Abstract
Purpose of review
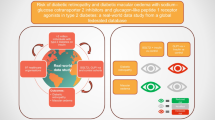
The relationship between pancreatic ductal adenocarcinoma (PDAC) and diabetes mellitus (DM) is complex. We reviewed the recent medical literature regarding the effect of anti-diabetic medication on PDAC risk and survival, risk of PDAC in DM, and role of DM in early detection of PDAC.
Recent findings
Studies report that while some anti-diabetic medications (e.g., metformin) may decrease the risk of PDAC, others (insulin, sulfonylureas and incretin-based therapies) may increase the risk. However, these observations may be subject to protopathic biases. Metformin’s anti-tumor activity may have influence overall survival of PDAC, but epidemiological reports have largely been inconsistent to defend these findings due to heterogeneous methodologies. There is congruent data to support the association between DM and PDAC, with an inverse relationship to DM duration. Older subjects with new-onset DM are the only known high-risk group for PDAC, and strategy using this group for early detection has led to development of clinical risk prediction models that define a very high-risk PDAC group.
Summary
Role of anti-diabetic medication in PDAC risk modification or survival is controversial. With successful efforts to distinguish type 2-DM from PDAC-DM using risk-stratifying models, there is an opportunity to initiate screening protocols for early detection of PDAC in a sub-set of DM subjects.

Similar content being viewed by others
Abbreviations
- ADM:
-
Anti-diabetes medication
- DM:
-
Diabetes mellitus
- NOD:
-
New-onset diabetes
- PDAC:
-
Pancreatic ductal adenocarcinoma
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Sah RP, Nagpal SJS, Mukhopadhyay D, Chari ST. New insights into pancreatic cancer-induced paraneoplastic diabetes. Nat Rev Gastroenterol Hepatol. 2013;10(7):423–33.
• Andersen DK, Korc M, Petersen GM, Eibl G, Li D, Rickels MR, et al. Diabetes, pancreatogenic diabetes, and pancreatic cancer. Diabetes. 2017;66(5):1103–10. https://doi.org/10.2337/db16-1477. This reports highlights key research gaps in relationship between PDAC and DM as identified by experts at a symposium held at the American Diabetic Association 76th scientific session.
Bosetti C, Rosato V, Buniato D, Zambon A, La Vecchia C, Corrao G. Cancer risk for patients using thiazolidinediones for type 2 diabetes: a meta-analysis. Oncologist. 2013;18(2):148–56. https://doi.org/10.1634/theoncologist.2012-0302.
Colmers IN, Bowker SL, Tjosvold LA, Johnson JA. Insulin use and cancer risk in patients with type 2 diabetes: a systematic review and meta-analysis of observational studies. Diabetes Metab. 2012;38(6):485–506. https://doi.org/10.1016/j.diabet.2012.08.011.
Bodmer M, Becker C, Meier C, Jick SS, Meier CR. Use of antidiabetic agents and the risk of pancreatic cancer: a case-control analysis. Am J Gastroenterol. 2012;107(4):620–6. https://doi.org/10.1038/ajg.2011.483.
Singh S, Singh PP, Singh AG, Murad MH, McWilliams RR, Chari ST. Anti-diabetic medications and risk of pancreatic cancer in patients with diabetes mellitus: a systematic review and meta-analysis. Am J Gastroenterol. 2013;108(4):510–9; quiz 20. https://doi.org/10.1038/ajg.2013.7.
• Chen H, Zhou X, Chen T, Liu B, Jin W, Gu H, et al. Incretin-based therapy and risk of pancreatic Cancer in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Diabetes Ther. 2016;7(4):725–42. https://doi.org/10.1007/s13300-016-0198-3. The only meta-analysis including all randomized control trials of incretin-based therapy showing no short-term increased risk of PDAC in DM patients on incretin-based therapy.
Boniol M, Franchi M, Bota M, Leclercq A, Guillaume J, van Damme N, et al. Incretin-based therapies and the short-term risk of pancreatic Cancer: results from two retrospective cohort studies. Diabetes Care. 2018;41(2):286–92. https://doi.org/10.2337/dc17-0280.
