Abstract
Background
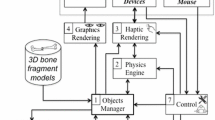
Accurate reproduction of the preoperative plan at the time of surgery is critical for wide resection of primary bone tumors. Robotic technology can potentially help the surgeon reproduce a given preoperative plan, but yielding control of cutting instruments to a robot introduces potentially serious complications. We developed a novel passive (“haptics”) robot-assisted resection technique for primary bone sarcomas that takes advantage of robotic accuracy while still leaving control of the cutting instrument in the hands of the surgeon.
Questions/Purposes
We asked whether this technique would enable a preoperative resection plan to be reproduced more accurately than a standard manual technique.
Methods
A joint-sparing hemimetaphyseal resection was precisely outlined on the three-dimensionally reconstructed image of a representative Sawbones femur. The indicated resection was performed on 12 Sawbones specimens using the standard manual technique on six specimens and the haptic robotic technique on six specimens. Postresection images were quantitatively analyzed to determine the accuracy of the resections compared to the preoperative plan, which included measuring the maximum linear deviation of the cuts from the preoperative plan and the angular deviation of the resection planes from the target planes.
Results
Compared with the manual technique, the robotic technique resulted in a mean improvement of 7.8 mm of maximum linear deviation from the preoperative plan and 7.9° improvement in pitch and 4.6° improvement in roll for the angular deviation from the target planes.
Conclusions
The haptic robot-assisted technique improved the accuracy of simulated wide resections of bone tumors compared with manual techniques.
Clinical Relevance
Haptic robot-assisted technology has the potential to enhance primary bone tumor resection. Further bench and clinical studies, including comparisons with recently introduced computer navigation technology, are warranted.






Similar content being viewed by others
References
Alyafei S, Inoue T, Zhang H, Ahmed K, Oriuchi N, Sato N, Suzuki H, Endo K. Image fusion system using PACS for MRI, CT, and PET images. Clin Positron Imaging. 1999;2:137–143.
Anstey JB, Smith EJ, Rasquinha B, Rudan JF, Ellis RE. On the use of laser scans to validate reverse engineering of bony anatomy. Stud Health Technol Inform. 2011;163:18–24.
Avedian RS, Haydon RC, Peabody TD. Multiplanar osteotomy with limited wide margins: a tissue preserving surgical technique for high-grade bone sarcomas. Clin Orthop Relat Res. 2010;468:2754–2764.
Bacci G, Ferrari S, Mercuri M, Bertoni F, Picci P, Manfrini M, Gasbarrini A, Forni C, Cesari M, Campanacci M. Predictive factors for local recurrence in osteosarcoma: 540 patients with extremity tumors followed for minimum 2.5 years after neoadjuvant chemotherapy. Acta Orthop Scand. 1998;69:230–236.
Bacci G, Forni C, Longhi A, Ferrari S, Mercuri M, Bertoni F, Serra M, Briccoli A, Balladelli A, Picci P. Local recurrence and local control of non-metastatic osteosarcoma of the extremities: a 27-year experience in a single institution. J Surg Oncol. 2007;96:118–123.
Bickels J, Wittig JC, Kollender Y, Henshaw RM, Kellar-Graney KL, Meller I, Malawer MM. Distal femur resection with endoprosthetic reconstruction: a long-term followup study. Clin Orthop Relat Res. 2002;400:225–235.
Cartiaux O, Docquier PL, Paul L, Francq BG, Cornu OH, Delloye C, Raucent B, Dehez B, Banse X. Surgical inaccuracy of tumor resection and reconstruction within the pelvis: an experimental study. Acta Orthop. 2008;79:695–702.
Cartiaux O, Paul L, Docquier PL, Raucent B, Dombre E, Banse X. Computer-assisted and robot-assisted technologies to improve bone-cutting accuracy when integrated with a freehand process using an oscillating saw. J Bone Joint Surg Am. 2010;92:2076–2082.
Cho HS, Oh JH, Han I, Kim HS. Joint-preserving limb salvage surgery under navigation guidance. J Surg Oncol. 2009;100:227–232.
Gebhardt MC, Flugstad DI, Springfield DS, Mankin HJ. The use of bone allografts for limb salvage in high-grade extremity osteosarcoma. Clin Orthop Relat Res. 1991;270:181–196.
Hoffer FA, Nikanorov AY, Reddick WE, Bodner SM, Xiong X, Jones-Wallace D, Gronemeyer SA, Rao BN, Kauffman WM, Laor T. Accuracy of MR imaging for detecting epiphyseal extension of osteosarcoma. Pediatr Radiol. 2000;30:289–298.
