Abstract
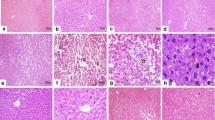
Chitosan has attracted much attention as a biomedical material, owing to its unique biological activities. In this study, hepatoprotective effect of β-chitosan obtained from the gladius of squid Sepioteuthis lessoniana was studied against carbon tetrachloride (CCl4)-induced oxidative stress and liver injury in rats. The rats that received β-chitosan along with the administration of CCl4 showed significantly decreased plasma and tissue alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities and total cholesterol, triglyceride (TG) and free fatty acid (FFA) contents, whereas the treatment with β-chitosan alone markedly increased rat hepatic and circulatory superoxide dismutase (SOD), catalase and glutathione peroxidase (GPx) and reduced glutathione (GSH) levels and decreased the malondialdehyde level. Histopathological observations recommended the marked hepatoprotective effect of β-chitosan. The CCl4-induced alterations on circulatory and hepatic antioxidant defence system were normalised by β-chitosan, and it could be concluded that the hepatoprotective effect of chitosan may be due to its antioxidant and antilipidemic property. Therefore, β-chitosan could be considered as antihepatotoxic agent.

Similar content being viewed by others
Abbreviations
- CCl4 :
-
Carbon tetrachloride
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- TG:
-
Triglycerides
- FFA:
-
Free fatty acids
- SOD:
-
Superoxide dismutase
- GPx:
-
Glutathione peroxidase
- GSH:
-
Glutathione
References
Hasegawa, R., Chujo, T., Sai-kato, K., Umemura, T., Tanimura, A., & Kurokawa, Y. (1995). Preventive effects of green tea against liver oxidative DNA damage and hepatotoxicity in rats treated with 2-nitropropane. Food and Chemical Toxicology, 33, 961–965.
Cho, E. J., Yokozawa, T., Rhyu, D. Y., Kim, H. Y., Shibahara, N., & Park, J. C. (2003). The inhibitory effects of 12 medicinal plants and their component compounds on lipid peroxidation. The American Journal of Chinese Medicine, 31, 907–917.
Datta, S., Basu, K., Sinha, S., & Bhattacharyya, P. (1998). Hepatoprotective effect of a protein isolated from Cajanus indicus (Spreng) on carbon tetrachloride induced hepatotoxicity. Indian Journal of Experimental Biology, 36, 175–181.
Sheweita, S. A., Abd El-Gabar, M., & Bastawy, M. (2001). Carbon tetra chloride changes the activity of cytochrome P450 system in the liver of male rats: role of antioxidants. Toxicology, 169, 83–92.
Kodavanti, P. R., Joshi, U. M., Young, Y. A., Meydrech, E. F., & Mehendale, H. M. (1989). Protection of hepatotoxic and lethal effects of CCl4 by partial hepatectomy. Toxicology and Pathology, 17, 494–505.
Srivastava, A., & Shivanandappa, T. (2010). Hepatoprotective effect of the root extract of Decalepis hamiltonii against carbon tetrachloride-induced oxidative stress in rats. Food Chemistry, 118, 411–417.
Demirdag, K., Bakcecioglu, I. H., Ozercan, I. H., Ozden, M., Yilmaz, S., & Kalkan, A. (2004). Role of l-carnitine in the prevention of acute liver damage induced by carbon tetrachloride in rats. Journal of Gastroenterology and Hepatology, 19, 333–338.
Lee, C. P., Shih, P. H., Hsu, C. L., & Yen, G. C. (2007). Hepatoprotection of tea seed oil (Camellia oleifera Abel.) against CCl4-induced oxidative damage in rats. Food and Chemical Toxicology, 45, 888–895.
Kim, K. W., & Thomas, R. L. (2007). Antioxidative activity of chitosans with varying molecular weights. Food Chemistry, 101, 308–313.
Shigemasa, Y., & Minami, S. (1996). Applications of chitin and chitosan for biomaterials. Biotechnology & Genetic Engineering Reviews, 13, 383–420.
Han, L. K., Kimura, Y., & Okuda, H. (1999). Reduction in fat storage during chitin-chitosan treatment in mice fed a high-fat diet. International Journal of Obesity and Related Metabolic Disorders, 23, 174–179.
Yan, Y., Wanshun, L., Baoqin, H., Bing, L., & Chenwei, F. (2006). Protective effects of chitosan oligosaccharide and its derivatives against carbon tetrachloride-induced liver damage in mice. Hepatology Research, 35, 178–184.
Jeon, T., Hwang, S. G., Park, N. G., Jung, Y. R., Shin, S. I., Choi, S. D., & Park, D. K. (2003). Antioxidative effect of chitosan on chronic carbon tetrachloride induced hepatic injury in rats. Toxicology, 187, 67–73.
Subhapradha, N., Ramasamy, P., Vairamani, S., Madeswaran, P., Srinivasan, A., & Shanmugam, A. (2013). Physicochemical characterization of β-chitosan from Sepioteuthis lessoniana gladius. Food Chemistry, 141, 907–913.
