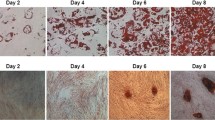
Abstract
The arachidonate cyclooxygenase (COX) pathway is involved in the generation of several types of endogenous prostaglandins (PGs) with opposite effects on adipogenesis at different life stages of adipocytes. However, the specific role of COX isoforms, the rate-limiting enzymes for the pathway, remains elusive in the regulation of the endogenous synthesis of PGs. This study was aimed at the selective suppression of the constitutive COX-1 in cultured preadipocytes by the isolation of cloned preadipocytes transfected stably with a mammalian expression vector harboring cDNA encoding mouse COX-1 in the antisense direction. The gene expression analysis revealed that the transcript and protein levels of the constitutive COX-1 were substantially suppressed in the isolated cloned transfectants with antisense COX-1. By contrast, the expression of the inducible COX-2 was not affected in the stable transfectants with antisense COX-1. All of the cloned stable transfectants with antisense COX-1 exhibited a significant reduction in the immediate synthesis of PGE2 serving as an anti-adipogenic factor. The sustained expression of COX-1 in the antisense direction induced the appreciable stimulation of fat storage in adipocytes during the maturation phase, which was associated with the higher expression levels of adipocyte-specific genes, indicating the positive regulation of adipogenesis program. Moreover, the up-regulation of adipogenesis is accompanied by a higher production of J2 series PGs including 15-deoxy-Δ12,14-PGJ2 and Δ12-PGJ2, known as pro-adipogenic factors by the transfectants with antisense COX-1. The results suggest that the inducible COX-2 can contribute to the endogenous synthesis of PGJ2 derivatives acting as autocrine mediators to simulate adipogenesis during the maturation phase by way of compensation for the suppressed expression of the constitutive COX-1.







Similar content being viewed by others
Abbreviations
- COX:
-
Cyclooxygenase
- PG:
-
Prostaglandin
- 15d-PGJ2 :
-
15-Deoxy-Δ12,14-PGJ2
- PPARγ:
-
Peroxisome proliferator-activated receptor γ
- AA:
-
Arachidonic acid
- DME-HEPES:
-
Dulbecco's modified Eagle's medium with 25 mM HEPES
- PMA:
-
Phorbol 12-myristate 13-acetate
- FBS:
-
Fetal bovine serum
- IBMX:
-
3-Isobutyl-1-methylxanthine
- G418:
-
Geneticin disulfate
- RT:
-
Reverse transcriptase
- PCR:
-
Polymerase chain reaction
- ELISA:
-
Enzyme-linked immunosorbent assay
- GM:
-
Growth medium
- DM:
-
Differentiation medium
- MM:
-
Maturation medium
- C/EBPα:
-
CCAAT/enhancer-binding protein α
- GLUT-4:
-
Glucose transporter-4
- LPL:
-
Lipoprotein lipase
- aP2:
-
Adipocyte protein 2
- SDS-PAGE:
-
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- PVDF:
-
Polyvinylidene difluoride
References
MacDougald, O. A., & Lane, M. D. (1995). Transcriptional regulation of gene expression during adipocyte differentiation. Annual Review of Biochemistry, 64, 535–559.
Gregoire, F. M., Smas, C. M., & Sul, H. S. (1998). Understanding adipocyte differentiation. Physiological Reviews, 78, 783–809.
Rangwala, S. M., & Lazar, M. A. (2000). Transcriptional control of adipogenesis. Annual Review of Nutrition, 20, 535–559.
Guilherme, A., Virbasius, J. V., Puri, C., & Czech, M. P. (2008). Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nature Reviews Molecular Cell Biology, 9, 367–377.
Wajchenberg, B. L. (2000). Subcutaneous and visceral adipose tissue: their relation to the metabolic syndrome. Endocrine Reviews, 21, 697–738.
Tsuboi, H., Sugimoto, Y., Kainoh, T., & Ichikawa, A. (2004). Prostanoid EP4 receptor is involved in suppression of 3T3-L1 adipocyte differentiation. Biochemical and Biophysical Research Communications, 322, 1066–1072.