Tseng CM, Liao WC, Chang CY, Lee CT, Tseng CH, Hsu YC, et al. Incretin-based pharmacotherapy and risk of adverse pancreatic events in the ethnic Chinese with diabetes mellitus: a population-based study in Taiwan. Pancreatology. 2017;17(1):76–82. https://doi.org/10.1016/j.pan.2016.10.003.
Madiraju AK, Erion DM, Rahimi Y, Zhang XM, Braddock DT, Albright RA, et al. Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature. 2014;510(7506):542–6. https://doi.org/10.1038/nature13270.
Chen K, Qian W, Jiang Z, Cheng L, Li J, Sun L, et al. Metformin suppresses cancer initiation and progression in genetic mouse models of pancreatic cancer. Mol Cancer. 2017;16(1):131. https://doi.org/10.1186/s12943-017-0701-0.
Hong H, Yong F, Hongming P, et al. Relationship of metformin with the risk of pancreatic cancer in patients with type 2 diabetes: a meta-analysis. Biomedical Research (0970-938X) [serial online]. 2017;28(10):4439–4444. Available from: Academic Search Premier, Ipswich, MA. Accessed September 10, 2018.
Wang Z, Lai ST, Xie L, Zhao JD, Ma NY, Zhu J, et al. Metformin is associated with reduced risk of pancreatic cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetes Res Clin Pract. 2014;106(1):19–26. https://doi.org/10.1016/j.diabres.2014.04.007.
Olson SH, Xu Y, Herzog K, Saldia A, DeFilippis EM, Li P, et al. Weight loss, diabetes, fatigue, and depression preceding pancreatic cancer. Pancreas. 2016;45(7):986–91. https://doi.org/10.1097/MPA.0000000000000590.
Azar M, Lyons TJ. Diabetes, insulin treatment, and cancer risk: what is the evidence? F1000 Med Rep. 2010;2:4. doi:https://doi.org/10.3410/M2-4.
Mayer D, Shukla A, Enzmann H. Proliferative effects of insulin analogues on mammary epithelial cells. Arch Physiol Biochem. 2008;114(1):38–44. https://doi.org/10.1080/13813450801900645.
Lee DY, Yu JH, Park S, Han K, Kim NH, Yoo HJ, et al. The influence of diabetes and antidiabetic medications on the risk of pancreatic cancer: a nationwide population-based study in Korea. Sci Rep. 2018;8(1):9719. https://doi.org/10.1038/s41598-018-27965-2.
Chan MT, Lim GE, Skovso S, Yang YH, Albrecht T, Alejandro EU, et al. Effects of insulin on human pancreatic cancer progression modeled in vitro. BMC Cancer. 2014;14:814. https://doi.org/10.1186/1471-2407-14-814.
Horwitz RI, Feinstein AR. The problem of “protopathic bias” in case-control studies. Am J Med. 1980;68(2):255–8.
Drucker DJ. The biology of incretin hormones. Cell Metab. 2006;3(3):153–65. https://doi.org/10.1016/j.cmet.2006.01.004.
Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M, et al. Management of hyperglycemia in type 2 diabetes: a patient-centered approach: position statement of the American Diabetes Association (ADA) and the European Association for the Study of diabetes (EASD). Diabetes Care. 2012;35(6):1364–79. https://doi.org/10.2337/dc12-0413.
Elashoff M, Matveyenko AV, Gier B, Elashoff R, Butler PC. Pancreatitis, pancreatic, and thyroid cancer with glucagon-like peptide-1-based therapies. Gastroenterology. 2011;141(1):150–6. https://doi.org/10.1053/j.gastro.2011.02.018.
Spranger J, Gundert-Remy U, Stammschulte T. GLP-1-based therapies: the dilemma of uncertainty. Gastroenterology. 2011;141(1):20–3. https://doi.org/10.1053/j.gastro.2011.05.019.
