Abstract
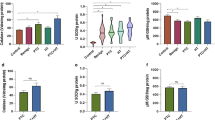
The aim of this study was to evaluate and compare the oxidative profiles of three thyroid disorders: Graves’ disease (GD), Hashimoto thyroiditis (HT), and papillary thyroid cancer (PTC). Malondialdehyde levels (MDA), glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT) activities were examined in the plasma of 52 patients (29 untreated HT, 16 untreated GD, and 7 PTC who underwent surgical therapy). Results were compared with those of 30 healthy controls and among the three groups of patients. The GD, HT, and PTC patients exhibited increased plasma MDA levels and SOD activities compared with the controls (p < 0.05, p < 0.05, and p < 0.001, respectively). CAT activities significantly increased only for the PTC and HT patients (p < 0.001 and p < 0.05, respectively), whereas GPx activities significantly decreased only in the GD and PTC (p < 0.05 and p < 0.01, respectively). The comparison among the three groups of patients has shown increased MDA level and SOD activity for the PTC patients as compared to the GD patients (p < 0.01 and p < 0.001, respectively). Compared with HT, PTC patients exhibited significant higher MDA level, SOD, and CAT activities and a significant lower GPx activity (p < 0.01, p < 0.001, p < 0.05, and p < 0.05, respectively). No significant discrepancies were noted between the GD and HT patients. Our results have clearly shown an oxidative profile that is highly disturbed for the PTC patients as compared to those of autoimmune disorders. Future studies are needed to determine whether or not the oxidative stress has a prognostic value in this pathology.

Similar content being viewed by others
Abbreviations
- CAT:
-
Catalase
- FT4:
-
Free thyroxin
- FT3:
-
Free tri-iodothyronin
- GPx:
-
Glutathione peroxidase
- GSH:
-
Glutathione
- GD:
-
Grave’s disease
- HT:
-
Hashimoto thyroiditis
- PTC:
-
Papillary thyroid cancer
- MDA:
-
Malondialdehyde
- SOD:
-
Superoxide dismutase
- TBARs:
-
Thiobarbituric acid reactive species
- TPO:
-
Thyroperoxidase
- TG:
-
Thyroglobulin
- T4:
-
Thyroxin
- T3:
-
Tri-iodothyronin
- ROS:
-
Reactive oxygen species
- TSH:
-
Thyroid-stimulating hormone
- Anti-TPO:
-
Antibody anti-thyroperoxidase
- Anti-TG:
-
Antibody anti-thyroglobulin
- TSH-R:
-
Thyroid-stimulating hormone receptor
- TSH:
-
Thyroid-stimulating hormone
References
Valko M, Leibfritz D, Moncol J et al (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem B 39:44–84
Mates JM, Perez-Gomez C, Nunez de Castro I (1999) Antioxidant enzymes and human disease. Clin Biochem 1411:385–400
Akinci M, Kosova F, Çetin B et al (2008) Oxidant/antioxidant balance in patients with thyroid cancer. Acta Cir Bras 23(6):551–554
Gargouri B, Lassoued S, Ayadi W et al (2009) Lipid peroxidation and antioxidant system in the tumor and in the blood of patients with nasopharyngeal carcinoma. Biol Trace Elem Res 132:27–34
Guarino V, Castellone MD, Avilla E et al. (2010) Thyroid cancer and inflammation. Mol Cell Endocrinol (in press)
Hultqvist M, Olsson LM, Gelderman KA et al (2009) The protective role of ROS in autoimmune disease. Trends Immunol 30(5):201–208
Chabchoub G, Mnif M, Maalej A et al (2006) Epidemiologic study of autoimmune thyroid disease in south Tunisia. Ann Endocrinol (Paris) 67:591–595
Oueslati Z, Aloui M, Gritli S et al (2002) Thyroid papillary microcarcinoma. Rev Laryngol Otol Rhinol 123(1):39–42
Figge J (1999) Epidemiology of thyroid cancer. In: Wartofsky L (ed) Thyroid cancer: a comprehensive guide to clinical management. Humana, Totowa, pp 77–83 (from article: Expression of functional metallothionein isoforms in papillary thyroid cancer 2009)
