Abstract
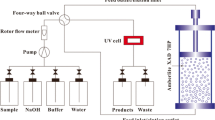
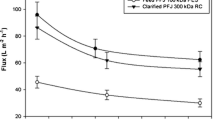
Obtaining native protein from potato fruit water (PFW) acceptable for food consumption was attempted by comparing expanded bed adsorption (EBA) and ultrafiltration (UF).The methods were assessed on their process performance and the product quality. Extractable tuber proteins were recovered from lab-prepared PFW either by adsorption to an EBA column using a mixed mode resin (0.31 L) or by batch concentration in an UF (10 kDa MWCO, 0.093 m2) unit and then freeze dried. The yields on protein and esterase activity were higher (p < 0.05 and p < 0.01; Mann–Whitney U-test) in UF (3.2 g l−1 PFW and 3.17 kU l−1 PFW) than in EBA (1.8 and 1.21). The performance difference was also reflected in process productivity for esterase activity which was fivefold better (p < 0.01) in UF (4.30 kU h−1) than with EBA (0.84) due to the higher enzyme retention; protein productivities were the same. The content of solanidine glycoalkaloids was depleted to moderate levels but came out unaffected by the processing method: EBA 286 ppm, UF 213 ppm. The low levels of chlorogenic acid in all EBA preparations were negatively correlated to high brightness score (L* = 73.8%), a favorable attribute in food-quality proteins. Both methodologies produced native preparations of comparable protein content (75%). EBA processing, however, increased the fraction of the patatin protein which may offer advantages in food applications.






Similar content being viewed by others
Abbreviations
- C :
-
Concentration outlet
- C 0 :
-
Concentration inlet
- CF:
-
Concentration factor
- CV:
-
Column volume of settled resin
- CQA:
-
Chlorogenic acid
- EAAI:
-
Essential amino acid index
- EBA:
-
Expanded bed adsorption
- expCV:
-
Column volume of the expanded resin
- L*:
-
Brightness
- MiMo:
-
Mixed mode
- MWCO:
-
Molecular weight cutoff
- PFW:
-
Potato fruit water
- PI:
-
Protease inhibitor
- R app :
-
Apparent rejection coefficient
- RO:
-
Reverse osmosis
- TGA:
-
Total glycoalkaloids
- TMP:
-
Transmembrane pressure
- UF:
-
Ultrafiltration
References
Alt, V., Steinhof, R., Lotz, M., Ulber, R., Kasper, C., & Scheper, T. (2005). Optimization of glycoalkaloid analysis for use in industrial potato fruit juice downstreaming. Engineering in Life Sciences, 5(6), 562–567.
Andersson, J., Sahoo, D., & Mattiasson, B. (2008). Isolation of potato proteins using simulated moving bed technology. Biotechnology and Bioengineering, 101(6), 1256–1263.
Bartova, V., & Barta, J. (2009). Chemical composition and nutritional value of protein concentrates isolated from potato (Solanum tuberosum L.) fruit juice by precipitation with ethanol or ferric chloride. Journal of Agricultural and Food Chemistry, 57(19), 9028–9034.
Bidinger, S. C. (1992). Wertstoffgewinnung aus Abwässern/Recovery of valuable substances from waste water. Internationale Zeitschrift für Lebensmittel-Technologie (ZFL), 43(5), 222–228.
Bohac, J. R. (1991). A modified method to purify patatin from potato-tubers. Journal of Agricultural and Food Chemistry, 39(8), 1411–1415.
Bolivar, J. M., Batalla, P., Mateo, C., Carrascosa, A. V., Pessela, B. C., & Guisána, J. M. (2010). Selective adsorption of small proteins on large-pore anion exchangers coated with medium size proteins. Colloids and Surfaces, B: Biointerfaces, 78, 140–145.
Byrne, D. (2002). Commission decision of 15 February 2002 authorizing the placing on the market of coagulated potato proteins and hydrolysates thereof as novel food ingredients under Regulation (EC) No 258/97 of the European Parliament and of the Council. Official Journal of the European Communities, L, 50, 92–93.
Claussen, I.C., Strømmen, I., Egelandsdal, B., Strætkvern, K.O. (2007). Effects of drying methods on functionality of a native potato protein concentrate drying technology, 25(6), 1091–1098.
D’Alvise, N., Lesueur-Lambert, C., Fertin, B., Dhulster, P., & Guillochon, D. (2000). Removal of polyphenols and recovery of proteins from alfalfa white protein concentrate by ultrafiltration and adsorbent resin separations. Separation Science and Technology, 35(15), 2453–2472.
Dao, L., & Friedman, M. (1992). Chlorogenic acid content of fresh and processed potatoes determined by ultraviolet spectrophotometry. Journal of Agricultural and Food Chemistry, 40(11), 2152–2156.
Eriksson, G., & Sivik, B. (1976). Ultrafiltration of potato process water—influence of processing variables. Potato Research, 19, 279–287.
Giuseppin, M.L.F., van der Sluis, C., Laus, M.C. (2008). Native potato protein isolates. Patent No WO 2008/069650.
Graf, A. M., Steinhof, R., Lotz, M., Tippkotter, N., Kasper, C., Beutel, S., et al. (2009). Downstream-processing with membrane adsorbant on the isolation of native protein fractions from potato amniotic liquid. Chemie Ingenieur Technik, 81(3), 267–274.
