Abstract
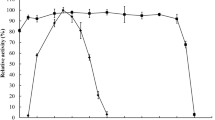
For xylooligosaccharide (XO) production, endo-xylanase from Thermobifida fusca was modified by error-prone PCR and DNA shuffling. The G4SM1 mutant (S62T, S144C, N198D, and A217V) showed the most improved hydrolytic activity and was two copies expressed in Pichia pastoris under the control of GAP promoter. The maximum xylanase activity in culture supernatants was 165 ± 5.5 U/ml, and the secreted protein concentration reached 493 mg/l in a 2-l baffled shake flask. After 6× His-tagged protein purification, the specific activity of G4SM1 was 2036 ± 45.8 U/mg, 2.12 times greater than that of wild-type enzyme. Additionally, G4SM1 was stable over a wide pH range from 5.0 to 9.0. Meanwhile, half-life of G4SM1 thermal inactivation at 70 °C increased 8.5-fold. Three-dimensional structures suggest that two amino acid substitutions, S62T and S144C, located at catalytic domain may be responsible for the enhanced activity and thermostability of xylanase. Xylobiose was the dominant end product of xylan hydrolysis by G4SM1. Due to its attractive biochemical properties, G4SM1 has potential value in commercial XO production.





Similar content being viewed by others
References
Vieira, A. T., Teixeira, M. M., & Martins, F. S. (2013). The role of probiotics and prebiotics in inducing gut immunity. Frontiers in Immunology, 4, 445. doi:10.3389/fimmu.2013.00445.
Vázquez, M. J., Alonso, J. L., Domínguez, H., & Parajó, J. C. (2000). Xylooligosaccharides: manufacture and applications. Trends in Food Science & Technology, 11, 387–393.
Joo, G. J., Rhee, I. K., Kim, S. O., & Rhee, S. J. (1998). Effect of dietary xylooligosaccharide on indigestion and retarding effect of bile acid movement across a dialysis membrane. Journal-Korean Society of Food Science and Nutrition, 27, 705–711.
Chapla, D., Pandit, P., & Shah, A. (2012). Production of xylooligosaccharides from corncob xylan by fungal xylanase and their utilization by probiotics. Bioresource Technology, 115, 215–221.
Finegold, S. M., Li, Z., Summanen, P. H., Downes, J., Thames, G., Corbett, K., Dowd, S., Krak, M., & Heber, D. (2014). Xylooligosaccharide increases bifidobacteria but not lactobacilli in human gut microbiota. Food & Function. doi:10.1039/C3FO60348B.
Hansen, C. H., Frøkiær, H., Christensen, A. G., Bergström, A., Licht, T. R., Hansen, A. K., & Metzdorff, S. B. (2013). Dietary xylooligosaccharide downregulates IFN-γ and the low-grade inflammatory cytokine IL-1β systemically in mice. Journal of Nutrition, 143, 533–540.
Howard, M. D., Gordon, D. T., Garleb, K. A., & Kerley, M. S. (1995). Dietary fructooligosaccharide, xylooligosaccharide and gum arabic have variable effects on cecal and colonic microbiota and epithelial cell proliferation in mice and rats. Journal of Nutrition, 125, 2604–2609.
Mussatto, S. I., & Mancilha, I. M. (2007). Non-digestible oligosacharides: a review. Carbohydrate Polymers, 68, 587–597.
Sun, Z. P., Lv, W. T., Yu, R. K., Li, J., Liu, H. H., Sun, W., Wang, Z. M., Li, J. P., Shan, Z., & Qin, Y. L. (2013). Effect of a straw-derived xylooligosaccharide on broiler growth performance, endocrine metabolism, and immune response. Canadian Journal of Veterinary Research, 77, 105–109.
Haddar, A., Driss, D., Frikha, F., Ellouz-Chaabouni, S., & Nasri, M. (2012). Alkaline xylanases from Bacillus mojavensis A21: production and generation of xylooligosaccharides. International Journal of Biological Macromolecules, 51, 647–656.
Samanta, A. K., Jayapal, N., Kolte, A. P., Senani, S., Sridhar, M., Suresh, K. P., & Sampath, K. T. (2012). Enzymatic production of xylooligosaccharides from alkali solubilized xylan of natural grass (Sehima nervosum). Bioresource Technology, 112, 199–205.
Bian, J., Peng, F., Peng, X. P., Peng, P., Xu, F., & Sun, R. C. (2013). Structural features and antioxidant activity of xylooligosaccharides enzymatically produced from sugarcane bagasse. Bioresource Technology, 127, 236–241.
Boler, B.M.V, & Fahey, G.C. Jr. (2012) Prebiotics of Plant and Microbial Origin. Direct-Fed Microbials and Prebiotics for Animals (Callaway, T.R. and Ricke, S.C., ed.), Springer, New York, NY, pp13-26.
Sun, J. Y., Liu, M. Q., Weng, X. Y., Qian, L. C., & Gu, S. H. (2007). Expression of recombinant Thermomonospora fusca xylanase A in Pichia pastoris and xylooligosaccharides released from xylans by it. Food Chemistry, 104, 1055–1064.
Zhang, Z. G., Yi, Z. L., Pei, X. Q., & Wu, Z. L. (2010). Improving the thermostability of Geobacillus stearothermophilus xylanase XT6 by directed evolution and site-directed mutagenesis. Bioresource Technology, 101, 9272–9278.
