Abstract
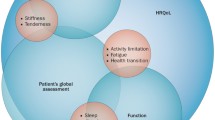
Patient reported outcomes (PRO) are at the core of assessing RA treatment response with patient assessments of global health or disease activity, pain, and physical function included in the calculation of American College of Rheumatology (ACR) responses. Progress has been made in assessing PROs that include additional patient-valued aspects of disease in recent RA randomized clinical trials (RCTs), particularly fatigue. Importantly, the National Institute of Health (NIH)—Patient Reported Outcomes Measurement Information System (PROMIS) development of psychometrically advanced generic health measures that span the range of symptoms potentially affected in RA, with high precision across the entire range of a symptom are undergoing additional study in RA and other rheumatologic diseases to establish their construct validity, responsiveness, and clinically meaningful cutoffs. PRO measures that are currently used and widely available can provide important perspectives not captured in composite clinical response criteria with the potential of better informing treatment decisions in clinical practice.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Boers M, Kirwan JR, Wells G, et al. Developing core outcome measurement sets for clinical trials: OMERACT filter 2.0. J Clin Epidemiol. 2014;67:745–53. The newly endorsed OMERACT Filter 2.0 provides a framework and methodology for selecting RCT core sets.
Tugwell PS, Petersson IF, Boers M, et al. Domains selection for patient-reported outcomes: current activities and options for future methods. J Rheumatol. 2011;38:1702–10.
Frank L, Forsythe L, Ellis L, Schrandt S, Sheridan S, Gerson J, et al. Conceptual and practical foundations of patient engagement in research at the patient-centered outcomes research institute. Quality of life research: an international journal of quality of life aspects of treatment, care and rehabilitation. 2015 Jan 6.
Frank L, Basch E, Selby JV. The PCORI perspective on patient-centered outcomes research. JAMA. 2014;312(15):1513–4.
Boers M, Tugwell P, Felson DT, et al. World Health Organization and International League of Associations for Rheumatology core endpoints for symptom modifying antirheumatic drugs in rheumatoid arthritis clinical trials. J Rheumatol Suppl. 1994;41:86–9.
Felson DT, Anderson JJ, Boers M, et al. The American college of rheumatology preliminary core set of disease activity measures for rheumatoid arthritis clinical trials. The committee on outcome measures in rheumatoid arthritis clinical trials. Arthritis Rheum. 1993;36:729–40.
Kirkham JJ, Boers M, Tugwell P, et al. Outcome measures in rheumatoid arthritis randomised trials over the last 50 years. Trials. 2013;14:324.
Boers M, Brooks P, Strand CV, et al. The OMERACT filter for Outcome Measures in Rheumatology. J Rheumatol. 1998;25:198–9.
van Tuyl LH, Hewlett S, Sadlonova M, Davis B, Flurey C, Hoogland W, et al. The patient perspective on remission in rheumatoid arthritis: ‘You’ve got limits, but you’re back to being you again’. Annals of the rheumatic diseases. 2014 Feb 12.
Bingham 3rd CO, Alten R, Bartlett SJ, et al. Identifying preliminary domains to detect and measure rheumatoid arthritis flares: report of the OMERACT 10 RA Flare Workshop. J Rheumatol. 2011;38:1751–8.
Bingham 3rd CO, Alten R, de Wit MP. The importance of patient participation in measuring rheumatoid arthritis flares. Ann Rheum Dis. 2012;71:1107–9.
Bartlett SJ, Hewlett S, Bingham 3rd CO, et al. Identifying core domains to assess flare in rheumatoid arthritis: an OMERACT international patient and provider combined Delphi consensus. Ann Rheum Dis. 2012;71:1855–60.
de Wit M, Abma T, Koelewijn-van Loon M, et al. Involving patient research partners has a significant impact on outcomes research: a responsive evaluation of the international OMERACT conferences. BMJ Open. 2013;3(5).
Cella D, Yount S, Rothrock N, et al. The Patient-Reported Outcomes Measurement Information System (PROMIS): progress of an NIH Roadmap cooperative group during its first two years. Med Care. 2007;45(5 Suppl 1):S3–11.
