Abstract
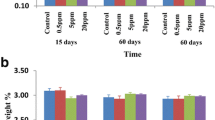
Glucose intolerance in fluorosis areas and when fluoride is administered for the treatment of osteoporosis has been reported. Controlled fluoridation of drinking water is regarded as a safe and effective measure to control dental caries. However, the effect on glucose homeostasis was not studied so far. The aim of this study was to evaluate the effect of the intake of fluoridated water supply on glucose metabolism in rats with normal and deficient renal function. Male Sprague–Dawley rats were divided into eight groups of four rats. Renal insufficiency was induced in four groups (NX) which received drinking water containing 0, 1, 5, and 15 ppm F (NaF) for 60 days. Four groups with simulated surgery acted as controls. There were no differences in plasma glucose concentration after a glucose tolerance test between controls and NX rats and among rats with different intakes of fluoride. However, plasma insulin level increased as a function of fluoride concentration in drinking water, both in controls and in NX rats. It is concluded that the consumption of fluoridated water from water supply did not affect plasma glucose levels even in cases of animals with renal disease. However, a resistance to insulin action was demonstrated



Similar content being viewed by others
References
McDonagh MS, Whiting PF, Wilson PM et al (2000) Systematic review of water fluoridation. BMJ 321:855–859
Whitford GM (1996) Metabolism and toxicity of fluoride, 2nd edn. Karger, Basel
Schmidt CW, Leuschke W (1986) Metabolism of fluorine in terminal renal failure. Urol Nephrol 79:481–484
Turner CH, Owan I, Brisendine EJ et al (1996) High fluoride intakes cause osteomalacia and diminished bone strength in rats with renal deficiency. Bone 19:595–601
Menoyo I, Rigalli A, Puche RC (2005) Effect of fluoride on the secretion of insulin in the rat. Arzneimittelforschung 55:455–460
Rigalli A, Ballina JC, Roveri E et al (1990) Inhibitory effect of fluoride on the secretion of insulin. Calcif Tissue Int 46:333–338
Rigalli A, Ballina JC, Puche RC (1992) Bone mass increase and glucose tolerance in rats chronically treated with sodium fluoride. Bone Miner 16:101–108
Chehoud KA, Chiba FY, Sassaki KT et al (2008) Effect of fluoride intake on insulin sensitivity and insulin signal transduction. Fluoride 41:270–275
De la Sota M, Puche RC, Rigalli A et al (1997) Bone mass and glucose homeostasis of subjects with high spontaneous fluoride intake. Medicina (B Aires) 57:417–420
Trivedi N, Mithal A, Gupta SK et al (1993) Reversible impairment of glucose tolerance in patients with endemic fluorosis. Diabetologia 36:826–828
Whitford GM, Sampaio FC, Pinto CS et al (2008) Pharmacokinetics of ingested fluoride: lack of effect of chemical compound. Arch Oral Biol 53:1037–1041
Gretz N, Ealdherr A, Strauch N (1993) The remnant kidney model. In: Gretz N, Strauch N (eds) Experimental and genetic rat models of chronic renal failure. Karger, Basel, pp 1–28
Rigalli A, Alloatti R, Puche RC (1999) Measurement of total and diffusible serum fluoride. J Clin Lab Anal 13:151–157
Dunipace AJ, Brizendine EJ, Zhang W et al (1995) Effect of aging on animal response to chronic fluoride exposure. J Dent Res 74:358–368
Pessan JP, Silva SM, Lauris JR et al (2008) Fluoride uptake by plaque from water and from dentifrice. J Dent Res 87:461–465
Parfitt AM (1976) The actions of parathyroid hormone on bone: relation to bone remodeling and turnover, calcium homeostasis, and metabolic bone disease. Part IV of IV parts: The state of the bones in uremic hyperaparathyroidism–the mechanisms of skeletal resistance to PTH in renal failure and pseudohypoparathyroidism and the role of PTH in osteoporosis, osteopetrosis, and osteofluorosis. Metabolism 25:1157–1188
Acknowledgments
This work was funded by a grant from Fundación Alberto J. Roemmers. The authors thank Valeria Dalmau and Agustin Ramirez by the invaluable collaboration in the surgery of induction of renal insufficiency, Hilda Moreno for fluoride measurement, and Digno Alloatti for providing the animals.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
After and intravenous dose of NaF (Do), the distribution of fluoride in the body can be represented as detailed in Fig. 4.
Distribution of fluoride in plasma, bone, and kidney after an intravenous dose (Do). [F] plasma concentration of fluoride, Vo − rate of fluoride uptake by bone tissue, Vo + rate of fluoride released from bone tissue, Vu − rate of renal fluoride excretion, Vu + rate of renal fluoride re-absorption, U cumulative urinary fluoride excretion
In this situation, the variation of plasma fluoride concentration can be represented by the following differential equation:
where
rearranging Eq. 1
After an intravenous dose of NaF, it can be supposed that Vo− > Vo+. This assumption is supported by the high plasma fluoride levels reached after the fluoride injection.
Under this consideration, Vo+ can be considered negligible; as a consequence, Eq. 2 can be written as follows:
The preceding differential equation states that after an intravenous dose of fluoride, the decrease in plasma fluoride concentration is a function of the rate of fluoride uptake by bone tissue and the renal excretion of fluoride.
It can be considered that both rates are linear functions of plasma fluoride concentration.
As a consequence
From Eqs. 3, 4, and 5, the following is obtained:
where ke is the rate constant of elimination of fluoride from plasma compartment.
Integrating both members of Eq. 7 and solving
where [F] and [F o] are the values of plasma fluoride concentration at a definite time and immediately after the injection of NaF, respectively.
Considering the initial concentration of fluoride as the ratio between the intravenous dose (Do) and the distribution volume of fluoride (V d), Eq. 8 can be rewritten as follows
Equation 9 represents the logarithm of plasma fluoride concentration as a linear function of the time. With two or more values of plasma fluoride concentration after an intravenous dose (Do), V d and ke can be obtained with the last square method of linear regression.
On the other hand, the cumulative urinary excretion of fluoride can be described by the following differential equation:
where ku is the rate constant of renal fluoride excretion. This equation states that the excretion of fluoride linearly depends on the plasma fluoride levels
Rearranging Eq. 8
and replacing in Eq. 10, the following is obtained:
Integrating both terms of equation
as U 0 = 0:
After 24 h of the mini dose of NaF, approximately 100% of fluoride not incorporated in bone tissue has been excreted in urine. The cumulative fluoride excretion after 24 h can be considered the limit of Eq. 13 when time tends to infinite. As a consequence, the limit of Eq. 13 can be written:
Solving
where [U]∞ is the cumulative fluoride excretion after 24 h of the intravenous dose. The value of ku is obtained with Eq. 14 and the values of [U]∞, Do, V d, and ke.
With the values of ke and ku using Eq. 6, ko − is obtained.
Finally, with Eq. 4 and the values of ko− and basal plasma fluoride concentration, the value of Vo− is obtained.
Rights and permissions
About this article
Cite this article
Lupo, M., Buzalaf, M.A.R. & Rigalli, A. Effect of Fluoridated Water on Plasma Insulin Levels and Glucose Homeostasis in Rats with Renal Deficiency. Biol Trace Elem Res 140, 198–207 (2011). https://doi.org/10.1007/s12011-010-8690-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-010-8690-5