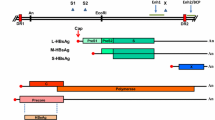
Abstract
Purpose of review
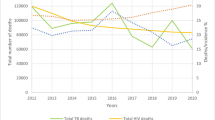
Hepatitis C virus (HCV) infection is a principal cause of morbidity, mortality, and economic burden, and the global elimination of HCV is now considered a public health objective. Globally, an estimated 71 million have chronic HCV infection with an estimated annual mortality of 3,99,000.
Recent findings
The 69th World health Assembly has endorsed the global HCV elimination strategy by 2030, which is made possible because of all oral, cost-effective, efficacious, generic direct-acting antivirals. The public health strategy targets not only diagnosed chronic hepatitis C patients but also asymptomatic undiagnosed HCV-infected individuals, persons living with human immunodeficiency virus (HIV), post-transplant, thallasemia, persons who inject drugs, and the pediatric population.
Summary
Mobilization of state-sponsored HCV elimination programs in a global coalition against HCV has taken root in Egypt, India, Georgia, Mongolia, China, Australia, and Brazil. This review summarizes current HCV elimination initiatives in terms of medical progress, barriers to HCV elimination, and current gaps in knowledge and social outreach from the public health perspective.

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance
Polaris Observatory HCV Collaborators. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol Hepatol. 2017;2:161–76.
Gower E, Estes C, Blach S, Razavi-Shearer K, Razavi H. Global epidemiology and genotype distribution of the hepatitis C virus infection. J Hepatol. 2014;61:S45–57.
Ward JW, Holtzman DA, Hepatitis C. In: Boyer TD, Manns MP, Sanyal AJ, et al., editors. Zakim and Boyer’s hepatology: a textbook of liver disease. 7th ed. London: Elsevier Health Sciences; 2017. p. 428–45.
World Health Organization. Global Health Sector Strategy on Viral Hepatitis 2016–2021: towards ending viral hepatitis WHO/HIV/2016.06. Available at: http://www.who.int/hepatitis/strategy2016-2021/ghss-hep/en/. Accessed 15 Dec 2018. This WHO document indicates the work of the Global Coalition against HCV and the methodology adopted in countries to suit their individual need and purpose.
Marshall AD, Pawlotsky JM, Lazarus JV, Aghemo A, Dore GJ, Grebely J. The removal of DAA restrictions in Europe - one step closer to eliminating HCV as a major public health threat. J Hepatol. 2018;69(5):1188–96. https://doi.org/10.1016/j.jhep.2018.06.016.
Draft global health sector strategies. Viral hepatitis, 2016–2021 Sixty-Ninth World Health Assembly. Available at: http://apps.who.int/gb/ebwha/pdf_files/WHA69/A69_32-en.pdf?ua ¼ 1. Accessed 18 Dec 2018.
Cochi SL, Dowdle WR. Introduction 2010. In: Cochi SL, Dowdle WR, editors. Disease eradication in the 21st century: implications for global health. Cambridge: The MIT Press. p. 33–61.
Pradat P, Virlogeux V, Trépo E. Epidemiology and elimination of HCV-related liver Disease. Viruses. 2018;(10):10, E545. https://doi.org/10.3390/v10100545.
Morris MD, Shiboski S, Bruneau J, Hahn JA, Hellard M, Prins M, et al. Geographic differences in temporal incidence trends of hepatitis C virus infection among people who inject drugs: the InC3 collaboration. Clin Infect Dis. 2017;64:860–8.
WHO. Global Hepatitis Report 2017. Geneva: World Health Organization; 2017. Licence: CC BY-NC-SA 3.0 IGO
Dhiman RK, Satsangi S, Grover GS, Puri P. Tackling the hepatitis C disease burden in Punjab, India. J Clin Exp Hepatol. 2016;6:224–32.
Dhiman R, Grover G, Premkumar M, et al. Decentralized care is effective in the management of patients with hepatitis C in a public health care setting: the Punjab Model. European Association for the Study of the Liver 2018 Congress. Hepatology. 2018;68(suppl 1):S37–64.
Omran D, Alboraie M, Zayed RA, Wifi MN, Naguib M, Eltabbakh M, et al. Towards hepatitis C virus elimination: Egyptian experience, achievements and limitations. World J Gastroenterol. 2018;24(38):4330–40. https://doi.org/10.3748/wjg.v24.i38.4330.
Dore GJ, Hajarizadeh B. Elimination of hepatitis C virus in Australia: laying the foundation. Infect Dis Clin North Am. 2018;32:269–79.