WHO. WHO: The International Classification of Functioning, Disability and Health 2002. http://www.who.int/classifications/icf/training/icfbeginnersguide.pdf?ua=1; 2001 [updated 2001; cited]; Available from: http://www.who.int/classifications/icf/training/icfbeginnersguide.pdf?ua=1.
US Food and Drug Administration. US Food and Drug Administration. Guidance for Industry Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims (2009). 2009 [updated 2009 2009; cited 2014 1/23/2014]; Available from: www.fda.gov/downloads/Drugs/Guidances/UCM193282.pdf.
Frost MH, Reeve BB, Liepa AM, et al. What is sufficient evidence for the reliability and validity of patient-reported outcome measures? Value Health. 2007;10 Suppl 2:S94–105.
Coons SJ, Kothari S, Monz BU, et al. The patient-reported outcome (PRO) consortium: filling measurement gaps for PRO end points to support labeling claims. Clin Pharmacol Ther. 2011;90:743–8.
Mokkink LB, Terwee CB, Knol DL, et al. The COSMIN checklist for evaluating the methodological quality of studies on measurement properties: a clarification of its content. BMC Med Res Methodol. 2010;10:22.
Mokkink LB, Terwee CB, Patrick DL, et al. The COSMIN checklist for assessing the methodological quality of studies on measurement properties of health status measurement instruments: an international Delphi study. Qual Life Res: Int J Qual Life Asp Treat Care Rehabil. 2010;19:539–49.
Patrick DL, Burke LB, Gwaltney CJ, et al. Content validity–establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO good research practices task force report: part 1–eliciting concepts for a new PRO instrument. Value Health: J Int Soc Pharmacoeconomics Outcomes Res. 2011;14:967–77.
Patrick DL, Burke LB, Gwaltney CJ, et al. Content validity—establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO Good Research Practices Task Force report: part 2—assessing respondent understanding. Value Health: J Int Soc Pharmacoeconomics Outcomes Res. 2011;14:978–88.
Reeve BB, Wyrwich KW, Wu AW, et al. ISOQOL recommends minimum standards for patient-reported outcome measures used in patient-centered outcomes and comparative effectiveness research. Qual Life Res: Int J Qual Life Asp Treat Care Rehabil. 2013;22:1889–905.
Brundage M, Blazeby J, Revicki D, et al. Patient-reported outcomes in randomized clinical trials: development of ISOQOL reporting standards. Qual Life Res: Int J Qual LIfe Asp Treat Care Rehabil. 2013;22:1161–75.
Wyrwich KW, Norquist JM, Lenderking WR, et al. Methods for interpreting change over time in patient-reported outcome measures. Qual Life Res: Int J Qual Life Asp Treat Care Rehabil. 2013;22:475–83.
Snyder CF, Aaronson NK, Choucair AK, et al. Implementing patient-reported outcomes assessment in clinical practice: a review of the options and considerations. Qual Life Res: Int J Qual Life Asp Treat Care Rehabil. 2012;21:1305–14.
de Wit MP, Berlo SE, Aanerud GJ, et al. European league against rheumatism recommendations for the inclusion of patient representatives in scientific projects. Ann Rheum Dis. 2011;70:722–6.
Gossec L, Paternotte S, Aanerud GJ, et al. Finalisation and validation of the rheumatoid arthritis impact of disease score, a patient-derived composite measure of impact of rheumatoid arthritis: a EULAR initiative. Ann Rheum Dis. 2011;70:935–42.
Gossec L, de Wit M, Kiltz U, et al. A patient-derived and patient-reported outcome measure for assessing psoriatic arthritis: elaboration and preliminary validation of the Psoriatic Arthritis Impact of Disease (PsAID) questionnaire, a 13-country EULAR initiative. Ann Rheum Dis. 2014;73:1012–9.
Castrejon I, Gossec L, Carmona L. The EULAR outcome measures library: an evolutional database of validated patient-reported instruments. Ann Rheum Dis. 2015;74:475–6. This article describes the EULAR electronic resource of patient reported outcome measures.
Methodology Committee of the Patient-Centered Outcomes Research I. Methodological standards and patient-centeredness in comparative effectiveness research: the PCORI perspective. JAMA. 2012;307:1636–40.