Funch D, Gydesen H, Tornoe K, Major-Pedersen A, Chan KA. A prospective, claims-based assessment of the risk of pancreatitis and pancreatic cancer with liraglutide compared to other antidiabetic drugs. Diabetes Obes Metab. 2014;16(3):273–5. https://doi.org/10.1111/dom.12230.
Gokhale M, Buse JB, Gray CL, Pate V, Marquis MA, Sturmer T. Dipeptidyl-peptidase-4 inhibitors and pancreatic cancer: a cohort study. Diabetes Obes Metab. 2014;16(12):1247–56. https://doi.org/10.1111/dom.12379.
Butler PC, Elashoff M, Elashoff R, Gale EA. A critical analysis of the clinical use of incretin-based therapies: are the GLP-1 therapies safe? Diabetes Care. 2013;36(7):2118–25. https://doi.org/10.2337/dc12-2713.
Hart PA, Law RJ, Frank RD, Bamlet WR, Burch PA, Petersen GM, et al. Impact of diabetes mellitus on clinical outcomes in patients undergoing surgical resection for pancreatic cancer: a retrospective, cohort study. Am J Gastroenterol. 2014;109(9):1484–92. https://doi.org/10.1038/ajg.2014.193.
• Sharma A, Smyrk TC, Levy MJ, Topazian MA, Chari ST. Fasting blood glucose levels provide estimate of duration and progression of pancreatic cancer before diagnosis. Gastroenterology. 2018; https://doi.org/10.1053/j.gastro.2018.04.025. This is the first study that estiamted the pre-diagnostic duration of invasive PDAC using blood glucose as a bio-marker of early PDAC and show hyperglycemia in PDAC preceeds by 30 to 36 months prior to PDAC diagnosis and this hyperglycemia is associated with a certain tumor volume.
Zhou PT, Li B, Liu FR, Zhang MC, Wang Q, Li YY, et al. Metformin is associated with survival benefit in pancreatic cancer patients with diabetes: a systematic review and meta-analysis. Oncotarget. 2017;8(15):25242–50. https://doi.org/10.18632/oncotarget.15692.
Zhou DC, Gong H, Tan CQ, Luo JQ. Prognostic significance of anti-diabetic medications in pancreatic cancer: a meta-analysis. Oncotarget. 2017;8(37):62349–57. https://doi.org/10.18632/oncotarget.17728.
Dong YW, Shi YQ, He LW, Cui XY, Su PZ. Effects of metformin on survival outcomes of pancreatic cancer: a meta-analysis. Oncotarget. 2017;8(33):55478–88. https://doi.org/10.18632/oncotarget.18233.
Xin W, Fang L, Fang Q, Zheng X, Huang P. Effects of metformin on survival outcomes of pancreatic cancer patients with diabetes: a meta-analysis. Mol Clin Oncol. 2018;8(3):483–8. https://doi.org/10.3892/mco.2017.1541.
Frouws MA, Sibinga Mulder BG, Bastiaannet E, Zanders MM, van Herk-Sukel MP, de Leede EM, et al. No association between metformin use and survival in patients with pancreatic cancer: an observational cohort study. Medicine (Baltimore). 2017;96(10):e6229. https://doi.org/10.1097/MD.0000000000006229.
Jang WI, Kim MS, Kang SH, Jo AJ, Kim YJ, Tchoe HJ, et al. Association between metformin use and mortality in patients with type 2 diabetes mellitus and localized resectable pancreatic cancer: a nationwide population-based study in korea. Oncotarget. 2017;8(6):9587–96. https://doi.org/10.18632/oncotarget.14525.
•• Chaiteerakij R, Petersen GM, Bamlet WR, Chaffee KG, Zhen DB, Burch PA, et al. Metformin use and survival of patients with pancreatic Cancer: a cautionary lesson. J Clin Oncol. 2016;34(16):1898–904. https://doi.org/10.1200/JCO.2015.63.3511. This study highlights the importance of patient selection and appropriate statistical analysis methods used when studying medication exposure and cancer survival.