Haridy J, Wigg A, Muller K, Ramachandran J, Tilley E, Waddell V, et al. Real-world outcomes of unrestricted direct-acting antiviral treatment for hepatitis HCV in Australia: the South Australian statewide experience. J Viral Hepat. 2018;25:1287–97.
El Kassas M, Elbaz T, Elsharkawy A, et al. HCV, prevention, treatment and key barriers to elimination. Expert Rev Anti-Infect Ther. 2018;16:345–50.
Nasrullah M, Sergeenko D, Gamkrelidze A, Averhoff F. HCV elimination—lessons learned from a small Eurasian country, Georgia. Nat Rev Gastroenterol Hepatol. 2017;14:447–8.
Olafsson S, Tyrfingsson T, Runarsdottir V. Treatment as prevention for hepatitis C (TraP Hep C)—a nationwide elimination programme in Iceland using direct-acting antiviral agents. J Intern Med. 2018;283:500–7.
Kracht PAM, Arends JE, van Erpecum KJ, Urbanus A, Willemse JA, Hoepelman AIM, et al. Strategies for achieving viral hepatitis C micro-elimination in the Netherlands. Hepatol Med Policy. 2018;3:12. https://doi.org/10.1186/s41124-018-0040-9.
Tsertsvadze T, Gamkrelidze A, Chkhartishvili N, Abutidze A, Sharvadze L, Kerashvili V, et al. Three years of progress towards achieving hepatitis C elimination in the country of Georgia, April 2015 – March 2018. Hepatology. 2018;68(Suppl-1):32A–3A.
Shiha G, Soliman R, Mikhail N. Easterbrook P. Educate, Test and treat model towards elimination of hepatitis C in Egypt: implementation in 63 villages. Hepatology. 2018;68(Suppl-1):33A.
Elsharkawy A, Fouad R, El Akel W, et al. Sofosbuvir-based treatment regimens: real life results of 14 409 chronic HCV genotype 4 patients in Egypt. Aliment Pharmacol Ther. 2017;45:681–7.
Estes C, Abdel-Kareem M, Abdel-Razek W, Abdel Sameea E, Abuzeid M, Gomaa A, et al. Economic burden of hepatitis C in Egypt: the future impact of highly effective therapies. Aliment Pharmacol Ther. 2015;42(5):696–706. https://doi.org/10.1111/apt.13316.
Sood A, Suryaprasad A, Trickey A, Kanchi S, Midha V, Foster MA, et al. The burden of hepatitis C virus infection in Punjab, India: a population-based serosurvey. PLoS ONE. 2018;13(7):e0200461. https://doi.org/10.1371/journal.pone.0200461.
National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Population Health and Public Health Practice. Committee on a National Strategy for the Elimination of Hepatitis B and C. In: Strom BL, Buckley GJ, editors. A national strategy for the elimination of hepatitis B and C: phase two report. Washington, DC: National Academies Press; 2017.
Shy R, Cui Y. Global analysis of a mathematical model for Hepatitis C virus transmissions. Virus Res. 2016;17(8–17).
Degenhardt L, Peacock A, Colledge S, Leung J, Grebely J, Vickerman P, et al. Global prevalence of injecting drug use and sociodemographic characteristics and prevalence of HIV, HBV, and HCV in people who inject drugs: a multistage systematic review. Lancet Glob Health. 2017;5:e1192–207.
WHO. Progress report on access to hepatitis C treatment: focus on overcoming barriers in low- and middle-income countries, March 2018. Geneva: World Health Organization; 2018. (WHO/CDS/HIV/18.4). Licence: CC BY-NC-SA 3.0 IGO
Platt L, Minozzi S, Reed J, et al. Needle syringe programmes and opioid substitution therapy for preventing hepatitis C transmission in people who inject drugs. Cochrane Database Syst Rev. 2017;9:CD012021.
World Health Organization. Global status report on blood safety and availability 2016. Available at: http://apps.who.int/iris/bitstream/handle/10665/254987/9789241565431-eng.pdf;jsessionid¼48C0B196CB2889E6E6D 43D0DA6A8796C?sequence¼1. Accessed 4 Dec 2018.
• Arora S, Kalishman S, Thornton K, Dion D, Murata G, Deming P, et al. Expanding access to hepatitis C virus treatment – Extension for Community Healthcare Outcomes (ECHO) project: disruptive innovation in specialty care. Hepatology. 2010;52:1124–33 This article helps us to understand the process of decentralizing public health care.