Lihme, A., Hansen, M., Olander, M., & Zafirakos, E. (2000). Expanded bed adsorption in the purification of biomolecules. In Desai (Ed.), Methods in biotechnology (pp. 121–141). Totowa: Humana.
Løkra, S., & Strætkvern, K. O. (2009). Industrial proteins from potato juice. A review. Food, 3(Special Issue 1), 88–95.
Løkra, S., Helland, M. H., Claussen, I. C., Strætkvern, K. O., & Egelandsdal, B. (2008). Chemical characterization and functional properties of a potato protein concentrate prepared by large-scale expanded bed adsorption chromatography. LWT Food Science and Technology, 41(6), 1089–1099.
Løkra, S., Schüller, R. B., Egelandsdal, B., Engebretsen, B., & Strætkvern, K. O. (2009). Comparison of composition, enzyme activity and selected functional properties of potato proteins isolated from potato juice with two different expanded bed resins. LWT Food Science and Technology, 42(4), 906–913.
Nabetani, H., Abbott, T. P., & Kleiman, R. (1995). Optimal separation of jojoba protein using membrane processes. Industrial and Engineering Chemistry Research, 34(5), 1779–1788.
Oosten, B. J. (1976). Ultrafiltration of potato juice results in high yield of protein. Starch/Stärke, 28(4), 135–137.
Osman, A., El Agha, A., Makris, D. P., & Kefalas, P. (2009). Chlorogenic acid oxidation by a crude peroxidase preparation: biocatalytic characteristics and oxidation products. Food and Bioprocess Technology. doi:10.1007/s11947-009-0241-8.
Ralet, M. C., & Guéguen, J. (2000). Fractionation of potato proteins: solubility, thermal coagulation and emulsifying properties. LWT Food Science and Technology, 33, 380–387.
Rausch, K. D. (2002). Front end to backpipe: membrane technology in the starch processing industry. Starch/Stärke, 54, 273–284.
Rüffer, H., Kremser, U., & Seekamp, M. (1997). Experiences with reverse osmosis pilot plant for the concentration of potato fruit water in the potato starch industry. Starch/Stärke, 49(9), 354–359.
Stevens, L. H., & Davelaar, E. (1996). Isolation and characterization of blackspot pigments from potato tubers. Phytochemistry, 42(4), 941–947.
Strætkvern, K. O., Schwarz, J. G., Wiesenborn, D. P., Zafirakos, E., & Lihme, A. (1999). Expanded bed adsorption for recovery of patatin from crude potato juice. Bioseparation, 7(6), 333–345.
Strætkvern, K.O., Aae Olander, M., Lihme, A. (2002). EBA processing of potato fruit water on mixed mode adsorbent for functional protein recovery: A difficult separation task made possible. In: DOWNSTREAM—Proceedings 4th International Conference on Expanded Bed Adsorption; EBA 02 St. Petersburg Beach, Florida, USA, pp 51–52. Amersham Biosciences, Uppsala, Sweden
Strætkvern, K. O., Løkra, S., Olander, M. A., & Lihme, A. (2005). Food-grade protein from industrial potato starch effluent recovered by an expanded bed adsorption process. Journal of Biotechnology, 118, S33–S33.
Tsagaraki, E. V., & Lazarides, H. N. (2010). Fouling analysis and performance of tubular ultrafiltration on pretreated olive mill waste water. Food and Bioprocess Technology. doi:10.1007/s11947-010-0326-4.
van Koningsveld, G. A., Walstra, P., Voragen, A. G. J., Kuijpers, I. J., Van Boekel, M., & Gruppen, H. (2006). Effects of protein composition and enzymatic activity on formation and properties of potato protein stabilized emulsions. Journal of Agricultural and Food Chemistry, 54(17), 6419–6427.
Wojnowska, I., Poznanski, S., & Bednarski, W. (1981). Processing of potato protein concentrates and their properties. Journal of Food Science, 47, 167–172.
Xu, L., & Diosady, L. L. (2002). Removal of phenolic compounds in the production of high-quality canola protein isolates. Food Research International, 35, 23–30.
Yang, H., & Etzel, M. R. (2003). Evaluation of three kinetic equations in models of protein purification using ion-exchange membranes. Industrial and Engineering Chemistry Research, 42, 890–896.
Zwijnenberg, H. J., Kemperman, A. J. B., Boerrigter, M. E., Lotz, M., Dijksterhuis, J. F., Poulsen, P. E., et al. (2002). Native protein recovery from potato fruit juice by ultrafiltration. Desalination, 144, 331–334.
Acknowledgements
We are grateful for the technical assistance of Mr. Matthew Whittiker, UMES food science technician, on potato juice extraction and various analytical procedures and of Mrs. Else Berit Stenseth, HUC senior lab technician, for performing the CQA analysis. Dr. Arne Linløkken, HUC, is acknowledged for the valuable advice on statistical analysis. We are especially indebted to Dr. Sacha Beutel, at the Institut für Technische Chemie, Gottfried Wilhelm Leibniz Universität Hannover, for providing the HPLC analysis of glycoalkaloids.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Strætkvern, K.O., Schwarz, J.G. Recovery of Native Potato Protein Comparing Expanded Bed Adsorption and Ultrafiltration. Food Bioprocess Technol 5, 1939–1949 (2012). https://doi.org/10.1007/s11947-010-0494-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-010-0494-2