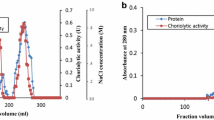
Abstract
Hatching enzyme (HE) is of importance to degrade egg membrane to let the larvae be free. HE was purified and characterized from starfish blastula. The specific activity and the purification ratio of the purified HE with 110.9 kDa of molecular weight were 449.62 U/mg and 7.42-fold, respectively. Its optimal pH and temperature for activity were pH 8.0 and 30 °C, respectively. This enzyme was relatively stable in the range of pH 4.0–6.0 and 30–40 °C. This enzyme was inhibited by ethylene diamine tetraacetic acid (EDTA) and ethylene glycol tetraacetic acid, and also done moderately by Leupeptin, tosyl-lysine chloromethyl ketone, tosyl-phenylalanine chloromethyl ketone, and phenyl-methanesulfonyl fluoride. Zn2+ ion activated HE activity strongly and recovered the EDTA-pretreated activity more than did Ca2+, Mg2+, and Cu2+. Based on the results above, the starfish HE was classified as a zinc metallo- and trypsin-like serine protease. The values of Km, Vmax, and Kcat of the starfish HE on dimethyl casein were 0.31 mg/ml, 0.17 U/ml, and 122.70 s−1, respectively, whereas 1.09 mg/ml, 0.12 U/ml, and 771.98 s−1 on type I collagen. Therefore, the starfish HE could be a potential cosmeceutical because of its strong cleavage specificity on type I collagen.






Similar content being viewed by others
References
Katagiri, C. (1975). The Journal of Experimental Zoology, 193, 109–118.
Denuc’, J. M. (985). Meded Kon Acad Wet Belgie, 46, 1–30.
Fan, T. J. (2000). Acta Zoologica Sinica, 46, 308–313.
Yamagami, K. (1988). Fish Phil, 11, 447–499.
Kofatos, F. C., & Williams, C. M. (1964). Science, 146, 538–540.
Hoshi, M., Numakunai, T., & Sawada, H. (1981). Developmental Biology, 86, 117–121.
Kitamura, Y., & Katagiri, C. (1998). Biochimica et Biophysica Acta, 387, 153–164.
Fan, T. J., & Katagiri, C. (2001). European Journal of Biochemistry, 268, 4892–4898.
Yasumasu, S., Iuchi, I., & Yamagami, K. (1989). Journal of Biochemistry, 105, 212–218.
Yasumasu, S., Iuchi, I., & Yamagami, K. (1989). Journal of Biochemistry, 105, 204–211.
Perona, R. M., & Wassarman, P. M. (1986). Developmental Biology, 114, 42–52.
Ben, F., Edwards, W., Ross, A., & Dennis, B. (1977). Archives of Biochemistry and Biophysics, 182, 696–704.
Kiyoshi, T., Hideyoshi, Y., & Motonori, H. (1979). European Journal of Biochemistry, 100, 257–265.
Li, B., Fan, T., Yang, L., Cong, R., Li, L., Sun, W., Cui-xian, L., & Shi, Z. (2006). Archives of Biochemistry and Biophysics, 451, 188–193.
D’Aniello, A., de Vincentiis, M., Di Fiore, M. M., & Scippa, S. (1997). Biochimica et Biophysica Acta, 1339, 101–112.
Shi, Z. P., Fan, T. J., Cong, R. S., Wang, X. F., Sun, W. J., & Yang, L. L. (2006). Fish Physiology and Biochemistry, 32, 35–42.
Lepage, T., & Gache, C. (1990). EMBO Journal, 9, 3003–3012.
Ala-Kokko, L., Rintala, A., & Savolainen, E.-R. (1987). Journal of Investigative Dermatology, 89, 238–244.
Sabatino, Gregory L. (Chester Springs, PA, US), Del Tito Jr., Benjamin J. (Doylestown, PA, US), Bassett, Phillip J. (Newcastle-under-Lyme, GB), Tharia, Hazel A. (Nr Crewe, GB), Hitchcock, Antony G. (Crewe, GB). (2010). Compositions and methods for treating collagen-mediated diseases. US patent 007811560B2
Norway. Aqua Bio Technology, Norway, July. (2012). Available: http://www.aquabiotechnology.com/index.php?id=5.