Dhiman RK, Grover GS, Premkumar M, Taneja S, Duseja A, Rathi S, et al. Direct-acting antiviral Therapy Is Safe and Effective in Pediatric Chronic Hepatitis C: the public health perspective. J Pediatr Gastroenterol Nutr. 2019;68:74–80.
Hill A, Simmons B, Gotham D, Fortunak J. Rapid reductions in prices for generic sofosbuvir and daclatasvir to treat hepatitis C. J Virus Erad. 2016;2:28–31.
Aggarwal R, Chen Q, Goel A, Seguy N, Pendse R, Ayer T, et al. Cost-effectiveness of hepatitis C treatment using generic direct-acting antivirals available in India. PLoS One. 2017. https://doi.org/10.1371/journal.pone.0176503 This study indicates the health economics of the public health approach to HCV Elimination which make it cost effective and cost saving.
WHO. WHO guideline on the use of safety-engineered syringes for intramuscular, intradermal and subcutaneous injections in health care settings: Organization WH; 2016. p. Geneva.
Harb AC, Tarabay R, Diab B, Ballout RA, Khamassi S, Akl EA. Safety engineered injection devices for intramuscular, subcutaneous and intradermal injections in healthcare delivery settings: a systematic review and meta-analysis. BMC Nurs. 2015;14(1):71.
Csete J, Kamarulzaman A, Kazatchkine M, Altice F, Balicki M, Buxton J, et al. Public health and international drug policy. Lancet. 2016;387(10026):1427–80.
Mathers BM, Degenhardt L, Ali H, Wiessing L, Hickman M, Mattick RP, et al. HIV prevention, treatment, and care services for people who inject drugs: a systematic review of global, regional, and national coverage. Lancet. 2010;375(9719):1014–28.
Grebely J, Dalgard O, Conway B, Cunningham EB, Bruggmann P, Hajarizadeh B. Sofosbuvir and velpatasvir for hepatitis C virus infection in people with recent injection drug use (SIMPLIFY): an open-label, single-arm, phase 4, multicentre trial. Lancet Gastroenterol Hepatol. 2018;3(3):153–61.
Rosenthal E, Hill K, Nussdorf L, et al. : Collocation of Buprenorphine with HCV Treatment to Improve Adherence and Reduce Harm in PWID with HCV Preliminary Data from the ANCHOR STUDY. EASL,2018. J. Hepatol. 68(1):S51. https://doi.org/10.1016/S0168-8278(18)30322-2
Terrault NA. Hepatitis C elimination: challenges with under-diagnosis and under-treatment. F1000Res. 2019;14:8. https://doi.org/10.12688/f1000research.15892.1.
World Health Organization. Guidelines on Hepatitis B and C Testing. Geneva: World Health Organization. Available at: http://apps.who.int/iris/bitstream/handle/10665/254621/9789241549981-eng.pdf;jsessionid¼47D22C07727811EA3B053EF3C997A6F3?sequence¼1. Published February 2017. Accessed 28 Dec 2018
World Health Organization. Guidelines for the care and treatment of persons diagnosed with chronic hepatitis C virus infection. Geneva: World Health Organization. Available at: http://apps.who.int/iris/bitstream/handle/10665/273174/9789241550345-eng.pdf?ua¼1. Published July 2018. Accessed 28 Dec 2018
Pedrana A, Howell J, Schröder S, Scott N, Wilson D, Kuschel C, et al. Eliminating viral hepatitis: the investment case. Doha: World Innovation Summit for Health; 2018.
da Fonseca EM, Shadlen K, Bastos FI. Brazil’s fight against hepatitis C - universalism, local production, and patents. N Engl J Med. 2019;380(7):605–7. https://doi.org/10.1056/NEJMp1812959.
Mesquita F, Santos ME, Benzaken A, Corrêa RG, Cattapan E, Sereno LS, et al. The Brazilian comprehensive response to hepatitis C: from strategic thinking to access to interferon-free therapy. BMC Public Health. 2016;16:1132.
Moon S, Erickson E. Universal medicine access through lump-sum remuneration - Australia’s approach to hepatitis C. N Engl J Med. 2019;380(7):607–10. https://doi.org/10.1056/NEJMp1813728.
Dashdorj N, Dashdorj N, Bungert AS, Genden Z, et al. Hepatitis prevention, control, and elimination program in Mongolia: national hepatitis elimination program in a country with the highest burden of viral hepatitis in the world. J Hep. 2017;66(1):S412–3. https://doi.org/10.1016/S0168-8278(17)31184-4.
