Abstract
Background
Primary tumor growth during neoadjuvant chemotherapy is believed to be a sign of resistance to chemotherapy (chemoresistance), and often is associated with poor histologic response, local recurrence, and poorer survival. Currently there are no proven indicators to predict poor response to chemotherapy at the time of diagnosis.
Questions/purposes
We asked (1) what clinicopathologic factors present at diagnosis predict primary tumor growth during neoadjuvant chemotherapy, (2) what factors at presentation predict survival, and (3) when the factors at presentation and the treatment-related factors are considered, what factors independently correlate with survival.
Methods
We studied 567 patients with Stage IIB osteosarcomas. The factors assessed included age, sex, location, pattern on plain radiographs (radiodense, radiolucent, mixed), MRI findings, pathologic subtype, initial tumor volume determined by MRI, tumor volume change after chemotherapy, surgical margin, and histologic response to preoperative chemotherapy. Logistic modeling was used to identify risk factors.
Results
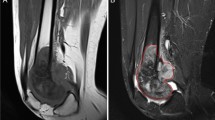
Independent risk factors associated with primary tumor growth after neoadjuvant chemotherapy were proximal tumor location (p < 0.01; relative risk [RR], 2.41; 95% CI, 1.5–3.86) and fluid-fluid level on initial MRI (p < 0.01; RR, 5.56; 95% CI, 3.48–8.87). Among factors at presentation, large initial tumor volume (p < 0.01; RR, 1.58; 95% CI, 1.22–2.04), proximal tumor site (p < 0.01; RR, 1.61; 95% CI, 1.19–2.19), and presence of fluid-fluid level (p < 0.01; RR, 1.83; 95% CI, 1.37–2.5) independently predicted reduced event–free survival. When we consider the factors at presentation and treatment-related factors, large initial tumor volume (p < 0.01; RR, 1.54), tumor growth after neoadjuvant chemotherapy (p < 0.01; RR, 3.88), inadequate surgical margin (p < 0.01; RR, 2.42), and poor histologic response (p = 0.03; RR, 1.43) were independent poor prognostic factors of event-free survival.
Conclusions
Proximal tumor location and the presence of the fluid-fluid level on initial MRI were predictors of tumor progression and poor survival in patients presenting with Stage IIB osteosarcomas. If confirmed in other studies, patients with these risk factors should be considered for trials of other treatment strategy.
Level of Evidence
Level III, prognostic study. See the Instructions for Authors for a complete description of levels of evidence.


Similar content being viewed by others
References
Bacci G, Bertoni F, Longhi A, Ferrari S, Forni C, Biagini R, Bacchini P, Donati D, Manfrini M, Bernini G, Lari S. Neoadjuvant chemotherapy for high-grade central osteosarcoma of the extremity: histologic response to preoperative chemotherapy correlates with histologic subtype of the tumor. Cancer. 2003;97:3068–3075.
Bacci G, Ferrari S, Ruggieri P, Biagini R, Fabbri N, Campanacci L, Bacchini P, Longhi A, Forni C, Bertoni F. Telangiectatic osteosarcoma of the extremity: neoadjuvant chemotherapy in 24 cases. Acta Orthop Scand. 2001;72:167–172.
Bacci G, Mercuri M, Longhi A, Ferrari S, Bertoni F, Versari M, Picci P. Grade of chemotherapy-induced necrosis as a predictor of local and systemic control in 881 patients with non-metastatic osteosarcoma of the extremities treated with neoadjuvant chemotherapy in a single institution. Eur J Cancer. 2005;41:2079–2085.
Bacci G, Picci P, Ferrari S, Orlandi M, Ruggieri P, Casadei R, Ferraro A, Biagini R, Battistini A. Prognostic significance of serum alkaline phosphatase measurements in patients with osteosarcoma treated with adjuvant or neoadjuvant chemotherapy. Cancer. 1993;71:1224–1230.
Bielack SS, Kempf-Bielack B, Delling G, Exner GU, Flege S, Helmke K, Kotz R, Salzer-Kuntschik M, Werner M, Winkelmann W, Zoubek A, Jurgens H, Winkler K. Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1,702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol. 2002;20:776–790.
