Abstract
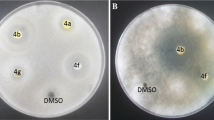
The acylhydrazone compound named ethyl N′-furan-2-carbonylbenzohydrazonate was synthesized by the condensation of ethyl benzimidate hydrochloride with furan-2-carbohydrazide. The treatment of the acylhydrazone with hydrazine hydrate afforded 4-amino-3-furan-2-yl-5-phenyl-1,2,4-triazole. The usage of this compound with various aromatic aldehydes resulted in the formation of 4-arylidenamino-3-furan-2-yl-5-phenyl-1,2,4-triazoles. Sodium borohydride reduction of 4-arylidenamino derivatives afforded 4-alkylamino-3-furan-2-yl-5-phenyl-1,2,4-triazoles. The obtained products were identified by FT-IR, 1H-NMR, 13C-NMR. A series of compounds were evaluated for their antibacterial, antiurease, and antioxidant activities. The results showed that the synthesized new compounds had effective antiurease and antioxidant activities.

Similar content being viewed by others
References
Klimesova, V., Zahajka, L., Waisser, K., Kaustova, J., & Mollmann, U. (2004). II Farmaco, 59, 279–288.
Zani, F., Vicini, P., & Incerti, M. (2004). European Journal of Medicinal Chemistry, 39, 135–140.
Tehranchian, S., Akbarzadeh, T., Fazeli, M. R., Jamalifar, H., & Shafiee, A. (2005). Bioorganic & Medicinal Chemistry Letters, 15, 1023–1025.
Awad, L. F., & El Ashry, S. H. (1998). Carbohydrate Researchs, 312, 9–22.
Amir, M., & Shikha, K. (2004). European Journal of Medicinal Chemistry, 39, 535–545.
Palaska, E., Sahin, G., Kelicen, P., Durlu, N. T., & Altınok, G. (2002). II Farmaco, 57, 101–107.
Holla, B. S., Poorjary, K. N., Rao, B. S., & Shivananda, M. K. (2002). European Journal of Medicinal Chemistry, 37, 511–517.
Henichart, J. P., Houssin, R., & Berier, J. L. (1986). Journal of Heterocyclic Chemistry, 23, 1531–1533.
Gumrukcuoglu, N., Serdar, M., Celik, E., Sevim, A., & Demirbas, N. (2007). Turkish Journal of Chemistry, 31(3), 335–348.
Serdar, M., Gumrukcuoglu, N., Karaoglu, S. A., & Demirbas, N. (2007). Turkish Journal of Chemistry, 31, 315–326.
Bekircan, O., & Gümrükçüoğlu, N. (2005). Indian Journal of Chemistry, 44B, 2107–2113.
Ocak, M., Gümrükçüoğlu, N., Ocak, Ü., Buschmann, H. J., & Schollmeyer, E. (2008). Journal of Solution Chemistry, 37, 1489–1497.
Weidinger, H., & Kranz, J. (1963). Chemische Berichte, 96(4), 1064–1070.
Gümrükçüoğlu, N., Uğraş, S., Uğraş, H. I., & Cakır, U. (2012). Journal of Inclusion Phenomena and Macrocyclic Chemistry, 73, 359–367.
Zerner, B. (1991). Bioorganic Chemistry, 19, 116–131.
Krajewska, B., Van-Eldik, R., & Brindell, M. (2012). Journal of Biological Inorganic Chemistry, 17, 1123–1134.
Visek, W. J. (1984). Journal of Dairy Science, 67(3), 481–498.
Kleiner, D., Traglauer, A., & Domm, S. (1998). Bulletin de I’Institut Pasteur, 96(4), 257–265.
Mobley, L. T. H., Island, M. D., & Hausinger, R. P. (1995). Microbiological Review, 59, 451–480.
McLean, R. J. C., Nickel, J. C., Cheng, K. J., & Costerton, J. W. (1988). CRC Critical Review in Microbiology, 16(1), 37–79.
Vaira, D., Holton, J., Ricci, C., Baset, C., Gatta, L., Perna, F., Tampieri, M. A., & Miglioli, M. (2002). Alimentary Pharmacology & Therapeutics, 16, 105–113.
Amtul, Z., & Rasheed, M. (2004). Biochemical and Biophysical Research Communications, 319, 1053–1063.
Chung, K. T., Thomasson, W. R., & Wu-Yuan, C. D. (1990). Journal of Applied Bacteriology, 69, 498–503.
Rabe, T., Mullholland, D., & van Staden, J. (2002). Journal of Ethnopharmacology, 80, 91–94.
Van Slyke, D. D., & Archibald, R. M. (1944). Journal of Biological Chemistry, 154, 623–642.
Benzie, I. F. F., & Strain, J. J. (1999). Methods in Enzymology, 299, 15–27.
Blois, M. S. (1958). Nature, 181, 1199–1200.
Miller, N. J., Rice-Evans, C. A., & Davies, M. J. (1993). Clinical Science, 84, 407–412.
Bilgin Sokmen, B., Gumrukcuoglu, N., Ugras, S., Ugras, H. I., & Yanardag, R. (2013). Journal of Enzyme Inhibition and Medicinal Chemistry, 28, 72–77.
Gumrukcuoglu, N., Bilgin Sokmen, B., Ugras, S., Ugras, H. I., & Yanardag, R. (2013). Journal of Enzyme Inhibition and Medicinal Chemistry, 28, 89–94.
Conflict of Interest
The authors declare no financial or other relationship that might lead to a conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1768 kb)
Rights and permissions
About this article
Cite this article
Sokmen, B.B., Gumrukcuoglu, N., Ugras, S. et al. Synthesis, Antibacterial, Antiurease, and Antioxidant Activities of Some New 1,2,4-Triazole Schiff Base and Amine Derivatives. Appl Biochem Biotechnol 175, 705–714 (2015). https://doi.org/10.1007/s12010-014-1307-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1307-2