Abstract
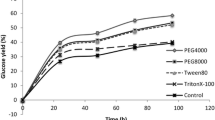
Extrusion processing has shown potential to be used as a pretreatment method for second-generation bioethanol production. Furthermore, surfactants have been shown to reduce enzyme deactivation and increase the efficiency of hydrolysis. Therefore, a sequential pretreatment technique was developed for corn stover (CS) and prairie cordgrass (PCG) in which a single screw extruder was used for the first pretreatment according to a previously optimized condition using 70–180 °C for feed, barrel, and die zones with 65–155 rpm screw speed. The second pretreatment was optimized in this study at 45–55 °C, 1–4 h, 0.15–0.6 g Tween 20/g glucan according to response surface methodology. Optimization of surfactant pretreatment facilitated the estimation of interaction and higher-order effects for major factors involved in surfactant treatment (temperature, time, surfactant loading). Using 8.6 FPU/g glucan cellulase, the optimum conditions found by fitting appropriate quadratic models to the data increased glucose and xylose yield by 27.5 and 33 % for CS and by 21.5 and 27 % for PCG, respectively. Tween 20 concentrations and pretreatment temperature were the most significant factors affecting sugar yield (p value <0.05). Studies of SDS concentration at and beyond critical micelle concentration (5.2–100 mM) demonstrated a decrease in sugar yield compared to control.




Similar content being viewed by others
References
Taherzadeh, M. J., & Karimi, K. (2007). Enzyme based hydrolysis processes for ethanol from lignocellulosic materials: a review. BioResources, 4, 707–738.
Huber, W. G., & Dale, B. E. (2009). The fuel of the future is gasoline. American Scientist. (9 April).
Bush, T. (2002). Plant fact sheet; prairie cordgrass Spartina pectinata Bosc ex link. USDA NRCS.
Ferreira, S. M. P., Duarte, A. P., Queiroz, J. A., & Domingues, F. C. (2009). Influence of buffer systems on Trichoderma ressei Rut C-30 morphology and cellulase production. Electronic Journal of Biotechnol, 12, 1–9.
Ma, A. Z., Hu, Q., Qu, Y. B., Bai, Z. H., Liu, W. F., & Zhuang, G. Q. (2008). The enzymatic hydrolysis rate of cellulose decrease with irreversible adsorption of cellobiohydrolase I. Enzyme and Microbial Technology, 42, 543–547.
Sadana, A. (1988). Enzyme deactivation. Biotechnology Advances, 16, 349–446.
Gunjikar, T. P., Sawant, S. B., & Joshi, J. B. (2001). Shear deactivation of cellulase, exoglucanase, endoglucanase, and â-glucosidase in a mechanically agitated reactor. Bio Programming, 17, 1161–1168.
Yang, B., Deidre, M., Charles, W., & Wyman, E. (2006). Changes in the enzymatic hydrolysis rate of Avicel cellulose with conversion. Biotechnology and Bioengineering, 94, 1122–1128.
Yang, S., Jia, W. Z., Quian, Q. Y., Zhou, Y. G., & Xia, X. H. (2009). Simple approach for efficient encapsulation of enzyme in silica matrix with retained bioactivity. Analytical Chemistry, 7, 3478–3484.
Khaled, M. A., Male, K. B., Hrapovic, S., & Luong, J. H. T. (2009). Cellulose nanocrystal/gold nanoparticle composite as a matrix for enzyme immobilization. Applied Materials and Interfaces, 1, 1383–1386.
Helle, S. S., Duff, S. J. B., & Copper, D. G. (1993). Effect of surfactants on cellulose hydrolysis. Biotechnology and Bioengineering, 42, 611–617.
Castanon, M., & Wilke, C. R. (1981). Effect of the surfactant Tween 80 on enzymatic hydrolysis of newspaper. Biotechnology and Bioengineering, 23, 1365–1372.
Erriksson, T., Karlsson, J., & Tjerland, F. (2002). Mechanism of surfactant effect in enzymatic hydrolysis of lignocellusoe. Enzyme and Microbial Technology, 3, 353–364.
Kaar, W. K., & Holtzapple, M. T. (1998). Benefits from Tween during enzymatic hydrolysis of corn stover. Biotechnology and Bioengineering, 59, 419–427.
Ballstros, M., Oliva, J. M., Negro, M. J., Manzanares, P., & Ballstros, I. (2004). Ethanol from lignocellulosic materials by a simultaneous saccharification and fermentation process (SSF) with Kluyveromyces marxianus CECT 10875. Process Biochemistry, 39, 1843–1848.
Zheng, Y., Pan, Z., & Zhang, R. (2008). Non-ionic surfactants and non-catalytic protein treatment on enzymatic hydrolysis of pretreated creeping wild ryegrass. Applied Biochemistry and Biotechnology, 146, 231–248.
Errikson, T., Borjesson, J., & Tjerlend, F. (2002). Mechanism of surfactant in enzymatic hydrolysis of lignocellulose. Enzyme and Microbial Technology, 31, 353–364.
Badley, R. A., Carruthers, L., & Phillips, M. C. (1977). Hydrophobic free energy and the denaturation of proteins. Biochemica et Biophysica Acta, 495, 110–113.