Tan J, You Y, Guo F, Xu J, Dai H, Bie P. Association of elevated risk of pancreatic cancer in diabetic patients: a systematic review and meta-analysis. Oncol Lett. 2017;13(3):1247–55. https://doi.org/10.3892/ol.2017.5586.
Makhoul I, Yacoub A, Siegel E. Type 2 diabetes mellitus is associated with increased risk of pancreatic cancer: a veteran administration registry study. SAGE Open Med. 2016;4:2050312116682257. https://doi.org/10.1177/2050312116682257.
Pannala R, Leirness JB, Bamlet WR, Basu A, Petersen GM, Chari ST. Prevalence and clinical profile of pancreatic cancer–associated diabetes mellitus. Gastroenterology. 2008;134(4):981–7.
Chari ST, Leibson CL, Rabe KG, Ransom J, De Andrade M, Petersen GM. Probability of pancreatic cancer following diabetes: a population-based study. Gastroenterology. 2005;129(2):504–11.
Muniraj T, Chari ST. Diabetes and pancreatic cancer. Minerva Gastroenterol Dietol. 2012;58(4):331–45.
•• Aggarwal G, Rabe KG, Petersen GM, Chari ST. New-onset diabetes in pancreatic cancer: a study in the primary care setting. Pancreatology. 2012;12(2):156–61. https://doi.org/10.1016/j.pan.2012.02.003. This is the first study highlighting the importance of delays in diagnosis of diabetes by physcian’s and its implications in early detection of PDAC.
Munigala S, Singh A, Gelrud A, Agarwal B. Predictors for pancreatic cancer diagnosis following new-onset diabetes mellitus. Clin Transl Gastroenterol. 2015;6:e118. https://doi.org/10.1038/ctg.2015.44.
•• Boursi B, Finkelman B, Giantonio BJ, Haynes K, Rustgi AK, Rhim AD, et al. A clinical prediction model to assess risk for pancreatic cancer among patients with new-onset diabetes. Gastroenterology. 2017;152(4):840–50.e3. This clinical risk prediction model uses physcian diagnosis of new-onset diabetes and based on various demographic, clinical and biochemical parameters at the time of NOD diagnosis predict 3-year risk of PDAC.
Pang Y, Kartsonaki C, Guo Y, Bragg F, Yang L, Bian Z, et al. Diabetes, plasma glucose and incidence of pancreatic cancer: a prospective study of 0.5 million Chinese adults and a meta-analysis of 22 cohort studies. Int J Cancer. 2017;140(8):1781–8. https://doi.org/10.1002/ijc.30599.
•• Sharma A, Kandlakunta H, Singh Nagpal SJ, Ziding F, Hoos W, Petersen GM, et al. Model to determine risk of pancreatic cancer in patients with new-onset diabetes. Gastroenterology. 2018; https://doi.org/10.1053/j.gastro.2018.05.023. This is the first clinical risk prediction model that uses glycemic definition of new-onset DM and identifies a very high risk group for PDAC (~4% 3-year risk) that may benefit from clinical work-up.
Harris MI, Klein R, Welborn TA, Knuiman MW. Onset of NIDDM occurs at least 4-7 yr before clinical diagnosis. Diabetes Care. 1992;15(7):815–9.
Fraser LA, Twombly J, Zhu M, Long Q, Hanfelt JJ, Narayan KM, et al. Delay in diagnosis of diabetes is not the patient’s fault. Diabetes Care. 2010;33(1):e10. https://doi.org/10.2337/dc09-1129.
Menke A, Casagrande S, Geiss L, Cowie CC. Prevalence of and trends in diabetes among adults in the United States, 1988-2012. JAMA. 2015;314(10):1021–9. https://doi.org/10.1001/jama.2015.10029.
Chari ST. Detecting early pancreatic cancer: problems and prospects. Semin Oncol. 2007;34(4):284–94. https://doi.org/10.1053/j.seminoncol.2007.05.005.
CenterforDiseaseControl. https://gis.cdc.gov/grasp/diabetes/diabetesatlas.html accessed on January 17, 2018. https://gis.cdc.gov/grasp/diabetes/diabetesatlas.html.