Santacroce L, Gagliardi S, Scott Kennedy A (2009) Thyroid, papillary carcinoma. Carcinomas of Endocrine Organs (emedicine from WebMD)
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convient assay for superoxide dismutase. Eur J Biochem 47:469–474
Flohe L, Gunzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–121
Degroot LJ, Quintans J (1989) The causes of auto-immune thyroid disease. Endocr Rev 10:537–562
Weetman AP, McGregor AM (1974) Autoimmune thyroid disease: further development in our understanding. Endocr Rev 15:788–830
Tomer Y, Davies TF (1993) Infection, thyroid disease and auto-immunity. Endocr Rev 14:107–120
Ademoglu E, Özbey N, Erbil Y (2006) Determination of oxidative stress in thyroid tissue and plasma of patients with Graves’ disease. Eur J Int Med 17:545–550
Bednarek J, Wysocki H, Sowinski J (2005) Peripheral parameters of oxidative stress in patients with infiltrative Graves’ ophthalmopathy treated with corticosteroids. Immunol Lett 93:227–232
Komosinska-Vassev K, Olczyk K, Kucharz EJ et al (2000) Free radical activity and antioxidant defense mechanisms in patients with hyperthyroidism due to Graves’ disease during therapy. Clin Chim Acta 300:107–117
Gerenova J, Gadjeva V (2005) Changes in parameters of oxidative stress in patients with Graves’ disease. Trakia Journal of Sciences 3:32–36
Rastogi L, Godbole MM, Madhur R et al (2006) Reduction in oxidative stress and cell death explains hypothyroidism induced neuropretection subsequent to ischemia/reperfusion insult. Exp Neur 200:290–300
Baskol G, Atmaca H, Tanriverdi F et al (2007) Oxidative stress and antioxidant status in patients with hypothyroidism before and after treatment. Exp Clin Endocrinol Diabetes 115:522–526
Videla LA (2000) Energy metabolism thyroid calorigenesis and oxydative stress: functional and cytotoxic consequences. Redox Rep 5:265–275
Fernandez V, Barrientos X, Kipreos K et al (1985) Superoxide radical generation, NADPHoxidase activity, and cytochrome P450 content in an experimental hyperthyroid state: relation to lipid peroxidation. Endocrinology 117:496–501
Nanda N, Bobby Z, Hamide A et al (2007) Association between oxidative stress and coronary lipid risk factors in hypothyroid women is independent of body mass index. Metabolism 56:1350–1355
Nobar-Rahbani M, Bahrami A, Norazarian M et al (2004) Correlation between serum levels of cholesterol and homocysteine with oxidative stress in hypothyroid patients. Int J Endocrinol Metab 2:103–109
Erdamar H, Demirci H, Yaman H et al (2008) The effect of hypothyroidism, hyperthyroidism, and their treatment on parameters of oxidative stress and antioxidant status. Clin Chem Lab Med 46:1004–1010
Messarah M, Boulakoud SM, Boumendjel A et al (2007) The impact of thyroid activity variations on some oxidizing-stress parameters in rats. Anim Biol Pathol 330:107–112
Paller MS, Sikova JJ (1986) Hypothyroïdism protects against free radical damage in ishchemic acute renal failure. Kidney Int 29:1162–1166
Monteiro Gil O, Oliveira NG, Rodrigues AS et al (2000) Cytogenetic alterations and oxidative stress in thyroid cancer patients after iodine-131 therapy. Mutagenesis 15:69–75
Weetman AP (2004) Cellular immune responses in autoimmune thyroid disease. Clin Endocrinol 61:405–413
Author information
Authors and Affiliations
Corresponding author
Additional information
Saloua Lassoued and Malek Mseddi have equally contributed in this paper.
Rights and permissions
About this article
Cite this article
Lassoued, S., Mseddi, M., Mnif, F. et al. A Comparative Study of the Oxidative Profile in Graves’ Disease, Hashimoto’s Thyroiditis, and Papillary Thyroid Cancer. Biol Trace Elem Res 138, 107–115 (2010). https://doi.org/10.1007/s12011-010-8625-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-010-8625-1