Her M, Kavanaugh A. Patient-reported outcomes in rheumatoid arthritis. Curr Opin Rheumatol. 2012;24:327–34.
Fleischmann R, Kremer J, Cush J, et al. Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N Engl J Med. 2012;9(367):495–507.
van Vollenhoven RF, Fleischmann R, Cohen S, et al. Tofacitinib or adalimumab versus placebo in rheumatoid arthritis. N Engl J Med. 2012;9(367):508–19.
Burmester GR, Blanco R, Charles-Schoeman C, et al. Tofacitinib (CP-690,550) in combination with methotrexate in patients with active rheumatoid arthritis with an inadequate response to tumour necrosis factor inhibitors: a randomised phase 3 trial. Lancet. 2013;381:451–60.
Lee EB, Fleischmann R, Hall S, et al. Tofacitinib versus methotrexate in rheumatoid arthritis. N Engl J Med. 2014;370:2377–86.
Weinblatt ME, Schiff M, Valente R, et al. Head-to-head comparison of subcutaneous abatacept versus adalimumab for rheumatoid arthritis: findings of a phase IIIb, multinational, prospective, randomized study. Arthritis Rheum. 2013;65:28–38.
Schiff M, Weinblatt ME, Valente R, et al. Head-to-head comparison of subcutaneous abatacept versus adalimumab for rheumatoid arthritis: two-year efficacy and safety findings from AMPLE trial. Ann Rheum Dis. 2014;73:86–94.
Gabay C, Emery P, van Vollenhoven R, et al. Tocilizumab monotherapy versus adalimumab monotherapy for treatment of rheumatoid arthritis (ADACTA): a randomised, double-blind, controlled phase 4 trial. Lancet. 2013;381:1541–50.
Kavanaugh A, Fleischmann RM, Emery P, et al. Clinical, functional and radiographic consequences of achieving stable low disease activity and remission with adalimumab plus methotrexate or methotrexate alone in early rheumatoid arthritis: 26-week results from the randomised, controlled OPTIMA study. Ann Rheum Dis. 2013;72:64–71.
Smolen JS, Emery P, Fleischmann R, et al. Adjustment of therapy in rheumatoid arthritis on the basis of achievement of stable low disease activity with adalimumab plus methotrexate or methotrexate alone: the randomised controlled OPTIMA trial. Lancet. 2014;383:321–32.
Bingham 3rd CO, Pohl C, Woodworth TG, et al. Developing a standardized definition for disease “flare” in rheumatoid arthritis (OMERACT 9 Special Interest Group). J Rheumatol. 2009;36:2335–41.
Hewlett S, Sanderson T, May J, et al. ‘I’m hurting, I want to kill myself’: rheumatoid arthritis flare is more than a high joint count—an international patient perspective on flare where medical help is sought. Rheumatology (Oxford, England). 2012;51:69–76.
Bykerk VP, Lie E, Bartlett SJ, Alten R, Boonen A, Christensen R, et al. Establishing a core domain set to measure rheumatoid arthritis flares: report of the OMERACT 11 RA flare Workshop. J Rheumatol. 2014;41:799–809.
Kirwan JR, Minnock P, Adebajo A, et al. Patient perspective: fatigue as a recommended patient centered outcome measure in rheumatoid arthritis. J Rheumatol. 2007;34:1174–7.
Felson DT, Anderson JJ, Boers M, et al. American College of Rheumatology. Preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum. 1995;38:727–35.
Felson DT, Smolen JS, Wells G, et al. American College of Rheumatology/European League Against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Arthritis Rheum. 2011;63:573–86.
Fries JF, Spitz P, Kraines RG, et al. Measurement of patient outcome in arthritis. Arthritis Rheum. 1980;23:137–45.
Pincus T, Summey JA, Soraci SA, et al. Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis Rheum. 1983;26:1346–53.
Keystone EC, Genovese MC, Hall S, et al. Golimumab in patients with active rheumatoid arthritis despite methotrexate therapy: results through 2 years of the GO-FORWARD study extension. J Rheumatol. 2013;40:1097–103.
de Jong PH, Hazes JM, Han HK, et al. Randomised comparison of initial triple DMARD therapy with methotrexate monotherapy in combination with low-dose glucocorticoid bridging therapy; 1-year data of the tREACH trial. Ann Rheum Dis. 2014;73:1331–9.