Verma, D., Kawarabayasi, Y., Miyazaki, K., & Satyanarayana, T. (2013). Cloning, expression and characteristics of a novel alkalistable and thermostable xylanase encoding gene (Mxyl) retrieved from compost-soil metagenome. PLoS one, 8, e52459.
Collins, T., Gerday, C., & Feller, G. (2005). Xylanases, xylanase families and extremophilic xylanases. FEMS Microbiology Reviews, 29, 3–23.
Irwin, D., Jung, E. D., & Wilson, D. B. (1994). Characterization and sequence of a Thermomonospora fusca xylanase. Applied Environmental Microbiology, 60, 63–770.
Wang, Q., & Xia, T. (2008). Enhancement of the activity and alkaline pH stability of Thermobifida fusca xylanase A by directed evolution. Biotechnology Letters, 30, 937–944.
Wang, Q., Zhao, L. L., Sun, J. Y., Liu, J. X., & Weng, X. Y. (2012). Enhancing catalytic activity of a hybrid xylanase through single substitution of Leu to Pro near the active site. World Journal of Microbiology and Biotechnology, 28, 929–935.
Stemmer, W. P. C. (1994). Rapid evolution of a protein in-vitro by DNA shuffling. Nature, 370, 389–391.
Bailey, M. J., Biely, P., & Poutanen, K. (1992). Interlaboratory testing of methods for assay of xylanase activity. Journal of Biotechnology, 23, 257–270.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anaytical Biochemistry, 72, 248–254.
Zhu, H., Qu, F., & Zhu, L. H. (1993). Isolation of genomic DNAs from plants, fungi and bacteria using benzyl chloride. Nucleic Acid Research, 21, 5279–5280.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods, 25, 402–408.
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227, 680–685.
Arnold, K., Bordoli, L., Kopp, J., & Schwede, T. (2006). The SWISS-MODEL Workspace: a web-based environment for protein structure homology modelling. Bioinformatics, 22, 195–201.
Kiefer, F., Arnold, K., Künzli, M., Bordoli, L., & Schwede, T. (2009). The SWISS-MODEL repository and associated resources. Nucleic Acids Research, 37, D387–D392.
Pettersen, E. F., Goddard, T. D., Huang, C. C., Couch, G. S., Greenblatt, D. M., Meng, E. C., & Ferrin, T. E. (2004). UCSF Chimera-a visualization system for exploratory research and analysis. Journal of Computational Chemistry, 25, 1605–1612.
Vassileva, A., Chugh, D. A., Swaminathan, S., & Khanna, N. (2001). Expression of hepatitis B surface antigen in the methylotrophic yeast Pichia pastoris using the GAP promoter. Journal of Biotechnology, 88, 21–35.
Han, Y., & Lei, X. G. (1999). Role of glycosylation in the functional expression of an Aspergillus niger phytase (phyA) in Pichia pastoris. Archives of Biochemistry and Biophysics, 364, 83–90.
Cereghino, J. L., & Cregg, J. M. (2000). Heterologous protein expression in the methylotrophic yeast Pichia pastoris. FEMS Microbiology Reviews, 24, 45–66.
Waterham, H. R., Digan, M. E., Koutz, P. J., Lair, S. V., & Cregg, J. M. (1997). Isolation of the Pichia pastoris glyceraldehyde-3-phosphate dehydrogenase gene and regulation and use of its promoter. Gene, 186, 37–44.
Sapag, A., Wouters, J., Lambert, C., De Ioannes, P., Eyzaguirre, J., & Depiereux, E. (2002). The endoxylanases from family 11: computer analysis of protein sequences reveals important structural and phylogenetic relationships. Journal of Biotechnology, 95, 109–131.
Miyazaki, K., Takenouchi, M., Kondo, H., Noro, N., Suzuki, M., & Tsuda, S. (2006). Thermal stabilization of Bacillus subtilis family-11 xylanase by directed evolution. Journal of Biological Chemistry, 281, 10236–10242.
Van den Burg, B., Dijkstra, B. W., Vriend, G., Van der Vinne, B., Venema, G., & Eijsink, V. G. (1994). Protein stabilization by hydrophobic interactions at the surface. European Journal of Biochemistry, 220, 981–985.
Funahashi, J., Takano, K., Yamagata, Y., & Yutani, K. (2000). Role of surface hydrophobic residues in the conformational stability of human lysozyme at three different positions. Biochemistry, 39, 14448–14456.
Loo, J. V., Cummings, J., Deizenne, N., Englyst, H., Franck, A., Hopkins, M., Kok, N., Macfariane, G., Newton, D., Quigley, M., Roberfroid, M., van Vliet, T., & van den Heuvel, E. (1999). Functional food properties of non-digestible oligosaccharides: a consensus report from the ENDO Project (DGXII-AIRII-CT94-1095). British Journal of Nutrition, 81, 121–132.
Acknowledgments
This work was supported in part by IAEA Coordinated Research Projects (No. 16327/R0), the National Natural Science Foundation of China (30971702), the Natural Science Foundation of Zhejiang Province (3090247), and research grants from the Science and Technology Department of Zhejiang Province, China (2010C32008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Q., Du, W., Weng, XY. et al. Recombination of Thermo-Alkalistable, High Xylooligosaccharides Producing Endo-Xylanase from Thermobifida fusca and Expression in Pichia pastoris . Appl Biochem Biotechnol 175, 1318–1329 (2015). https://doi.org/10.1007/s12010-014-1355-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1355-7