Reese, E. T., & Robinson, F. M. (1981). Denaturation of β-lactoglobulin by shaking and its subsequent renaturation. Journal of Colloid and Interface Science, 83, 393–400.
Ramo, L., Filho, Z., Deschamps, F., & Saddler, J. (1999). The effect of Thrichoderma cellulose on the fine structure of bleached softwood kraft pulp. Enzyme and Microbial Technology, 24, 371–380.
Kumar, R., & Wyman, C. E. (2008). Effect of additive on the digestibility of corn stover solids following pretreatment by leading technologies. Biotechnology and Bioengineering, 102, 1544–1557.
Kaar, W. E., & Holtzapple, M. T. (2000). Using lime pretreatment to facilitate the enzymatic hydrolysis of corn stover. Biomass and Bioenergy, 18, 189–199.
Naem, A., & Khan, R. H. (2004). Characterization of molten globule state of cytochrome c at alkaline, native and acidic pH induced by butanol and SDS. The International Journal of Biochemistry & Cell Biology, 36, 2281–2292.
Moore, B. M., & Flurkey, W. H. (1990). Sodium dodecyl sulfate activation of a plant polyphenoloxidase. Journal of Biological Chemistry, 265, 4982–4988.
Parker, W., & Song, P. S. (1992). Protein structures in SDS micelle–protein complexes. Biophysics, 61, 1435–1439.
Lee, S. H., Inoue, S., Teramato, Y., & Endo, T. (2010). Enzymatic saccharification of woody biomass micro/nanofibrillated by continuous extrusion process II. Effect of hot-compressed water treatment. Bioresource Technology, 101, 9645–9649.
Karunanithy, C., & Muthukumarappan, K. (2011). Optimization of alkali soaking and extrusion pretreatment of prairie cordgrass for maximum sugar recovery by enzymatic hydrolysis. Biochemical Engineering Journal, 54, 71–82.
Karunanithy, C., & Muthukumarappan, K. (2011). Application of response surface methodology to optimize the alkali concentration, corn stover particle size and extruder parameters for maximum sugar recovery. Biofuel ISBN 978-953-307-478-8.
Karunanithy, C., & Muthukumarappan, K. (2011). Optimization of corn stover and extruder parameters for enzymatic hydrolysis using response surface methodology. Biological Engineering, 3(2), 73–95.
Xiang, J., Fan, J. B., Chen, N., & Liang, Y. (2006). Interaction of cellulose with sodium dodecyl sulfate at critical micelle concentration level. 49, 175–180.
Borjesson, J., Engqvist, M., Slipos, B., & Tjerneld, F. (2007). Effect of Poly ethylene glycol on enzymatic hydrolysis and adsorption of cellulose enzymes to pretreated lignocellulose. Enzyme and Microbial Technology, 41, 186–195.
Kaya, F., Heitmann, J. A., & Joyce, T. W. (1995). Influence of surfactants on the enzymatic hydrolysis of xylan and cellulose. TAPPI Journal, 78, 150–157.
QI, B., Chen, X., & Wan, Y. (2010). Pretreatment of wheat straw by non-ionic surfactant assisted dilute acid for enhancing enzymatic hydrolysis and ethanol production. Bioresource Technology, 101, 4875–4883.
Montgomery D. C. Design and analysis of experiments. New York: John Wiley and Sons. pp. 427–450.
Saeki, S., Kuwahara, N., Nakata, M., & Kaneko, M. (1976). Upper an dlower critical solution temperature in poly ethylene glycol solutions. Polymer, 17, 685–689.
Tu, M., Chandra, P. R., & Saddler J. N. (2007). Recycling cellulase during the hydrolysis of steam exploded and ethanol pretreated Lodgepole pine. 23, 1130–1137.
Viparelli, P., Alfani, F., & Cantarella, M. (1999). Models for enzyme superactivity in aqueous solutions of surfactants. The Biochemical Journal, 344, 765–773.
Karlstrom, G. (1985). A new model for upper and lower critical solution temperature in poly ethylene oxide solusions. Journal of Physical Chemistry, 89, 4962–4964.
Cristenson, J. B., Borejsson, J., Bruun, M. H., Tjerneld, F., & Jorgensen, H. (2007). Use of surfactant active additives in enzymatic hydrolysis of wheat straw lignocellulose. Enzyme and Microbial Technology, 40, 888–895.
Fendler, V. J. H. (1982). Membranen membrane mimetic chemistry. New York, Chichester, Brisbane, Toronto, Singapore: John Wiley & Sons, XI 522 S.
Acknowledgments
Funding support was provided by Sun Grant project titled “Development of pretreatment strategies”. The authors would also like to thank Novozymes Inc. for providing the commercial enzymes. We would also thank Dr. Karunanithy who helped in the extrusion of corn stover and prairie cordgrass.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eckard, A.D., Muthukumarappan, K. & Gibbons, W. Modeling of Pretreatment Condition of Extrusion-Pretreated Prairie Cordgrass and Corn Stover with Poly (Oxyethylen)20 Sorbitan Monolaurate. Appl Biochem Biotechnol 167, 377–393 (2012). https://doi.org/10.1007/s12010-012-9698-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9698-4