Opinion statement
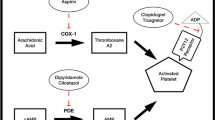
In patients with transient ischemic attack (TIA) or ischemic stroke of noncardiac origin, antiplatelet drugs are able to decrease the risk of stroke by 11% to 15%, and decrease the risk of stroke, myocardial infarction (MI), and vascular death by 15% to 22%. Aspirin leads to a moderate but significant reduction of stroke, MI, and vascular death in patients with TIA and ischemic stroke. Low doses are as effective as high doses, but are better tolerated in terms of gastrointestinal side effects. The recommended aspirin dose, therefore, is between 50 and 325 mg. Bleeding complications are not dose-dependent, and also occur with the lowest doses. The combination of aspirin (25 mg twice daily) with slow-release dipyridamole (200 mg twice daily) is superior compared with aspirin alone for stroke prevention. Ticlopidine is effective in secondary stroke prevention in patients with TIA and stroke. For some end points, it is superior to aspirin. Due to its side-effect profile (neutropenia, thrombotic thrombocytopenic purpura [TTP]), ticlopidine should be given to patients who are intolerant of aspirin. Prospective trials have not indicated whether ticlopidine is suggested for patients who have recurrent cerebrovascular events while on aspirin. Clopidogrel has a better safety profile than ticlopidine. Although not investigated in patients with TIA, clopidogrel should also be effective in these patients assuming the same pathophysiology than in patients with stroke. Clopidogrel is second-line treatment in patients intolerant for aspirin, and first-line treatment for patients with stroke and peripheral arterial disease or MI. A frequent clinical problem is patients who are already on aspirin because of coronary heart disease or a prior cerebral ischemic event, and then suffer a first or recurrent TIA or stroke. No single clinical trial has investigated this problem. Therefore, recommendations are not evidence-based. Possible strategies include the following: continue aspirin, add dipyridamole, add clopidogrel, switch to ticlopidine or clopidogrel, or switch to anticoagulation with an International Normalized Ratio (INR) of 2.0 to 3.0. The combination of low-dose warfarin and aspirin was never studied in the secondary prevention of stroke. In patients with a cardiac source of embolism, anticoagulation is recommended with an INR of 2.0 to 3.0. At the present time, anticoagulation with an INR between 3.0 and 4.5 cannot be recommended for patients with noncardiac TIA or stroke. Anticoagulation with an INR between 3.0 and 4.5 carries a high bleeding risk. Whether anticoagulation with lower INR is safe and effective is not yet known. Treatment of vascular risk factors should also be performed in secondary stroke prevention.
Similar content being viewed by others
References and Recommended Reading
Dennis M, Bamford J, Sandercock P, et al.: The prognosis of transient ischemic attacks in the community: the Oxfordshire Community Stroke project. Stroke 1991, 21:848–853.
Hankey GJ, Warlow CP, Slattery J: The prognosis of transient ischemic attacks. J Neurol 1990, 237:142.
Wilterdink JL, Easton JD: Vascular event rates in patients with atherosclerotic cerebrovascular disease. Arch Neurol 1992, 49:857–863.
The Amaurosis Fugax Study Group: Current management of amaurosis fugax. Stroke 1990, 21:201–208.
Gueyffier R, Boissel JP, Boutitie F, et al.: Effect of antihypertensive treatment in patients having already suffered from stroke: gathering the evidence. Stroke 1997, 28:2557–2562.
Randomised trial of a perindopril-based bloodpressure-lowering regimen among 6105 individuals with previous stroke or transient ischaemic attack. Progress Collaborative Group [no authors listed]. Lancet 2001,358:1033–1041.
The Scandinavian Simvastatin Survival Study Group: Randomized trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet 1994, 344:1383–1389.
Blauw GJ, Lagaay AM, Smelt AHM, et al.: Stroke, statins, and cholesterol: a meta-analysis of randomized, placebo-controlled, double-blind trials with HMGCoA reductase inhibitors. Stroke 1997, 28:946–950.
Plehn JF, Davis BR, Sacks FM, et al.: Reduction of stroke incidence after myocardial infarction with pravastatin: the Cholesterol and Recurrent Events (CARE) study. Circulation 1999, 99:216–223.
