Abstract
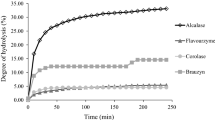
Enzymatic hydrolysis of proteins from low commercial value fish could be produced for uses like functional ingredients in a wide and always increasing zone of application in different food products. The objective of this work was to evaluate the functional properties and the amino acid profile of enzymatic hydrolysates from Bluewing searobin (Prionotus punctatus), using two microbial enzymes, Alcalase and Flavourzyme. The enzymatic hydrolysate obtained through the addition of the enzyme Alcalase reached the maximum solubility (42%) at pH 9, water holding capacity (WHC) of 2.4 gwater gprotein −1, 4.5 goil gprotein −1 of oil holding capacity (OHC) and an emulsifying activity index (EAI) of 54 m2 gsolids −1 at pH 3. On the other hand, the hydrolysate obtained from Flavourzyme attained 38% of solubility at pH 9, 3.7 gwater gprotein −1 and 5.5 goil gprotein −1 for the holding capacities, and an EAI of 71 m2 gsolids −1 at pH 11. The hydrolysate with Flavourzyme produced best results for WHC, OHC, and EAI because it had solubility lower than the hydrolysate of Alcalase. The hydrolysate produced by Alcalase had a higher amino acid content compared with Flavourzyme’s hydrolysate. However, both showed a good essential amino acid amounts. In general, these results indicate the potential utilization of the hydrolysate from Bluewing searobin in food formulations for the direct human consumption.



Similar content being viewed by others
References
Abdul-Hamid, A., Bakar, J., & Bee, G. H. (2002). Nutritional quality of spray dried protein hydrolysate from Black tilapia (Oreochromis mossambicus). Food Chemistry, 78, 69–74.
Adler-Nissen, J. (1986). A review of food hydrolysis specific areas. In enzymic hydrolysis of food proteins (pp. 57–109). Copenhagen: Elsevier Applied Science Publishers.
AOAC. (1995). Official methods of analysis of international (16th ed.). Virginia: Arlington.
Casarin, F., Cladera-Olivera, F., & Brandelli, A. (2008). Use of poultry byproduct for production of keratinolytic enzymes. Food and Bioprocess Technology, 1, 301–305.
Chandi, G. K., & Sogi, D. S. (2007). Functional properties of rice bran protein concentrates. Journal of Food Engineering, 79, 592–597.
Cheftel, J. C., Cuq, J. L., & Lorient, D. (1989). Proteínas alimentarías. Zaragoza: Acribia.
Chothia, C. (1974). Hydrophobic bonding and accessible surface-area in proteins. Nature, 9, 248–338.
Chothia, C. (1975). Structural invariants in protein folding. Nature, 8, 254–304.
Contreras-Guzmán, E. S. (1994). Bioquímica de Pescados e Derivados, FUNEP: São Paulo.
Demetriades, K., Coupland, J. N., & McClements, D. J. (1997). Physical properties of whey protein stabilized emulsions as related to pH and NaCl. Journal of Food Science, 62, 342–347.
Diniz, F. M., & Martin, A. M. (1996). Use of response surface methodology to describe the combined effects of pH, temperature and E/S ratio on the hydrolysis of dogfish (Squalus acanthias) muscle. International Journal of Food Science & Technology, 31, 419–426.
Dumay, J., Donnay-Moreno, C., Barnathan, G., Jaquen, P., & Bergé, J. P. (2006). Improvement of lipid and phospholipids recoveries from sardine (Sardina pilchardus) viscera using industrial proteases. Process Biochemistry, 41, 2327–2332.
El khalifa, A. E. O., Schiffler, B., & Bernhard, R. (2005). Effect of fermentation on the functional properties of sorghum flour. Food Chemistry, 92, 1–5.
Gauthier, S. F., Paquin, P., Pouliot, Y., & Turgeon, S. (1993). Surface activity and related functional properties of peptides obtained from whey proteins. Journal of Dairy Science, 76, 321–328.
Gildberg, A. (1993). Enzymatic processing of marine raw materials. Process Biochemistry, 28, 1–15.