Horowitz SM, Glasser DB, Lane JM, Healey JH. Prosthetic and extremity survivorship after limb salvage for sarcoma. How long do the reconstructions last? Clin Orthop Relat Res. 1993;293:280–286.
International Standard, ISO 1101:2004(E). Geometrical Product Specifications (GPS)—Geometrical Tolerancing—Tolerances of Form, Orientation, Location and Run-out. 2nd ed. Geneva, Switzerland: International Organization for Standardization; 2004.
Kawai A, Lin PP, Boland PJ, Athanasian EA, Healey JH. Relationship between magnitude of resection, complication, and prosthetic survival after prosthetic knee reconstructions for distal femoral tumors. J Surg Oncol. 1999;70:109–115.
Kawai A, Muschler GF, Lane JM, Otis JC, Healey JH. Prosthetic knee replacement after resection of a malignant tumor of the distal part of the femur. Medium to long-term results. J Bone Joint Surg Am. 1998;80:636–647.
Marcove RC, Sheth DS, Healey J, Huvos A, Rosen G, Meyers P. Limb-sparing surgery for extremity sarcoma. Cancer Invest. 1994;12:497–504.
Muscolo DL, Ayerza MA, Aponte-Tinao LA, Abalo E, Farfalli G. Unicondylar osteoarticular allografts of the knee. J Bone Joint Surg Am. 2007;89:2137–2142.
Muscolo DL, Ayerza MA, Aponte-Tinao LA, Abalo E, Farfalli G. Unicondylar osteoarticular allografts of the knee. Surgical technique. J Bone Joint Surg Am. 2008;90(Suppl 2):206–217.
Muscolo DL, Ayerza MA, Aponte-Tinao LA, Ranalletta M. Use of distal femoral osteoarticular allografts in limb salvage surgery. J Bone Joint Surg Am. 2005;87:2449–2455.
Nagarajan R, Neglia JP, Clohisy DR, Robison LL. Limb salvage and amputation in survivors of pediatric lower-extremity bone tumors: what are the long-term implications? J Clin Oncol. 2002;20:4493–4501.
Nathan SS, Gorlick R, Bukata S, Chou A, Morris CD, Boland PJ, Huvos AG, Meyers PA, Healey JH. Treatment algorithm for locally recurrent osteosarcoma based on local disease-free interval and the presence of lung metastasis. Cancer. 2006;107:1607–1616.
Onikul E, Fletcher BD, Parham DM, Chen G. Accuracy of MR imaging for estimating intraosseous extent of osteosarcoma. AJR Am J Roentgenol. 1996;167:1211–1215.
Ottaviani G, Jaffe N. The epidemiology of osteosarcoma. Cancer Treat Res. 2009;152:3–13.
Pearle AD, O’Loughlin PF, Kendoff DO. Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty. 2010;25:230–237.
Raabe A, Krishnan R, Seifert V. Actual aspects of image-guided surgery. Surg Technol Int. 2003;11:314–319.
Renard AJ, Veth RP, Schreuder HW, van Loon CJ, Koops HS, van Horn JR. Function and complications after ablative and limb-salvage therapy in lower extremity sarcoma of bone. J Surg Oncol. 2000;73:198–205.
Rougraff BT, Simon MA, Kneisl JS, Greenberg DB, Mankin HJ. Limb salvage compared with amputation for osteosarcoma of the distal end of the femur. A long-term oncological, functional, and quality-of-life study. J Bone Joint Surg Am. 1994;76:649–656.
Saifuddin A. The accuracy of imaging in the local staging of appendicular osteosarcoma. Skeletal Radiol. 2002;31:191–201.
San-Julian M, Aquerreta JD, Benito A, Cañadell J. Indications for epiphyseal preservation in metaphyseal malignant bone tumors of children: relationship between image methods and histological findings. J Pediatr Orthop. 1999;19:543–548.
Shin DS, Choong PF, Chao EY, Sim FH. Large tumor endoprostheses and extracortical bone-bridging: 28 patients followed 10–20 years. Acta Orthop Scand. 2000;71:305–311.
Shin M, Penholate MF, Lefaucheur JP, Gurruchaga JM, Brugieres P, Nguyen JP. Assessing accuracy of the magnetic resonance imaging-computed tomography fusion images to evaluate the electrode positions in subthalamic nucleus after deep-brain stimulation. Neurosurgery. 2010;66:1193–1202.
So TYC, Lam YL, Mak KL. Computer-assisted navigation in bone tumor surgery: seamless workflow model and evolution of technique. Clin Orthop Relat Res. 2010;468:2985–2991.