Rosendorff C: Statins for prevention of stroke. Lancet 1998, 351:1002–1003.
The Long-Term Intervention with Pravastatin in Ischemic Disease (LIPID) Study Group: Prevention of cardiovascular events and death with pravastatin in patients with coronary heart disease and a broad range of initial cholesterol levels. N Engl J Med 1998, 339:1349–1357.
White HD, Simes RJ, Anderson NE, et al.: Pravastatin therapy and the risk of stroke. N Engl J Med 2000, 343:317–326.
Shepherd J, Blauw GJ, Murphy MB, et al.: The design of a prospective study of pravastatin in the elderly at risk (PROSPER). Am J Cardiol 1999, 84:1192–1197.
Rubins HB, Robins SJ, Collins D, et al.: Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. N Engl J Med 1999, 341:410–418.
Diabetes Control and Complications Trial Research Group: The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993, 329:977–986.
Antiplatelet Trialists Collaboration: Secondary prevention of vascular disease by prolonged antiplatelet treatment. BMJ 1988, 296:320–331.
Patrono C, Roth GJ: Aspirin in ischemic cerebro-vascular disease. Stroke 1996, 27:756–760.
American-Canadian Co-operative Study Group: Persantin-aspirin in cerebral ischemia, part II: endpoint results. Stroke 1985, 16:406–415.
Antiplatelet Trialists Collaboration: Collaborative overview of randomised trials of antiplatelet therapy-I: prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients. BMJ 1994, 308:81–106.
Hart RG, Halperin JL: Atrial fibrillation and stroke: revisiting the dilemmas. Stroke 1994, 25:1337–1341.
North American Symptomatic Carotid Endarterectomy Trial Collaborators: Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med 1991, 325:445–453.
European Carotid Surgery Trialists Collaborative Group: Randomized trial of endarterectomy for recently symptomatic carotid stenosis: final results of the MRC European Carotid Surgery Trial (ECST). Lancet 1998, 351:1379–1387.
The ESPS Group: The European Stroke Prevention Study (ESPS). Principal end-points. Lancet 1987, 2:1351–1354.
Diener HC, Forbes C, Riekkinen PJ, et al.: European Stroke Prevention Study 2: efficacy and safety data. J Neurol Sci 1997, 151:S1-S77.
Gent M, Blakely JA, Easton JD, et al.: The Canadian American ticlopidine study (CATS) in thromboembolic stroke. Lancet 1989, 1:1215–1220.
International Stroke Trial Collaborative Group: The International Stroke Trial (IST): a randomized trial of aspirin, subcutaneous heparin, both, or neither among 19,435 patients with acute ischemic stroke. Lancet 1997, 349:1564–1565.
CAST (Chinese Acute Stroke Trial) Collaborative Group: CAST: randomized placebo-controlled trial of early aspirin use in 20,000 patients with acute ischemic stroke. Lancet 1997, 349:1641–1649.
UK-TIA Study Group: The United Kingdom transient ischemic attack (UK-TIA) aspirin trial: final results. J Neurol Neurosurg Psychiatry 1991, 54:1044–1054.
The SALT Collaborative Group: Swedish aspirin low-dose trial (SALT) of 75-mg aspirin as secondary prophylaxis after cerebrovascular ischaemic events. Lancet 1991, 338:1345–1349.
ESPS-Working Group: Second European stroke prevention study. J Neurol 1992, 239:299–301.
Diener HC, Cuhna L, Forbes C, et al.: European Stroke Prevention Study 2: dipyridamole and acetylsalicylic acid in the secondary prevention of stroke. J Neurol Sci 1996, 143:1–13.
Hass WK, Easton JD, Adams HP, et al.: A randomized trial comparing ticlopidine hydrochloride with aspirin for the prevention of stroke. N Engl J Med 1989, 321:501–507.
CAPRIE Steering Committee: A randomized, blinded, trial of clopidogrel versus aspirin in patients at risk of ischemic events (CAPRIE). Lancet 1996, 348:1329–1339.