Ní Cheallaigh C, O’Leary A, Keating S, Irish Hepatitis C, et al. Outcomes Research Network. Telementoring with project ECHO: a pilot study in Europe. BMJ Innov. 2017;3(3):144–51.
Becevic M, Mutrux R, Edison K. Show-Me ECHO: complex disease care capacity-building telehealth program. Stud Health Technol Inform. 2016;226:233–6.
Tahan V, Almashhrawi A, Kahveci AM, Mutrux R, Ibdah JA. Extension for community health outcomes-hepatitis C: small steps carve big footprints in the allocation of scarce resources for hepatitis C virus treatment to remote developing areas. World J Hepatol. 2016;8(11):509–12.
Chugh Y, Dhiman RK, Premkumar M, Prinja S, Grover GS, Bahuguna P. Cost-effectiveness of pan-genotypic sofosbuvir-velpatasvir combination versus genotype dependent directly acting anti-viral drugs for treatment of hepatitis C patients in the universal coverage scheme of Punjab state in India. PLoS ONE 2019 (In Publication).
Chhatwal J, He T, Hur C, Lopez-Olivo MA. Direct-acting antiviral agents for patients with hepatitis C virus genotype 1 infection are cost-saving. Clin Gastroenterol Hepatol. 2017;15:827–37.
Facente SN, Grebe E, Burk K, Morris MD, Murphy EL, Mirzazadeh A, et al. Estimated hepatitis C prevalence and key population sizes in San Francisco: a foundation for elimination. PLoS One. 2018;13:e0195575.
Janjua NZ, Islam N, Kuo M, Yu A, Wong S, Butt ZA, et al. Identifying injection drug use and estimating population size of people who inject drugs using healthcare administrative datasets. Int J Drug Policy. 2018;55:31–9.
Kapadia SN, Marks KM. Hepatitis C management simplification from test to cure: a framework for primary care providers. Clin Ther. 2018;40:1234–45.
Norton BL, Fleming J, Bachhuber MA, Steinman M, DeLuca J, Cunningham CO, et al. High HCV cure rates for people who use drugs treated with direct acting antiviral therapy at an urban primary care clinic. Int J Drug Policy. 2017;47:196–201.
van Tilborg M, Al Marzooqi SH, Wong WWL, et al. HCV core antigen as an alternative to HCV RNA testing in the era of direct-acting antivirals: retrospective screening and diagnostic cohort studies. Lancet Gastroenterol Hepatol. 2018;3:856–64. https://doi.org/10.1016/S2468-1253(18)30271-1.
Goel A, Chen Q, Chhatwal J, Aggarwal R. Cost-effectiveness of generic pan-genotypic sofosbuvir/velpatasvir versus genotype-dependent direct-acting antivirals for hepatitis C treatment. J Gastroenterol Hepatol. 2018;33:2029–36. https://doi.org/10.1111/jgh.14301.
Nwaohiri A, Schillie S, Bulterys M, Kourtis AP. Hepatitis C virus infection in children: how do we prevent it and how do we treat it? Expert Rev Anti-Infect Ther. 2018;16:689–94.
Lau G, Benhamou Y, Chen G, Li J, Shao Q, Ji D, et al. Efficacy and safety of 3-week response-guided triple direct-acting antiviral therapy for chronic hepatitis C infection: a phase 2, open-label, proof-of-concept study. Lancet Gastroenterol Hepatol. 2016;1:97–104.
Zeuzem S, Foster GR, Wang S, et al, Glecaprevir-pibrentasvir for 8 or 12 weeks in HCV genotype 1 or 3 infection. N Engl J Med. 2018;378:354–369
Government of India, World Health Organisation, Institute of Liver and Biliary Sciences. WHO GoI-ILBS national technical consultation on viral hepatitis - “towards a national action plan for viral hepatitis (NAP-VH)”. New Delhi: Institute of Liver and Biliary Sciences; 2016. 44 p
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Radha K. Dhiman declares that he has no conflict of interest. Gagandeep S. Grover declares that he has no conflict of interest. Madhumita Premkumar declares that she has no conflict of interest.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Liver
Rights and permissions
About this article
Cite this article
Dhiman, R.K., Grover, G.S. & Premkumar, M. Hepatitis C elimination: a Public Health Perspective. Curr Treat Options Gastro 17, 367–377 (2019). https://doi.org/10.1007/s11938-019-00240-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11938-019-00240-7