Xu, L., Nishimura, K., Jisaka, M., Nagaya, T., & Yokota, K. (2006). Gene expression of arachidonate cyclooxygenase pathway leading to the delayed synthesis of prostaglandin E2 and F2α in response to phorbol 12-myristate 13-acetate and action of these prostanoids during life cycle of adipocytes. Biochimica et Biophysica Acta, 1761, 434–444.
Mazid, M. A., Chowdhury, A. A., Nagaya, K., Nishimura, K., Jisaka, M., Nagaya, T., et al. (2006). Endogenous 15-deoxy-Δ12,14-PGJ2 synthesized by adipocytes during maturation phase contributes to upregulation of fat storage. FEBS Letters, 580, 6885–6890.
Hossain, M. S., Chowdhury, A. A., Rahman, M. S., Nishimura, K., Jisaka, M., Nagaya, T., et al. (2011). Development of enzyme-linked immunosorbent assay for Δ12-PGJ2 and its application to the measurement of the endogenous product generated by cultured adipocytes during the maturation phase. Prostaglandins & Other Lipid Mediators, 94, 73–80.
Hyman, B. T., Stoll, L. L., & Spector, A. A. (1995). Prostaglandin production by 3T3-L1 cells in culture. Biochimica et Biophysica Acta, 713(1982), 375–385.
Forman, B. M., Tontonoz, P., Chen, J., Brun, R. P., Spiegelman, B. M., & Evans, R. M. (1995). 15-Deoxy-Δ12,14-PGJ2 is a ligand for the adipocyte determination factor PPARγ. Cell, 83, 803–812.
Kliewer, S. A., Lenhard, J. M., Wilson, T. M., Patel, I., Morris, D. C., & Lehman, J. M. (1995). A prostaglandin J2 metabolite binds peroxisome proliferator-activated receptor γ and promotes adipocyte differentiation. Cell, 83, 813–819.
Smith, W. L., DeWitt, D. L., & Garavito, R. M. (2000). Cyclooxygenases: structure, cellular, and molecular biology. Annual Review of Biochemistry, 69, 145–182.
Fain, J. N., Ballou, L. R., & Bahouth, S. W. (2001). Obesity is induced in mice heterozygous for cyclooxygenase-2. Prostaglandins Other Lipids Mediat, 65, 199–209.
Green, H., & Kehinde, O. (1974). Sublines of mouse 3T3 cells that accumulate lipid. Cell, 1, 113–116.
Green, H., & Kehinde, O. (1975). An established preadipose cell line and its differentiation in culture. II. Factors affecting the adipocyte conversion. Cell, 5, 19–27.
Yan, H., Kermouni, A., Abdel-Hafez, M., & Lau, D. C. W. (2003). Role of cyclooxygenases COX-1 and COX-2 in modulating adipogenesis in 3T3-L1 cells. Journal of Lipid Research, 44, 424–429.
Petersen, R. K., Jørgensen, C., Rustan, A. C., Frøyland, L., Muller-Decker, K., Furstenberger, G., et al. (2003). Arachidonic acid-dependent inhibition of adipocyte differentiation requires PKA activity and is associated with sustained expression of cyclooxygenases. Journal of Lipid Research, 44, 2320–2330.
Lehmann, J. M., Lenhard, J. M., Oliver, B. B., Ringold, G. M., & Kliewer, S. A. (1997). Peroxisome proliferator-activated receptors α and γ are activated by indomethacin and other non-steroidal anti-inflammatory drugs. Journal of Biological Chemistry, 272, 3406–3410.
Chu, X., Nishimura, K., Jisaka, M., Nagaya, T., Shono, F., & Yokota, K. (2010). Up-regulation of adipogenesis in adiocytes expressing stably cylooxygenase-2 in the antisense direction. Prostaglandins & Other Lipid Mediators, 91, 1–9.