Fries JF, Cella D, Rose M, et al. Progress in assessing physical function in arthritis: PROMIS short forms and computerized adaptive testing. J Rheumatol. 2009;36:2061–6.
Sokka T. Long-term outcomes of rheumatoid arthritis. Curr Opin Rheumatol. 2009;21:284–90.
Ward MM, Guthrie LC, Alba MI. Clinically important changes in individual and composite measures of rheumatoid arthritis activity: thresholds applicable in clinical trials. Ann Rheum Dis. 2014 May 1. This article redefines the minimally clinically important improvement in RA for the health assessment questionnaire at higher levels.
Ward MM, Guthrie LC, Alba MI. Clinically important changes in individual and composite measures of rheumatoid arthritis activity: thresholds applicable in clinical trials. Annals of the rheumatic diseases. 2014May 1.
Hays RD, Spritzer KL, Fries JF, et al. Responsiveness and minimally important difference for the Patient-Reported Outcomes Measurement Information System (PROMIS) 20-item physical functioning short form in a prospective observational study of rheumatoid arthritis. Ann Rheum Dis. 2015;74:104–7. This article defines the minimally clinically important difference in RA for the PROMIS 20 item physical functioning form.
Oude Voshaar MA, Ten Klooster PM, Glas CA, et al. Relative performance of commonly used physical function questionnaires in rheumatoid arthritis and a patient-reported outcomes measurement information system computerized adaptive test. Arthritis Care Res (Hoboken, NJ). 2014;66:2900–8.
Bartlett SJ, Orbai AM, Duncan T, et al. How well do generic patient reported outcomes measurement information system instruments capture health status in rheumatoid arthritis? Arthritis Rheum. 2013;65(10 Suppl):972.
Broderick JE, Schneider S, Junghaenel DU, et al. Validity and reliability of patient-reported outcomes measurement information system instruments in osteoarthritis. Arthritis Care Res. 2013;65(10):1625–33.
Khanna D, Maranian P, Rothrock N, et al. Feasibility and construct validity of PROMIS and “legacy” instruments in an academic scleroderma clinic. Value Health: J Int Soc Pharmacoeconomics Outcome Res. 2012;15:128–34.
Pincus T, Bergman MJ, Yazici Y, et al. An index of only patient-reported outcome measures, routine assessment of patient index data 3 (RAPID3), in two abatacept clinical trials: similar results to disease activity score (DAS28) and other RAPID indices that include physician-reported measures. Rheumatology (Oxford). 2008;47:345–9.
Pincus T, Swearingen C, Wolfe F. Toward a multidimensional Health Assessment Questionnaire (MDHAQ): assessment of advanced activities of daily living and psychological status in the patient-friendly health assessment questionnaire format. Arthritis Rheum. 1999;42:2220–30.
Pincus T, Yazici Y, Castrejon I. Pragmatic and scientific advantages of MDHAQ/ RAPID3 completion by all patients at all visits in routine clinical care. Bull NYU Hosp Joint Dis. 2012;70 Suppl 1:30–6.
Pincus T, Furer V, Keystone E, et al. RAPID3 (Routine Assessment of Patient Index Data 3) severity categories and response criteria: similar results to DAS28 (Disease Activity Score) and CDAI (Clinical Disease Activity Index) in the RAPID 1 (Rheumatoid Arthritis Prevention of Structural Damage) clinical trial of certolizumab pegol. Arthritis Care Res. 2011;63:1142–9.
Pincus T, Swearingen CJ, Bergman MJ, et al. RAPID3 (Routine Assessment of Patient Index Data) on an MDHAQ (Multidimensional Health Assessment Questionnaire): agreement with DAS28 (Disease Activity Score) and CDAI (Clinical Disease Activity Index) activity categories, scored in five versus more than ninety seconds. Arthritis Care Res. 2010;62:181–9.
Yazici Y, Curtis JR, Ince A, et al. Efficacy of tocilizumab in patients with moderate to severe active rheumatoid arthritis and a previous inadequate response to disease-modifying antirheumatic drugs: the ROSE study. Ann Rheum Dis. 2012;71(2):198–205.