Dyken ML, Barnett HJM, Easton JD, et al.: Low-dose aspirin and stroke: “ It ain’t necessarily so”. Stroke 1992, 23:1395–1399.
Dyken ML: Controversies in stroke: past and present. The Willis lecture. Stroke 1993, 24:1251–1258.
Diener HC: Use of acetylsalicylic acid in the secondary prevention of stroke. Int J Clin Pract 1998, Suppl 97:12–15.
Algra A, van GijnJ: Aspirin at any dose above 30 mg offers only modest protection after cerebral ischemia. J Neurol Neurosurg Psychiatry 1996, 60:197–199.
Algra A, van Gijn J: Cumulative meta-analysis of aspirin efficacy after cerebral ischemia of arterial origin. J Neurol Neurosurg Psychiatry 1999, 65:255. A good review.
Department of Health and Human Services, US Food and Drug Administration: Internal analgesic, antipyretic, and anti-rheumatic drug products for overthe counter human use. Final rule for professional labeling of aspirin, buffered aspirin, and aspirin in combination with antacid drug products. Federal Register 1998, 63:56802–56819.
The Dutch TIA Trial Study Group: A comparison of two doses of aspirin (30 mg vs 283 mg/day) in patients after a transient ischemic attack or minor ischemic stroke. N Engl J Med 1991, 325:1261–1266.
Page Y, Tardy B, Zeni F, et al.: Thrombotic thrombocytopenic purpura related to ticlopidine. Lancet 1991, 337:774–776.
Bennett CL, Kiss JE, Weinberg PD, et al.: Thrombotic thrombocytopenic purpura after stenting and ticlopidine. Lancet 1998, 352:1036–1037.
Bennett CL, Connors JM, Carwile JM, et al.: Thrombotic thrombocytopenic purpura associated with clopidogrel. N Engl J Med 2000, 342:1773–1777. An excellent article.
Patrono C, Coller B, Dalen JE, et al.: Platelet-active drugs: the relationships among dose, effectiveness, and side effects. Chest 1998, 114(suppl):470S-488S.
Guiraud-Chaumeil B, Rascol A, David J, et al.: Prevention des recidives des accidents vasculaires cerebraux ischemiques par les anti-agregants plaquettaires. Rev Neurol (Paris) 1982, 138:367–385.
Bousser MG, Eschwege E, Haguenau M, et al.: " A.I.C.L.A." controlled trial of aspirin and dipyridamole in the secondary prevention of atherothrombotic cerebral ischemia. Stroke 1983, 13:5–14.
Müller TH, Su CA, Weisenberger H, et al.: Dipyridamole alone or combined with low dose acetylsalicylic acid inhibits platelet aggregation in human whole blood ex vivo. Br J Clin Pharmacol 1990, 30:179–186.
EAFT Group: Secondary prevention in non-rheumatic atrial fibrillation after transient ischaemic attack or minor stroke. Lancet 1993, 342:1255–1262.
The Stroke Prevention in Reversible Ischemia Trial (SPIRIT) Study Group: A randomized trial of anticoagulants versus aspirin after cerebral ischemia of presumed arterial origin. Ann Neurol 1997, 42:857–865.
Mohr JP, Thompson JL, Lazar RM, et al.: A comparison of warfarin and aspirin for the prevention of recurrent ischemic stroke. N Engl J Med 2001, 345:1444–1451.
Chimowitz M, Kokkinos J, Strong J, et al.: The warfarin-aspirin symptomatic intracranial disease study. Neurology 1995, 45:1488–1493.
Topol EJ, Byzova TV, Plow EF: Platelet GPIIb-IIIa blockers. Lancet 1999, 353:227–231.
Topol EJ, Eston JD, Amarenco P, et al.: Design of the blockade of the glycoprotein IIb/IIIa receptor to avoid vascular occlusion (BRAVO) trial. Am Heart J 2000, 139:927–933.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Diener, HC., Ringleb, P. Antithrombotic secondary prevention after stroke. Curr Treat Options Cardio Med 4, 429–440 (2002). https://doi.org/10.1007/s11936-002-0022-4
Issue Date:
DOI: https://doi.org/10.1007/s11936-002-0022-4