Springer BD, Hanssen AD, Sim FH, Lewallen DG. The kinematic rotating hinge prosthesis for complex knee arthroplasty. Clin Orthop Relat Res. 2001;392:283–291.
Springfield DS, Schmidt R, Graham-Pole J, Marcus RB Jr, Spanier SS, Enneking WF. Surgical treatment for osteosarcoma. J Bone Joint Surg Am. 1988;70:1124–1130.
Sugarbaker PH, Malawer MM. Musculoskeletal Surgery for Cancer: Principles and Techniques. New York, NY, USA: Thieme Medical Publishers; 1992.
Yamanaka Y, Kamogawa J, Katagi R, Kodama K, Misaki H, Kamada K, Okuda S, Morino T, Ogata T, Yamamoto H. 3-D MRI/CT fusion imaging of the lumbar spine. Skeletal Radiol. 2010;39:285–288.
Zatsepin ST, Burdygin VN. Replacement of the distal femur and proximal tibia with frozen allografts. Clin Orthop Relat Res. 1994;303:95–102.
Acknowledgments
We thank Mako Surgical Corporation for providing the robot and required robotic engineering support used in this study at no cost to the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
The institution of one or more of the authors (AP, PJB, JHH) has received funding from MAKO Surgical Corporation (Ft Lauderdale, FL, USA). One author (CL) is an employee of MAKO Surgical Corp.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
This work was performed at Memorial Sloan-Kettering Cancer Center, New York, NY, USA; the Hospital for Special Surgery, New York, NY, USA; and Mako Surgical Corporation.
Appendix
Appendix
Calculation of various parameters of accuracy
We used reverse engineering software (Version 12.0; Studio, Geomagic, Inc, Research Triangle Park, NC, USA) to analyze the pre- and postresection three-dimensionally reconstructed laser scan images. The preoperative image contained the three target planes of the resection, ie, the three planes that were defined as part of the preoperative plan as the superior, inferior, and vertical cut planes. For each specimen, the standard best alignment function of the software was used to perfectly superimpose the preoperative and postoperative images of the Sawbones specimen; this placed the preoperative and postoperative scans in a common coordinate system. We then used the software to highlight the cortical rim of each of the three cuts, deliberately excluding the cancellous surfaces to avoid the analysis problems associated with the cancellous voids of the cancellous surface. (These voids resulted in the software selecting points not only on the surface of the cancellous bone, but also in the depths of the voids, the latter of which were clearly not in the plane of the cut.) We believed this technique was well justified, because it was unlikely that a horizontal saw blade would produce cuts in the cancellous surface that deviated wildly from the plane of the cuts defined by just the cortical rims. The small values recorded for the deviation from flatness for both the robotic and manual cuts (see Results section) support this assumption.
The cortical rims of each of the three cuts—superior, inferior, and vertical—were exported as discrete points (relative to the common coordinate system created after the preoperative and postoperative images were superimposed) and imported into a technical computing software program (Matlab; MathWorks, Natick, MA, USA) for further analysis. This software was used to help analyze the various geometric relationships between points on the resection plane (and the best-fit plane) and the target planes. Using this software, the perpendicular distance between each point and the corresponding target plane was calculated. Additionally, the angular deviations between the target plane and best-fit planes were calculated.
The data were reported in a form that was consistent with the standards of the International Organization of Standardization (ISO) (Fig. A-1) [13]. According to these standards, the location error is defined as the perpendicular distance from the target plane to the point on the cut surface furthest from the target plane. For simplicity, in the text we usually refer to location error as the maximum deviation from the preoperative plan. Although it is not an ISO standard, we also calculated the mean deviation of the points from the target plane by calculating the mean of the (absolute) distance of each point from the target plane. The flatness was calculated by measuring the amplitude, or peak-to-peak distance, of the cut surface.
The pitch and roll errors were defined as angular deviations between the best-fit plane and the target plane along the axis of the blade and the front edge of the blade, in that order. Specifically, the roll error refers to a rotation about the AP axis of the bone; the pitch error refers to a rotation about the superoinferior axis for the medial cut and the mediolateral axis for the superior and inferior cuts. For the best-fit planes through the superior and inferior limbs of the cuts, the deviation from parallel was defined as the angle between these two planes.
About this article
Cite this article
Khan, F., Pearle, A., Lightcap, C. et al. Haptic Robot-assisted Surgery Improves Accuracy of Wide Resection of Bone Tumors: A Pilot Study. Clin Orthop Relat Res 471, 851–859 (2013). https://doi.org/10.1007/s11999-012-2529-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-012-2529-7