Gossec L, Dougados M, Rincheval N, et al. Elaboration of the preliminary Rheumatoid Arthritis Impact of Disease (RAID) score: a EULAR initiative. Ann Rheum Dis. 2009;68:1680–5.
Boers M. RAID: a valid tool to quantify the impact of rheumatoid arthritis. But what impact will it have on the core set for trials? Ann Rheum Dis. 2011;70:884–5.
Heiberg T, Austad C, Kvien TK, et al. Performance of the Rheumatoid Arthritis Impact of Disease (RAID) score in relation to other patient-reported outcomes in a register of patients with rheumatoid arthritis. Ann Rheum Dis. 2011;70:1080–2.
Dougados M, Brault Y, Logeart I, et al. Defining cut-off values for disease activity states and improvement scores for patient-reported outcomes: the example of the Rheumatoid Arthritis Impact of Disease (RAID). Arthritis Res Ther. 2012;14:R129. This article defines the minimally clinically important difference for the RAID questionnaire.
Ware Jr JE, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473–83.
Kosinski M, Zhao SZ, Dedhiya S, et al. Determining minimally important changes in generic and disease-specific health-related quality of life questionnaires in clinical trials of rheumatoid arthritis. Arthritis Rheum. 2000;43:1478–87.
Busija L, Pausenberger E, Haines TP, et al. Adult measures of general health and health-related quality of life: Medical Outcomes Study Short Form 36-Item (SF-36) and Short Form 12-Item (SF-12) Health Surveys, Nottingham Health Profile (NHP), Sickness Impact Profile (SIP), Medical Outcomes Study Short Form 6D (SF-6D), Health Utilities Index Mark 3 (HUI3), Quality of Well-Being Scale (QWB), and Assessment of Quality of Life (AQoL). Arthritis Care Res. 2011;63 Suppl 11:S383–412.
Hays RD, Morales LS. The RAND-36 measure of health-related quality of life. Ann Med. 2001;33:350–7.
Ware Jr J, Kosinski M, Keller SD. A 12-item short-form health survey: construction of scales and preliminary tests of reliability and validity. Med Care. 1996;34:220–33.
Gandek B, Ware JE, Aaronson NK, et al. Cross-validation of item selection and scoring for the SF-12 Health Survey in nine countries: results from the IQOLA Project. International Quality of Life Assessment. J Clin Epidemiol. 1998;51:1171–8.
Hays RD, Liu H, Spritzer K, et al. Item response theory analyses of physical functioning items in the medical outcomes study. Med Care. 2007;45(5 Suppl 1):S32–8.
Whalley D, McKenna SP, de Jong Z, et al. Quality of life in rheumatoid arthritis. Br J Rheumatol. 1997;36:884–8.
Kirwan JR, Hewlett S. Patient perspective: reasons and methods for measuring fatigue in rheumatoid arthritis. J Rheumatol. 2007;34:1171–3.
Sanderson T, Calnan M, Morris M, et al. Shifting normalities: interactions of changing conceptions of a normal life and the normalisation of symptoms in rheumatoid arthritis. Sociol Health Illn. 2011;33:618–33.
Bartlett SJ, Orbai AM, Duncan T, et al. Can generic PROMIS measures adequately capture fatigue in rheumatoid arthritis? Ann Rheum Dis. 2014;73 Suppl 2:1167.
Yellen SB, Cella DF, Webster K, et al. Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. J Pain Symptom Manag. 1997;13:63–74.
Cella D, Yount S, Sorensen M, et al. Validation of the functional assessment of chronic illness therapy fatigue scale relative to other instrumentation in patients with rheumatoid arthritis. J Rheumatol. 2005;32:811–9.
Hewlett S, Hehir M, Kirwan JR. Measuring fatigue in rheumatoid arthritis: a systematic review of scales in use. Arthritis Rheum. 2007;57:429–39.
Nicklin J, Cramp F, Kirwan J, et al. Measuring fatigue in rheumatoid arthritis: a cross-sectional study to evaluate the Bristol Rheumatoid Arthritis Fatigue Multi-Dimensional questionnaire, visual analog scales, and numerical rating scales. Arthritis Care Res. 2010;62:1559–68.