Barkhouse C L, M Niles, L-A Davidson. (2007). Can. Ind. Rep. Fish. Aquat. Sci, 279, vii + 38.
Kester, D. R., Duedall, I. W., Connors, D. N., & Pytkowicz, R. M. (1967). Limnology and Oceanography, 12, 176–179.
Thierry, L., & Christian, G. (1989). Journal of Biological Chemistry, 9, 4787–4793.
Yamagami, K. (1970). Zoology Japan, 43, 1–9.
Bradford, M. M. (196). Analytical Biochemistry, 72, 248–254.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Chandler, D. E., & Heuser, J. (1980). The Journal of Cell Biology, 84, 618–632.
Mozingo, N. M., Hollar, L. R., & Chandler, D. E. (1993). Cell Science, 104, 929–938.
Tingjun, F., Jing, W., Wenpeng, Y., Qiwang, Z., Ying, S., & Rishan, C. (2010). Acta Biochimica et Biophysica Sinica, 42, 165–171.
Roe, J. L., & Lennarz, W. J. (1990). Journal of Biological Chemistry, 265, 8704–8711.
Kohji, N., Hiroshi, T., Yamato, K., Masaaki, Y., & Norio, S. (1991). Biochemistry, 30, 6115–6123.
Leonard, D. M., Taylor, M. H., & Singleton, R., Jr. (1981). The Journal of Experimental Zoology, 216, 133–140.
Hagenmaier, H. E. (1974). Comparative Biochemistry and Physiology, 49B, 313–324.
Schoots, A. F., & Denuce, J. M. (1981). International Journal of Biochemistry, 13, 591–602.
Iwasawa, A., Mao, K. M., Yasumasu, S., & Yoshizaki, N. (2009). Poultry Science, 88, 2636–2643.
Fersht, A. (1999). Structure and mechanism in protein science: a guide to enzyme catalysis and protein folding (pp. 169–190). New York: Freeman.
Jiang, S. T., Moody, M. W., & Chen, H. S. (1991). Journal of Food Science, 56, 322–326.
Natalia, M., & Varinia, M. (2010). Milady’s skin care and cosmetic ingredients dictionary (3rd ed., pp. 9–11). Canada: Thomson Learning.
Auld, D. S. (1995). Methods in Enzymology, 248, 228–242.
Nakatsuka. (1079). Development, Growth & Differentiation, 21, 245–253.
Jongeneel, C. V., Bouvier, J., & Bairoch, A. (1989). FEBS Letters, 242, 211–214.
Juro, I. (1985). Zoological Science, 2, 1–10.
Denuce, J. M., & Thijssen, F. J. W. (1975). Archives Biology (Brussels), 86, 391–398.
Kaighn, M. E. (1964). Developmental Biology, 9, 56–80.
Sano, K., Inohaya, K., Kawaguchi, M., Yoshizaki, N., Iuchi, I., & Yasumasu, S. (2008). FEBS Journal, 275, 5934–5946.
Fujiwara, M., Muragaki, Y., & Ooshima, A. (2005). British Journal of Dermatology, 153, 295–300.
Babu, M., Diegelmann, R., & Oliver, N. (1989). Molecular and Cellular Biology, 9, 1642–1650.
Acknowledgments
This work was supported by grant no. RTI 05-01-02 from the Regional Technology Innovation Program of the Ministry of Knowledge Economics (MKE), Republic of Korea. Zhi Jiang Li is a recipient of a graduate fellowship provided by Brain Korea (BK21) program sponsored by the Ministry of Education, Science and Technology, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Z.J., Kim, S.M. A Novel Hatching Enzyme from Starfish Asterias amurensis: Purification, Characterization, and Cleavage Specificity. Appl Biochem Biotechnol 169, 1386–1396 (2013). https://doi.org/10.1007/s12010-012-0090-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-0090-1