Gildberg, A., Arnesen, J. A., & Carlehög, M. (2002). Utilization of cod backbone by biochemical fractionation. Process Biochemistry, 38, 475–480.
Guerard, F., Sumaya-Martinez, M. T., Laroque, D., Chabeaud, A., & Dufosse, L. (2007). Optimization of free radical scavenging activity by response surface methodology in the hydrolysis of shrimp processing discards. Process Biochemistry, 42, 1486–1491.
Hoyle, N., & Merritt, J. H. (1994). Quality of fish protein hydrolysates from herring (Clupea harengus). Journal of Food Science, 59, 76–79.
Idouraine, A., Yense, S. B., & Weber, C. W. (1991). Terapy bean flour, albumin and globulin fractions properties compared with soy protein isolate. Journal of Food Science, 56, 1316–1318.
Je, J. K. Y., Qian, Z. J., Byun, H. G., & Kim, S. K. (2007). Purification and characterization of an antioxidant peptide obtained from tuna backbone protein by enzymatic hydrolysis. Process Biochemistry, 42, 840–846.
Jeon, Y. J., Hee-Guk, B., & Se-Kwon, K. (1999). Improvement of functional properties of cod frame protein hydrolysates using ultrafiltration membranes. Process Biochemistry, 35, 471–478.
Kristinsson, H. G., & Rasco, B. A. (2000). Kinetics of the hydrolysis of Atlantic salmon (Salmon solar) muscle proteins by alkaline proteases and a visceral serine protease mixture. Process Biochemistry, 36, 131–139.
Lempek, T. S., Martins, V. G., & Prentice, C. H. (2007). Rheology of surimi-based products from fatty fish underutilized by the industry: Argentine croaker (Umbrina canosai). Journal of Aquatic Food Product Technology, 16(4), 27–44.
Liaset, B., & Espe, M. (2008). Nutritional composition of soluble and insoluble fractions obtained by enzymatic hydrolysis of fish-raw materials. Process Biochemistry, 43, 42–48.
Liceaga-Gesualdo, A. M., & Li-Chan, E. C. Y. (1999). Functional properties of fish protein hydrolysate from herring (Clupea harengus). Journal of Food Science, 64, 1000–1004.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry, 193, 265–275.
McNairney, J. (1984). Modification of a novel protein product. Journal of Chemistry, Technology and Biotechnology. B: Biotechnology, 34, 206–214.
Maldonado, A. S. (1994). Comparación de quatro métodos para la recuperación de proteínas solubles eliminadas en la elaboración de surimi. Instituto Tecnológico Pesquero del Peru, 4, 15–26.
Mahmoud, M. I. (1994). Physicochemical and functional properties of protein hydrolysates in nutritional products. Food Technology, 58, 89–95.
Márquez, U. M. L., Mira, N. V. M., & Neves, R. A. M. (2004). Caracterização de hidrolisados enzimáticos de pescado. Ciência e Tecnologia de Alimentos, 24, 101–108.
Martin, R. E., Flick, G. J., Hebard, C. E., & Ward, D. D. (1982). Chemistry and biochemistry of marine food products. Westport: AVI Publishing Company.
Martins, V. G., Costa, J. A. V., & Prentice-Hernández, C. (2009). Hidrolisado protéico de pescado obtido por vias química e enzimática a partir de corvina (Micropogonias furnieri). Química Nova, 32, 61–66.
Morais, C., Mantovani, D. M. B., & Carvalho, C. R. L. (1992). Rendimento cárneo e composição química da ictiofauna acompanhante na captura do camarão sete barbas (Xiphopenaeus kroyeri). Coletânea do Instituto de Tecnologia de Alimentos, 22, 62–72.
Morr, V., German, B., Kinsella, J. E., Regenstein, J. M., Van Buren, J. P., Kilara, A., et al. (1985). A collaborative study to develop a standardized food protein solubility procedure. Journal of Food Science, 50, 1715–1718.
Mullally, M. M., O’Callaghan, D. M., Fitzgerald, R. J., Donnelly, W. J., & Dalton, J. P. (1995). Zymogen activation in pancreatic endoproteolytic preparations and influence on some whey protein characteristics. Journal of Food Science, 60, 227–233.