Dures EK, Hewlett SE, Cramp FA, et al. Reliability and sensitivity to change of the Bristol Rheumatoid Arthritis Fatigue scales. Rheumatology (Oxford, England). 2013;52:1832–9.
Oude Voshaar MA, Ten Klooster PM, Bode C, et al. Assessment of Fatigue in Rheumatoid Arthritis: A Psychometric Comparison of Single-item, Multiitem, and Multidimensional Measures. J Rheumatol. 2015 Jan 15.
Oude Voshaar MA, Ten Klooster PM, Bode C, Vonkeman HE, Glas CA, Jansen T, et al. Assessment of Fatigue in Rheumatoid Arthritis: A Psychometric Comparison of Single-item, Multiitem, and Multidimensional Measures. The Journal of rheumatology. 2015 Jan 15.
Orbai AM, Smith KC, Bartlett SJ, et al. “Stiffness has different meanings, I think, to everyone”. examining stiffness from the perspective of people living with rheumatoid arthritis. Arthritis Care Res. 2014 May 28. This qualitative research article describes and provides a conceptual framework for stiffness in RA from the patient perspective.
Orbai AM, Smith KC, Bartlett SJ, de Leon E, Bingham CO, 3rd. "Stiffness has different meanings, I think, to everyone". examining stiffness from the perspective of people living with rheumatoid arthritis. Arthritis care & research. 2014 May 28.
Halls S, Dures E, Kirwan J, Pollock J, Baker G, Edmunds A, et al. Stiffness is more than just duration and severity: a qualitative exploration in people with rheumatoid arthritis. Rheumatology (Oxford). In press.
Buttgereit F, Doering G, Schaeffler A, Witte S, Sierakowski S, Gromnica-Ihle E, et al. Efficacy of modifiedrelease versus standard prednisone to reduce duration of morning stiffness of the joints in rheumatoid arthritis (CAPRA-1): a double-blind, randomised controlled trial. Lancet. 2008 Jan 19;371(9608):205-14.
Ward MM, Guthrie LC, Alba MI. Brief report: rheumatoid arthritis response criteria and patient-reported improvement in arthritis activity: is an American College of Rheumatology twenty percent response meaningful to patients? Arthritis & rheumatology (Hoboken, NJ). 2014 Sep;66(9):2339-43.
Inanc N, Yilmaz-Oner S, Can M, et al. The role of depression, anxiety, fatigue, and fibromyalgia on the evaluation of the remission status in patients with rheumatoid arthritis. J Rheumatol. 2014;41:1755–60.
Curtis JR, Shan Y, Harrold L, et al. Patient perspectives on achieving treat-to-target goals: a critical examination of patient-reported outcomes. Arthritis Care Res. 2013;65:1707–12.
Kvien TK, Heiberg T, Hagen KB. Minimal clinically important improvement/difference (MCII/MCID) and patient acceptable symptom state (PASS): what do these concepts mean? Ann Rheum Dis. 2007;66 Suppl 3:iii40–1.
Compliance with Ethics Guidelines
Conflict of Interest
Ana-Maria Orbai declares the receipt of grant money in the form of a Scientist Development Award from the Rheumatology Research Foundation (RRF). Clifton O. Bingham declares the receipt of grant money via a Patient-Centered Outcomes Research Institute (PCORI) Pilot Project Program Award (1IP2-PI000737-01). All statements in this report, including its findings and conclusions, are solely those of the authors and do not necessarily represent the views of the Patient-Centered Outcomes Research Institute (PCORI), its Board of Governors, or Methodology Committee. Dr. Bingham is a member of the Executive Committee of OMERACT (Outcome Measures in Rheumatology), an international group that develops and evaluates health outcomes; OMERACT receives hands-off funding from more than 23 pharmaceutical and research companies.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Rheumatoid Arthritis
Rights and permissions
About this article
Cite this article
Orbai, AM., Bingham, C.O. Patient Reported Outcomes in Rheumatoid Arthritis Clinical Trials. Curr Rheumatol Rep 17, 28 (2015). https://doi.org/10.1007/s11926-015-0501-8
Published:
DOI: https://doi.org/10.1007/s11926-015-0501-8