Bieling P, Rehan N, Winkler P, Helmke K, Maas R, Fuchs N, Bielack S, Heise U, Jurgens H, Treuner J, Romanowski R, Exner U, Kotz R, Winkler K. Tumor size and prognosis in aggressively treated osteosarcoma. J Clin Oncol. 1996;14:848–858.
Briccoli A, Rocca M, Salone M, Bacci G, Ferrari S, Balladelli A, Mercuri M. Resection of recurrent pulmonary metastases in patients with osteosarcoma. Cancer. 2005;104:1721–1725.
Cho WH, Song WS, Jeon DG, Kong CB, Kim MS, Lee JA, Yoo JY, Kim JD, Lee SY. Differential presentations, clinical courses, and survivals of osteosarcomas of the proximal humerus over other extremity locations. Ann Surg Oncol. 2010;17:702–708.
Davis AM, Bell RS, Goodwin PJ. Prognostic factors in osteosarcoma: a critical review. J Clin Oncol. 1994;12:423–431.
Enneking WF, Spanier SS, Goodman MA. A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop Relat Res. 1980;153:106–120.
Fellenberg J, Dechant MJ, Ewerbeck V, Mau H. Identification of drug-regulated genes in osteosarcoma cells. Int J Cancer. 2003;105:636–643.
Fuchs N, Bielack SS, Epler D, Bieling P, Delling G, Korholz D, Graf N, Heise U, Jurgens H, Kotz R, Salzer-Kuntschik M, Weinel P, Werner M, Winkler K. Long-term results of the co-operative German-Austrian-Swiss osteosarcoma study group’s protocol COSS-86 of intensive multidrug chemotherapy and surgery for osteosarcoma of the limbs. Ann Oncol. 1998;9:893–899.
Gobel V, Jurgens H, Etspuler G, Kemperdick H, Jungblut RM, Stienen U, Gobel U. Prognostic significance of tumor volume in localized Ewing’s sarcoma of bone in children and adolescents. J Cancer Res Clin Oncol. 1987;113:187–191.
Harting MT, Lally KP, Andrassy RJ, Vaporciyan AA, Cox CS Jr, Hayes-Jordan A, Blakely ML. Age as a prognostic factor for patients with osteosarcoma: an analysis of 438 patients. J Cancer Res Clin Oncol. 2010;136:561–570.
Holscher HC, Bloem JL, Vanel D, Hermans J, Nooy MA, Taminiau AH, Henry-Amar M. Osteosarcoma: chemotherapy-induced changes at MR imaging. Radiology. 1992;182:839–844.
Ifergan I, Meller I, Issakov J, Assaraf YG. Reduced folate carrier protein expression in osteosarcoma: implications for the prediction of tumor chemosensitivity. Cancer. 2003;98:1958–1966.
Khanna C, Wan X, Bose S, Cassaday R, Olomu O, Mendoza A, Yeung C, Gorlick R, Hewitt SM, Helman LJ. The membrane-cytoskeleton linker ezrin is necessary for osteosarcoma metastasis. Nat Med. 2004;10:182–186.
Kim MS, Cho WH, Song WS, Lee SY, Jeon DG. time dependency of prognostic factors in patients with stage II osteosarcomas. Clin Orthop Relat Res. 2007;463:157–165.
Kim MS, Lee SY, Cho WH, Song WS, Koh JS, Lee JA, Yoo JY, Jung ST, Jeon DG. Relationships between plain-film radiographic patterns and clinicopathologic variables in AJCC stage II osteosarcoma. Skeletal Radiol. 2008;37:997–1001.
Kim MS, Lee SY, Cho WH, Song WS, Koh JS, Lee JA, Yoo JY, Jung ST, Jeon DG. Effect of increases in tumor volume after neoadjuvant chemotherapy on the outcome of stage II osteosarcoma regardless of histological response. J Orthop Sci. 2009;14:292–297.
