zn-deficient dams, femoral Zn content, serum concentrations of Zn, and osteocalcin were reduced when compared with control dams. We conclude that maternal Zn deficiency causes impairment of bone matrix formation and bone mineralization in neonates, implying the importance of Zn intake during pregnancy for proper bone development of offspring.

Similar content being viewed by others
References
Prasad AS (1991) Discovery of human zinc deficiency and studies in an experimental human model. Am J Clin Nutr 53:403–412
Rossi L, Migliaccio S, Corsi A et al (2001) Reduced growth and skeletal changes in zinc-deficient growing rats are due to impaired growth plate activity and inanition. J Nutr 131:1142–1146
McClung JP, Stahl CH, Marchitelli LJ et al (2006) Effects of dietary phytase on body weight gain, body composition and bone strength in growing rats fed a low-zinc diet. J Nutr Biochem 17:190–196
Diamond I, Hurley LS (1970) Histopathology of zinc-deficient fetal rats. J Nutr 100:325–329
Yamaguchi M, Matsui T (1996) Stimulatory effect of zinc-chelating dipeptide on deoxyribonucleic acid synthesis in osteoblastic MC3T3-E1 cells. Peptides 17:1207–1211
Domenech E, Diaz-Gomez NM, Barroso F et al (2001) Zinc and perinatal growth. Early Hum Dev 65 Suppl:S111–S117
Angus RM, Sambrook PN, Pocock NA et al (1998) Dietary intake and bone mineral density. Bone Miner 4:265–277
Yamaguchi M, Oishi H, Suketa Y (1987) Stimulatory effects of zinc on bone matrix formation in tissue culture. Biochem Pharmacol 36:4007–4012
Hall SL, Dimai HP, Farley JR (1996) Effects of zinc human skeletal alkaline phosphatase activity in vitro. Calcif Tissue Int 64:163–172
Christian P, West KP Jr (1998) Interactions between zinc and vitamin A: an update. Am J Clin Nutr 68 Suppl:435S–441S
Merialdi M, Caufield LE, Zavaleta N et al (2004) Randomized controlled trial of prenatal zinc supplementation and fetal bone growth. Am J Clin Nutr 79:826–830
Leek JC, Vogler JB, Gershwin ME et al (1984) Studies on marginal zinc deprivation in rhesus monkeys. V. Fetal and infant skeletal effects. Am J Clin Nutr 40:1203–1212
Reeves PG, Nielsen FH, Fahey GC Jr (1993) AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr 123:1939–1951
Garrett IR (2003) Assessing bone matrix formation using mouse calvarial organ culture. In: Helfrich MP, Ralston SH (eds) Bone research protocols. Humana, Totowa, pp 183–200
Hapon MB, Simoncini M, Via G et al (2003) Effect of hypothyroidism on hormone profiles in virgin, pregnant and lactating rats, and on lactation. Reproduction 126:371–382
McConnell SD, Henkin RI (1974) Altered preference for sodium chloride, anorexia, and changes in plasma and urinary zinc in rats fed a zinc-deficient diet. J Nutr 104:1108–1114
Shay NF, Mangian HF (2000) Neurobiology of zinc-influenced eating behavior. J Nutr 130:1493S–1499S
Favus MJ, Christakos S (eds) (2003) Primer on the metabolic bone diseases and disorders of mineral metabolism, 5th edn. American Society for Bone and Mineral Research, Washington
Stracher BC, Hill CH, Madaras JG (1980) Effect of zinc deficiency on bone collagenase and collagen turnover. J Nutr 110:2095–2102
Tomat AL, Inserra F, Veiras L et al (2008) Moderate zinc restriction during fetal and postnatal growth of rats: effects on adult arterial blood pressure and kidney. Am J Physiol Regul Integr Comp Physiol 295:R543–R549
Hosea HJ, Taylor CG, Wood T et al (2004) Zinc-deficient rats have more limited bone recovery during repletion than diet-restricted rats. Exp Biol Med Maywood 229:303–311
Lowe NM, Woodhouse LR, Wee J et al (1999) Short-term zinc kinetics in pregnant rats fed marginal zinc diets. J Nutr 129:1020–1025
Fairweather-Tait SJ, Wright AJA, Cooke J et al (1984) Studies of zinc metabolism in pregnant and lactating rats. Br J Nutr 54:401–413
Hotz C, Lowe NM, Araya M, Brown KH (2003) Assessment of the trace element status of individuals and populations: the example of zinc and copper. J Nutr 133:1563S–1568S
Acknowledgments
We thank Dr. K. Nakayama (Kyoto Institute of Nutrition and Pathology Inc.) for helping with the histological technique and M. Yamada (Meiji Dairies Corp.) for giving technical assistance. This research was supported by Meiji Dairies Corporation. M. Nagata, M. Kayanoma, T. Takahashi, T. Kaneko, and H. Hara have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagata, M., Kayanoma, M., Takahashi, T. et al. Marginal Zinc Deficiency in Pregnant Rats Impairs Bone Matrix Formation and Bone Mineralization in Their Neonates. Biol Trace Elem Res 142, 190–199 (2011). https://doi.org/10.1007/s12011-010-8760-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-010-8760-8