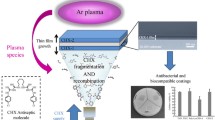
Abstract
Chitosan is an exciting alternative for the development of coating-surfaces due to its large action spectrum against pathogenic microorganisms. However, to produce a stable coating with effective antibacterial action, a compromise between deacetylation degree (DD) and molecular weight (MW) is essential. Four chitosan samples were characterized regarding Mw and DD and correlated with the minimum and bactericide concentrations against E. coli, P. aeruginosa, and S. aureus. CHI80MW (79.7% DD and 7.0 × 105 Da) showed the best antibacterial effect and was selected to functionalize polytetrafluoroethylene (PTFE) surfaces by plasma. CHI80MW was grafted onto the PTFE surfaces using two different spacer molecules: poly(ethylene glycol) bis (carboxymethyl) ether (PEG) and poly(ethylene-alt-maleic anhydride) (PA). PTFE-Plasma-PA-CHI80MW exhibited a coating with more attached chitosan and better antibacterial action if compared to PTFE-Plasma-PEG-CHI80MW: after 8 h, PTFE-Plasma-PEG-CHI80MW presented a bacterial reduction of 25-30% for the three bacterial strains, and PTFE-Plasma-PA-CHI80MW reduced them to 77-90%. Moreover, cytotoxicity tests showed that PTFE-Plasma-PA-CHI80MW samples were compatible with human fibroblasts.






Similar content being viewed by others
Data availability
All the data included in this study are available upon request by contacting the corresponding author.
References
Costerton, JW, Stewart, PS, Greenberg, EP, “Bacterial biofilms: A common cause of persistent infections.” Science, 284 1318–1322 (1999)
Borkow G, editor. Use of Biocidal Surfaces for Reduction of Healthcare Acquired Infections [Internet]. Springer International Publishing; 2014 [cited 2020 Mar 31]. https://www.springer.com/gp/book/9783319080567.
Kramer, A, Schwebke, I, Kampf, G, “How long do nosocomial pathogens persist on inanimate surfaces? A systematic review.” BMC Infect. Dis., 6 130 (2006)
Campoccia, D, Montanaro, L, Arciola, CR, “A review of the biomaterials technologies for infection-resistant surfaces.” Biomaterials, 34 8533–8554 (2013)
Ogino, A, Kral, M, Yamashita, M, et al. “Effects of low-temperature surface-wave plasma treatment with various gases on surface modification of chitosan.” Appl. Surf. Sci., 255 2347–2352 (2008)
Vaz J, Michel E, Chevallier P, et al. "Covalent grafting of chitosan on plasma-treated polytetrafluoroethylene surfaces for biomedical applications." J. Biomater. Tissue Eng., 4 (11), 915–924 (2014)
Kong, M, Chen, X, Xing, K, et al. “Antimicrobial properties of chitosan and mode of action: A state of the art review.” Int. J. Food Microbiol., 144 51–63 (2010)
Mourya, VK, Inamdar, NN, “Chitosan-modifications and applications: Opportunities galore.” React. Funct. Polym., 68 1013–1051 (2008)
Xu Y, Liu J, Guan S, et al. “A dual pH and redox-responsive Ag/AgO/carboxymethyl chitosan composite hydrogel for controlled dual drug delivery.” J. Biomater. Sci. Polym. Ed., 31 (13) 1706–1721 (2020)
Fan Z, Cheng P, Wang D, et al. “Design and investigation of salecan/chitosan hydrogel formulations with improved antibacterial performance and 3D cell culture function.” J. Biomater. Sci. Polym. Ed., 31 (17) 2268–2284 (2020)
Lavertu, M, Xia, Z, Serreqi, AN, et al. “A validated 1H NMR method for the determination of the degree of deacetylation of chitosan.” J. Pharm. Biomed. Anal., 32 1149–1158 (2003)
Clinical and Laboratory Standards Institute, editor. “Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: M07-A10; approved standard.” 10th ed. Wayne, PA: Committee for Clinical Laboratory Standards; 2015.
Huang, M, Ma, Z, Khor, E, et al. “Uptake of FITC-chitosan nanoparticles by A549 cells.” Pharm. Res., 19 1488–1494 (2002)
Tokura, S, Ueno, K, Miyazaki, S, et al. “Molecular weight dependent antimicrobial activity by chitosan.” Macromol. Symp., 120 1–9 (1997)
Guerreiro, SG, Brochhausen, C, Negrão, R, et al. “Implanted neonatal human dermal fibroblasts influence the recruitment of endothelial cells in mice.” Biomatter., 2 43–52 (2012)
Varma SR, Sivaprakasam TO, Mishra A, et al. “Protective effects of triphala on dermal fibroblasts and human keratinocytes.” PloS One, e0145921 (2016)
Dee, GJ, Rhode, O, Wachter, R, “Chitosan: Multi-functional marine polymer.” Chitosan Multi-Funct. Mar. Polym., 116 39–44 (2001)
Hernandez-Montelongo, J, Lucchesi, EG, Gonzalez, I, et al. “Hyaluronan/chitosan nanofilms assembled layer-by-layer and their antibacterial effect: A study using Staphylococcus aureus and Pseudomonas aeruginosa.” Colloids Surf. B Biointerfaces, 141 499–506 (2016)
Vasilev, K, Cook, J, Griesser, HJ, “Antibacterial surfaces for biomedical devices.” Expert Rev. Med. Devices, 6 553–567 (2009)
Hernandez-Montelongo, J, Corrales Ureña, YR, Machado, D, et al. “Electrostatic immobilization of antimicrobial peptides on polyethylenimine and their antibacterial effect against Staphylococcus epidermidis.” Colloids Surf. B Biointerfaces, 164 370–378 (2018)
Liu, H, Du, Y, Wang, X, et al. “Chitosan kills bacteria through cell membrane damage.” Int. J. Food Microbiol., 95 147–155 (2004)
Zivanovic, S, Chi, S, Draughon, AF, “Antimicrobial activity of chitosan films enriched with essential oils.” J. Food Sci., 70 M45–M51 (2005)
Croisier, F, Jérôme, C, “Chitosan-based biomaterials for tissue engineering.” Eur. Polym. J., 49 780–792 (2013)
Dash, M, Chiellini, F, Ottenbrite, RM, et al. “Chitosan—A versatile semi-synthetic polymer in biomedical applications.” Prog. Polym. Sci., 36 981–1014 (2011)
Funding
This work was supported by the Sao Paulo Research Foundation (Grant No 2013/05135-1), Coordination for the Improvement of Higher Education Personnel (CAPES: Procad 88882.151600/2017-01, Proex 33003017034P8), and National Council for Scientific and Technological Development (CNPq 421039/2016-7).
Author information
Authors and Affiliations
Contributions
JMV and RSV conceived and designed experiments; LMCGF performed the characterization of chitosan powders and obtained the MIC and MBC values for the chitosan powders; JMV functionalized the PTFE surfaces with chitosan by plasma grafting; JHM performed the photoluminescence characterization; JMV performed the antibacterial tests; CR performed the cytocompatibility tests; JMV, CR, and JHM analyzed the data; MMB and RSV contributed reagents/materials/analysis tools; JMV, TBT, JHM, and RSV wrote the manuscript. All authors revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vaz, J.M., Taketa, T.B., Hernandez-Montelongo, J. et al. Antibacterial noncytotoxic chitosan coatings on polytetrafluoroethylene films by plasma grafting for medical device applications. J Coat Technol Res 19, 829–838 (2022). https://doi.org/10.1007/s11998-021-00560-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11998-021-00560-3