Kakkar, P., Das, B., & Viswanathan, P. N. (1984). A modified spectrophotometric assay of superoxide dismutase (SOD). Indian Journal of Biochemistry & Biophysics, 21, 130–132.
Sinha, A. K. (1972). Colorimetric assay of catalase. Analytical Biochemistry, 47, 389–394.
Rotruck, J. T., Pope, A. L., Ganther, H. E., Swanson, A. B., Hafeman, D. G., & Hoekstra, W. G. (1973). Selenium: biochemical role as a component of glutathione peroxidase. Science, 179, 588–590.
Ellman, G. L. (1959). Tissue sulphydryl groups. Archives of Biochemistry and Biophysics, 82, 70–77.
Yagi, K. (1987). Lipid peroxides and human disease. Chemistry and Physics of Lipids, 45, 337–351.
Jiang, Z. Y., Hunt, J. V., & Wolf, S. P. (1992). Ferrous ion Fe2+ oxidation in the presence of xylenol orange for the detection of lipid hydroperoxides in low density lipoprotein. Analytical Biochemistry, 202, 384–389.
Folch, J., Lees, M., & Stanley, G. H. S. (1957). A simple method for the isolation and purification of total lipids from animal tissues. Journal of Biological Chemistry, 226, 497–509.
Falholt, K., Lund, B., & Falholt, W. (1973). An easy colorimetric method for routine determination of free fatty acids in plasma. Clinica Chimica Acta, 46, 105–111.
Mitra, S. K., Venkataranganna, M. V., Sundaram, R., & Gopumadhavan, S. (1998). Effect of HD-03, a herbal formulation, on the antioxidant defence system in rats. Phytotherapy Research, 12, 114–117.
Buettner, G. R., & Schafer, F. Q. (2000). Free radicals, oxidants and antioxidants. Teratology, 62, 234–235.
Manibusan, M. K., Odin, M., & Eastmond, D. A. (2007). Postulated mode of action carbon tetrachloride: a review. Journal of Environmental Science and Health C Environmental Carcinogenesis & Ecotoxicology Reviews, 25(3), 185–209.
Weber, L. W., Boll, M., & Stampfl, A. (2003). Hepatotoxicity and mechanism of action of haloalkanes: carbon tetrachloride as a toxicological model. Critical Reviews in Toxicology, 33, 105–136.
El-Shenawy, N. S., & Abdel-Rahman, M. S. (1993). The mechanism of chloroform toxicity in isolated rat hepatocytes. Toxicology Letters, 69, 77–85.
Kurata, M., Suzuki, M., & Agar, N. S. (1993). Antioxidant system and erythrocyte life span in mammals. Biochemistry and Physiology, 106, 477–487.
Sambath Kumar, R., Sivakumar, T., Sivakumar, P., Nethaji, R., Vijayabasker, M., Perumal, P., Gupta, M., & Mazumder, U. K. (2005). Hepatoprotective and in vivo antioxidant effects of Careya arborea against carbon tetrachloride induced liver damage in rats. International Journal of Molecular Medicine and Advance Science, 1(4), 418–424.
Hewawasam, R. P., Jayatilaka, K. A. P. W., Pathirana, C., & Mudduwa, L. K. B. (2003). Protective effect of Asteracantha longifolia extracts mouse liver injury induced by carbon tetrachloride and paracetamol. Journal of Pharmacy and Pharmacology, 55, 1413–1418.
Soliman, A. M., & Fahmy, S. R. (2011). Protective and curative effects of the 15 KD isolated protein from the Peganum harmala L. seeds against carbon tetrachloride induced oxidative stress in brain, tests and erythrocytes of rats. European Review for Medical and Pharmacological Sciences, 15, 888–899.
Lieber, C. S. (1992). Medical and nutritional complications of alcoholism: mechanisms and management (p. 579). New York: Plenum.
Kaffarnik, H., Schneider, J., Schubotz, R., Hausmann, L., Multilfellner, G., Muhlfellner, O., & Zofel, P. (1978). Plasma lipids. Triglycerides/fatty acid pattern and plasma insulin in fasted healthy volunteers during continuous ingestion of ethanol. Influence of lipids inhibited by nicotinic acid. Artherosclerosis, 29, 1–7.
Yeh, Y. H., Hsieh, Y. L., Lee, Y. T., & Hu, C. C. (2012). Protective effects of Geloina eros extract against carbon tetrachloride-induced hepatotoxicity in rats. Food Research International, 48, 551–558.
Acknowledgments
Authors are thankful to the Director and Dean, CAS in Marine Biology, Faculty of Marine Sciences, Annamalai University, for providing the necessary facilities. Three of the authors (AS, AS and SV) are also thankful to the Ministry of Environment and Forests (MoEnF), New Delhi, for the financial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Subhapradha, N., Saravanan, R., Ramasamy, P. et al. Hepatoprotective Effect of β-Chitosan from Gladius of Sepioteuthis lessoniana Against Carbon Tetrachloride-Induced Oxidative Stress in Wistar Rats. Appl Biochem Biotechnol 172, 9–20 (2014). https://doi.org/10.1007/s12010-013-0499-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0499-1