Abstract
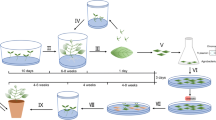
Cumin is an annual herbaceous medicinally important plant having diverse applications. An efficient and reproducible method of Agrobacterium-mediated genetic transformation was herein established for the first time. A direct regeneration method without callus induction was optimised using embryos as explant material in Gamborg’s B5 medium supplemented with 0.5-μM 6-benzyladenine and 2.0-μM α-naphthalene acetic acid. About 1,020 embryos (a mean of 255 embryos per batch) were used for the optimisation of transformation conditions. These conditions were an Agrobacterium cell suspension of 0.6 OD600, a co-cultivation time of 72 h, 300-μM acetosyringone and wounding of explants using a razor blade. Pre-cultured elongated embryos were treated using optimised conditions. About 720 embryos (a mean of 180 embryos per batch) were used for transformation and 95 % embryos showed transient β-glucuronidase expression after co-cultivation. Putative transformed embryos were cultured on B5 medium for shoot proliferation and 21 regenerated plants were obtained after selection and allowed to root. T0 plantlets showed β-glucuronidase expression and gene integration was confirmed via PCR amplification of 0.96 and 1.28 kb fragments of the hygromycin-phosphotransferase II and β-glucuronidase genes, respectively. In this study, a transformation efficiency of 1.5 % was demonstrated and a total of 11 transgenic plants were obtained at the hardening stage, however, only four plants acclimatised during hardening. Gene copy number was analysed by Southern blot analysis of hardened plants and single-copy gene integration was observed. This is the first successful attempt of Agrobacterium-mediated genetic transformation of cumin.



Similar content being viewed by others
References
Johri, R. K. (2011). Pharmacognosy Reviews, 5, 63–72.
Bettaieb, I., Bourgou, S., Wannes, W. A., Hamrouni, I., Limam, F., & Marzouk, B. (2010). Journal of Agricultural and Food Chemistry, 58, 10410–10418.
Bettaieb, I., Bourgou, S., Sriti, J., Msaada, K., Limam, F., & Marzouk, B. (2011). Journal of the Science of Food and Agriculture, 91, 2100–2107.
Einafshar, S., Poorazrang, H., Farhoosh, R., & Seiedi, S. M. (2012). European Journal of Lipid Science and Technology, 114, 168–174.
Thamaraikannan, M. and Sengottuvel, C. (2012). Cumin: Can India maintain a monopoly? Facts For You 3211–14.
Garg, B. K., Burman, U., & Kathju, S. (2002). Indian Journal of Plant Pathology, 7, 70–74.
Koocheki, A., Mehdi, N. M., & Azizi, G. (2008). Journal of Herbs Spices and Medicinal Plants, 14, 137–153.
Tatari, M. and Alikamar, R. A. (2008), In Proceedings of the second international salinity forum: Salinity, water and society, Global issues, local action, Adelaide, Australia, pp. 1–3.
Singh, N., Mishra, A., Joshi, M., & Jha, B. (2010). Plant Cell Tissue and Organ Culture, 103, 1–6.
Directorate of Agriculture (2012) Agriculture and co-operation department, Govt. of Gujarat, India. http://agri.gujarat.gov.in/hods/dire_agriculture/download/apy_1011_final.pdf. Accessed 7 November 2012.
Hunault, G., Desmarest, P., & Manoir, J. D. (1989). Foeniculum vulgare Miller: cell culture. Regeneration and the production of anethole. In Y. P. S. Bajaj (Ed.), Biotechnology in agriculture and forestry 7: medicinal and aromatic plants II (pp. 185–212). Berlin: Springer.
Baranski, R. (2008). Transgenic Plant Journal, 2, 18–38.
Tawfik, A. A., & Noga, G. (2001). Plant Cell Tissue and Organ Culture, 66, 141–147.
Tawfik, A. A., & Noga, G. (2002). Plant Cell Tissue and Organ Culture, 69, 35–40.
Li, R., Stelly, D. M., & Trolinder, N. L. (1989). Genome, 31, 1128–1134.
Stelly, D., Altman, D. W., Kohel, R. J., Rangan, T. S., & Comiskey, E. (1989). Genome, 32, 762–770.
Choi, H. W., Lemaux, P. G., & Cho, M. J. (2000). Crop Science, 40, 524–533.
Bregitzer, P., & Tonks, D. (2003). Crop Science, 43, 4–12.
Ebrahimie, E., Habashi, A. A., Ghareyazie, B., Ghannadha, M., & Mohammadie, M. (2003). Plant Cell Tissue and Organ Culture, 75, 19–25.
Ebrahimie, E., Habashi, A. A., Mohammadie-Dehcheshmeh, M., Ghannadha, M., Ghareyazie, B., & Yazdi-Amadi, B. (2006). In Vitro Cellular and Developmental Biology: Plant, 42, 455–460.
Ebrahimie, E., Naghavi, M. R., Hosseinzadeh, A., Behamta, M. R., Mohammadi-Dehcheshmeh, M., Sarrafi, A., & Spangenberg, G. (2007). Plant Cell Tissue and Organ Culture, 90, 293–311.
Tzfira, T., & Citovsky, V. (2006). Current Opinion in Biotechlogy, 17, 147–154.
Gamborg, O. L., Miller, R. A., & Ojima, K. (1968). Experimental Cell Research, 50, 151–158.
Joshi, M., Mishra, A., & Jha, B. (2011). Industrial Crops and Products, 33, 67–77.
Joshi, M., Mishra, A., & Jha, B. (2012). Industrial Crops and Products, 35, 313–316.
Jefferson, R. A. (1987). Plant Molecular Biology Reporter, 5, 387–405.
Doyle, J. J., & Doyle, J. L. (1987). Phytochemical Bulletin, 19, 11–15.
Komari, T. and Kubo, T. (1999), Advances in cellular and molecular biology of plants, vol. 5: Molecular improvement of cereal crops (Vasil, I. K., ed.), (pp. 43–83) Dordrecht: Kluwer Academic Publishers
Shou, H., Frame, B. R., Whittham, S. A., & Wang, K. (2004). Molecular Breeding, 13, 201–208.
Acknowledgments
The financial support received from CSIR, New Delhi (BSC0107–PlaGen) is thankfully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 466 kb)
Rights and permissions
About this article
Cite this article
Pandey, S., Mishra, A., Patel, M.K. et al. An Efficient Method for Agrobacterium-Mediated Genetic Transformation and Plant Regeneration in Cumin (Cuminum cyminum L.). Appl Biochem Biotechnol 171, 1–9 (2013). https://doi.org/10.1007/s12010-013-0349-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0349-1