Kong CB, Song WS, Cho WH, Oh JM, Jeon DG. Local recurrence has only a small effect on survival in high-risk extremity osteosarcoma. Clin Orthop Relat Res. 2012;470:1482–1490.
Lee SY, Jeon DG, Cho WH, Song WS, Kim MB, Park JH. Preliminary study of chemosenstivity tests in osteosarcoma using a histoculture drug response assay. Anticancer Res. 2006;26:2929–2932.
Meyers PA, Heller G, Healey J, Huvos A, Lane J, Marcove R, Applewhite A, Vlamis V, Rosen G. Chemotherapy for nonmetastatic osteogenic sarcoma: the Memorial Sloan-Kettering experience. J Clin Oncol. 1992;10:5–15.
Picci P, Bacci G, Campanacci M, Gasparini M, Pilotti S, Cerasoli S, Bertoni F, Guerra A, Capanna R, Albisinni U, et al. Histologic evaluation of necrosis in osteosarcoma induced by chemotherapy: regional mapping of viable and nonviable tumor. Cancer. 1985;56:1515–1521.
Provisor AJ, Ettinger LJ, Nachman JB, Krailo MD, Makley JT, Yunis EJ, Huvos AG, Betcher DL, Baum ES, Kisker CT, Miser JS. Treatment of nonmetastatic osteosarcoma of the extremity with preoperative and postoperative chemotherapy: a report from the Children’s Cancer Group. J Clin Oncol. 1997;15:76–84.
Rosen G, Marcove RC, Huvos AG, Caparros BI, Lane JM, Nirenberg A, Cacavio A, Groshen S. Primary osteogenic sarcoma: eight-year experience with adjuvant chemotherapy. J Cancer Res Clin Oncol. 1983;106(suppl):55–67.
Serra M, Picci P, Ferrari S, Bacci G. Prognostic value of P-glycoprotein in high-grade osteosarcoma. J Clin Oncol. 2007;25:4858–4860; author reply 4860–4861.
Shin KH, Moon SH, Suh JS, Yang WI. Tumor volume change as a predictor of chemotherapeutic response in osteosarcoma. Clin Orthop Relat Res. 2000;376:200–208.
Song WS, Cho WH, Jeon DG, Kong CB, Kim MS, Lee JA, Eid AS, Kim JD, Lee SY. Pelvis and extremity osteosarcoma with similar tumor volume have an equivalent survival. J Surg Oncol. 2010;101:611–617.
Song WS, Jeon DG, Kong CB, Cho WH, Koh JS, Lee JA, Yoo JY, Jung ST, Shin DS, Lee SY. Tumor volume increase during preoperative chemotherapy as a novel predictor of local recurrence in extremity osteosarcoma. Ann Surg Oncol. 2011;18:1710–1716.
Suresh S, Saifuddin A. Radiological appearances of appendicular osteosarcoma: a comprehensive pictorial review. Clin Radiol. 2007;62:314–323.
Wunder JS, Gokgoz N, Parkes R, Bull SB, Eskandarian S, Davis AM, Beauchamp CP, Conrad EU, Grimer RJ, Healey JH, Malkin D, Mangham DC, Rock MJ, Bell RS, Andrulis IL. TP53 mutations and outcome in osteosarcoma: a prospective, multicenter study. J Clin Oncol. 2005;23:1483–1490.
Acknowledgments
We thank Min Suk Kim, MD and Jae Soo Koh, MD for review of the pathologic specimens.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a grant of the Radiological Translational Research program, Korea Institute of Radiological & Medical Sciences (50471-2013)(D-GJ), Seoul, Korea.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Each author certifies that his or her institution approved the human protocol for this investigation, that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participation in the study was obtained.
About this article
Cite this article
Jeon, DG., Song, W.S., Cho, W.H. et al. Proximal Tumor Location and Fluid-fluid Levels on MRI Predict Resistance to Chemotherapy in Stage IIB Osteosarcoma. Clin Orthop Relat Res 472, 1911–1920 (2014). https://doi.org/10.1007/s11999-014-3521-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-014-3521-1