Nakai, S. (1983). Structure-function relationships of food proteins with an emphasis on the importance of protein hydrophobicity. Journal of Agricultural and Food Chemistry, 31, 676–683.
Nilsang, S., Lertsiti, S., Suphantharika, M., & Assavanig, A. (2005). Optimization of enzymatic hydrolysis of fish soluble concentrate by commercial proteases. Journal of Food Engineering, 70, 571–578.
Nolsoe, H., & Undeland, I. (2009). The acid and alkaline solubilization process for the isolation of muscle proteins. Food and Bioprocess Technology, 2, 1–27.
Normah, I., Jamilah, B., Saari, N., & Yaakob, B. C. M. (2005). Optimization of hydrolysis conditions for the production of threadfin bream (Nemipterus japonicus) hydrolysate by Alcalase. Journal of Muscle Foods, 16, 87–102.
Orban, E., Quaglia, G. B., Casini, I., Caproni, R., & Moneta, E. (1992). Composition and functionality of sardine native proteins. Lebensmittel-Wissenschaft und Technologie, 25, 371–373.
Pearce, K. N., & Kinsella, J. E. (1978). Emulsifying properties of proteins: evaluation of turbidimetric technique. Journal of Agriculture and Food Chemistry, 26, 716–723.
Poustie, U. J., & Rutherford, P. (2002). Dietary intervention for phenylketnuria. Oxford: The Cochrane Library.
Quaglia, G. B., & Orban, E. (1987a). Enzymatic solubilisation of proteins of sardine (Sardina pilchardus) by commercial proteases. Journal of Science and Food Agriculture, 38, 263–269.
Quaglia, G. B., & Orban, E. (1987b). Influence of the degree of hydrolysis on the solubility of protein hydrolysates from sardine (Sardina pilchardus). Journal of Science and Food Agriculture, 38, 271–276.
Rebeca, B. D., Peña-Vera, M. T., & Díaz-Castañeda, M. (1991). Production of fish protein hydrolysates with bacterial proteases; yield and nutritional value. Journal of Food Science, 56, 309–314.
Rodríguez-Ambriz, S. L., Martínez-Ayala, A. L., Millán, F., & Dávila-Ortiz, G. (2005). Composition and functional properties of Lupinus campestris protein isolates. Plant Foods for Human Nutrition, 60, 99–107.
Rosa, C. S. (2000). Estudo das propriedades funcionais do colágeno obtido da pele de frango. Dissertação em Mestrado em Ciência e Tecnologia de Alimentos. Universidade Federal de Santa Maria. Santa Maria: UFSM.
Safari, R., Motamedzadegan, A., Ovissipour, M., Regenstein, J. M., Gildberg, A., & Rasco, B. (2009). Use of hydrolysates from yellowfin tuna (Thunnus albacares) heads as a complex nitrogen source for lactic acid bacteria. Food and Bioprocess Technology. doi: 10.1007/s11947-009-0225-8.
Santos, S. D., Martins, V. G., Salas-Mellado, M., & Prentice-Hernández, C. (2009). Otimização dos parâmetros de produção de hidrolisados protéicos enzimáticos utilizando pescado de baixo valor comercial. Química Nova, 32, 72–77.
Skanderby, M. (1994). Protein hydrolysates: their functionality and applications. European Food Technology International, 10, 141–144.
Slizyte, R., Dauksas, E., Falch, E., Storrro, I., & Rustad, T. (2005). Characteristics of protein fractions generated from hydrolysed cod (Gadus morhua) by-products. Process Biochemistry, 40, 2021–2033.
Acknowledgements
The authors wish to thank the Coordination for Improvement of Superior Education Personal of Brazil (CAPES) and National Council for Scientific and Technological Development of Brazil (CNPq) for financial support to carry out experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
dos Santos, S.D., Martins, V.G., Salas-Mellado, M. et al. Evaluation of Functional Properties in Protein Hydrolysates from Bluewing Searobin (Prionotus punctatus) Obtained with Different Microbial Enzymes. Food Bioprocess Technol 4, 1399–1406 (2011). https://doi.org/10.1007/s11947-009-0301-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-009-0301-0