Chowdhury, A. A., Hossain, M. S., Rahman, M. S., Nishijima, K., Jisaka, M., Nagaya, T., et al. (2011). Sustained expression of lipocalin-type prostaglandin D synthase in the antisense direction positively regulates adipogenesis in cloned cultured preadipocytes. Biochemical and Biophysical Research Communications, 411, 287–292.
Yokota, K., Morishima, T., Nagaya, T., Jisaka, M., & Takinami, K. (1996). Modification of cultured Madin-Darby canine kidney cells with dietary unsaturated fatty acids and regulation of arachidonate cascade reaction. Bioscience, Biotechnology, and Biochemistry, 60, 1096–1103.
Lu, S., Nishimura, K., Hossain, M. A., Jisaka, M., Nagaya, T., & Yokota, K. (2004). Regulation and role of arachidonate cascade during changes in life cycle of adipocytes. Applied Biochemistry and Biotechnology, 118, 133–153.
Chu, X., Xu, L., Nishimura, K., Jisaka, M., Nagaya, T., Shono, F., et al. (2009). Suppression of adipogenesis program in cultured preadipocytes transfected stably with cyclooxygenase isoforms. Biochimica et Biophysica Acta, 1791, 273–280.
Chomczynski, P., & Sacchi, N. (1987). Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Analytical Biochemistry, 162, 156–159.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. L. (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry, 193, 265–275.
Markwell, M. A., Hass, S. M., Toleret, N. E., & Bieber, L. I. (1981). Protein determination in membrane and lipoprotein samples: manual and automated procedures. Methods in Enzymology, 72, 296–303.
Kuri-Harcuch, W., & Green, H. (1978). Adipose conversion of 3T3 cells depends on a serum factor. Proceedings of the National Academy of Science U S A, 75, 6107–6109.
Chowdhury, A. A., Rahman, M. S., Nishimura, K., Jisaka, M., Nagaya, T., Ishikawa, T., et al. (2011). 15-Deoxy-Δ12,14-PGJ2 interferes inducible synthesis of prostaglandins E2 and F2α that suppress subsequent adipogenesis program in cultured preadipocytes. Prostaglandins & Other Lipid Mediators, 95, 53–62.
Shibata, T., Kondo, M., Osawa, T., Shibata, N., Kobayashi, M., & Uchida, K. (2002). 15-Deoxy-Δ12,14-PGJ2: A prostaglandin D2 metabolite generated during inflammatory processes. Journal of Biological Chemistry, 277, 10459–10466.
Murakami, M., Kambe, T., Shimbara, S., & Kudo, I. (1999). Functional coupling between various phospholipase A2s and cyclooxygenases in immediate and delayed prostanoid biosynthetic pathways. Journal of Biological Chemistry, 274, 3103–3115.
Cornelius, P., MacDougald, O. A., & Lane, M. D. (1994). Regulation of adipocyte development. Annual Review of Nutrition, 14, 99–129.
Mandrup, S., & Lane, M. D. (1997). Regulating adipogenesis. Journal of Biological Chemistry, 272, 5367–5370.
Fitzpatrick, F. A., & Wynalda, M. A. (1983). Albumin-catalyzed metabolism of prostaglandin D2: identification of products formed in vitro. Journal of Biological Chemistry, 258, 11713–11718.
Ghoshal, S., Trivedi, D. B., Graf, G. A., & Loftin, C. D. (2011). Cycylooxygenase-2 deficiency attenuates adipose tissue differentiation and inflammation in mice. Journal of Biological Chemistry, 286, 889–898.
Munroe, D. G., Wang, E. Y., MacIntyre, J. P., Tam, S. S. C., Lee, D. H. S., Taylor, G. R., et al. (1995). Novel intracellular signaling function of prostaglandin H synthase-1 in NF-B activation. Journal of Inflammation, 45, 260–268.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rahman, M.S., Syeda, P.K., Khan, F. et al. Cultured Preadipocytes Undergoing Stable Transfection with Cyclooxygenase-1 in the Antisense Direction Accelerate Adipogenesis During the Maturation Phase of Adipocytes. Appl Biochem Biotechnol 171, 128–144 (2013). https://doi.org/10.1007/s12010-013-0347-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0347-3