Ortman JM, Velkoff VA, Hogan H. An aging nation: the older population in the United States, Current Population Reports, P25–1140. Washington, D.C.: U.S. Census Bureau. 2014. https://www.census.gov/content/dam/Census/library/publications/2014/demo/p25-1140.pdf. Accessed July 7, 2018.
Choe JW, Kim HJ, Kim JS, Cha J, Joo MK, Lee BJ, et al. Usefulness of CA 19-9 for pancreatic cancer screening in patients with new-onset diabetes. Hepatobiliary Pancreat Dis Int. 2018;17(3):263–8. https://doi.org/10.1016/j.hbpd.2018.04.001.
Murakami M, Nagai Y, Tenjin A, Tanaka Y. Proposed cut-off value of CA19-9 for detecting pancreatic cancer in patients with diabetes: a case-control study. Endocr J. 2018;65(6):639–43. https://doi.org/10.1507/endocrj.EJ17-0380.
O'Brien DP, Sandanayake NS, Jenkinson C, Gentry-Maharaj A, Apostolidou S, Fourkala EO, et al. Serum CA19-9 is significantly upregulated up to 2 years before diagnosis with pancreatic cancer: implications for early disease detection. Clin Cancer Res. 2015;21(3):622–31. https://doi.org/10.1158/1078-0432.CCR-14-0365.
Balasenthil S, Huang Y, Liu S, Marsh T, Chen J, Stass SA, et al. A plasma biomarker panel to identify surgically resectable early-stage pancreatic cancer. J Natl Cancer Inst. 2017;109(8) https://doi.org/10.1093/jnci/djw341.
Pelaez-Luna M, Takahashi N, Fletcher JG, Chari ST. Resectability of presymptomatic pancreatic cancer and its relationship to onset of diabetes: a retrospective review of CT scans and fasting glucose values prior to diagnosis. Am J Gastroenterol. 2007;102(10):2157–63. https://doi.org/10.1111/j.1572-0241.2007.01480.x.
Gangi S, Fletcher JG, Nathan MA, Christensen JA, Harmsen WS, Crownhart BS, et al. Time interval between abnormalities seen on CT and the clinical diagnosis of pancreatic cancer: retrospective review of CT scans obtained before diagnosis. AJR Am J Roentgenol. 2004;182(4):897–903. https://doi.org/10.2214/ajr.182.4.1820897.
• Bruenderman E, Martin RC II. A cost analysis of a pancreatic cancer screening protocol in high-risk populations. Am J Surg. 2015;210(3):409–16. https://doi.org/10.1016/j.amjsurg.2014.11.017. This is the first study that evaluates the cost effectivness of screening with imaging in patients over the age of 50 years for PDAC.
National Lung Screening Trial Research T, Church TR, Black WC, Aberle DR, Berg CD, Clingan KL, et al. Results of initial low-dose computed tomographic screening for lung cancer. N Engl J Med. 2013;368(21):1980–91. https://doi.org/10.1056/NEJMoa1209120.
Kenner BJ, Chari ST, Maitra A, Srivastava S, Cleeter DF, Go VL, et al. Early detection of pancreatic cancer—a defined future using lessons from other cancers: a white paper. Pancreas. 2016;45(8):1073–9. https://doi.org/10.1097/MPA.0000000000000701.
Rex DK. Colonoscopy: a review of its yield for cancers and adenomas by indication. Am J Gastroenterol. 1995;90(3):353–65.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Suresh Chari reports grants from NIH and the Kenner Family Research foundation, during the conduct of the study.
Ayush Sharma declares no conflict of interest.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any.
of the authors.
Additional information
This article is part of the Topical Collection on Pancreas
Rights and permissions
About this article
Cite this article
Sharma, A., Chari, S.T. Pancreatic Cancer and Diabetes Mellitus. Curr Treat Options Gastro 16, 466–478 (2018). https://doi.org/10.1007/s11938-018-0197-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11938-018-0197-8