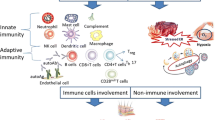
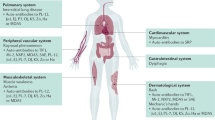
Abstract
In the past three decades, not much has changed in the pathophysiologic concepts of dermatomyositis and polymyositis. However, in the past couple of years, many changes have occurred reflecting the extremely complex nature of the immune response in general. New pathophysiologic models are needed, but at present, none of them encompasses all the recent findings. The changing concepts of dermatomyositis and polymyositis offer new opportunities for unraveling these diseases and developing better strategies for prevention and treatment. This article discusses the most important developments and their methodologic short-comings.
Article PDF
Similar content being viewed by others
References and Recommended Reading
Bohan A, Peter JB: Polymyositis and dermatomyositis (first of two parts). N Eng J Med 1975, 292:344–347.
Bohan A, Peter JB: Polymyositis and dermatomyositis (second of two parts). N Eng J Med 1975, 292:403–407.
Dalakas MC: Polymyositis, dermatomyositis, and inclusion-body myositis. N Eng J Med 1991, 325:1487–1498.
Dalakas MC, Hohlfeld R: Polymyositis and dermatomyositis. Lancet 2003, 362:971–982.
Kissel JT, Mendell JR, Rammohan KW: Microvascular deposition of complement membrane attack complex in dermatomyositis. N Eng J Med 1986, 314:329–334.
Greenberg SA, Amato AA: Uncertainties in the pathogenesis of adult dermatomyositis. Curr Opin Neurol 2004, 17:359–364.
Karpati G, Carpenter S, Melmed C, Eisen AA: Experimental ischemic myopathy. J Neurol Sci 1974, 23:129–161.
Greenberg SA, Pinkus JL, Pinkus GS, et al.: Interferon-alpha/beta-mediated innate immune mechanisms in dermatomyositis. Ann Neurol 2005, 57:664–678.
Strieter RM, Polverini PJ, Kunkel SL, et al.: The functional role of the ELR-motif in CXC chemokine-mediated angiogenesis. J Biol Chem 1995, 270:27348–27357.
Fall N, Bove KE, Stringer K, et al.: Association between lack of angiogenic response in muscle tissue and high expression of angiostatic ELR-negative CXC chemokines in patients with juvenile dermatomyositis. Arthritis Rheum 2005, 52:3175–3180.
Rossi M, Young JW: Human dendritic cells: potent antigen-presenting cells at the crossroads of innate and adaptive immunity. J Immunol 2005, 175:1373–1381.
Page G, Chevrel G, Miossec P: Anatomic localization of immature and mature dendritic cell subsets in dermatomyositis and polymyositis. Arthritis Rheum 2004, 50:199–208.
Dieu MC, Vanbervliet B, Vicari A, et al.: Selective recruitment of immature and mature dendritic cells by distinct chemokines expressed in different anatomic sites. J Exp Med 1998, 188:373–386.
Page G, Lebecque S, Miossec P: Anatomic localization of immature and mature dendritic cells in an ectopic lymphoid organ: correlation with selective chemokine expression in rheumatoid synovium. J Immunol 2002, 168:5333–5341.
Ngo VN, Tang HL, Cyster JG: Epstein-Barr virus-induced molecule 1 ligand chemokine is expressed by dendritic cells in lymphoid tissues and strongly attracts naïve T cells and activated B cells. J Exp Med 1998, 188:181–191.
Arahata K, Engel AG: Monoclonal antibody analysis of mononuclear cells in myopathies. I: Quantitation of subsets according to diagnosis and sites of accumulation and demonstration and counts of muscle fibers invaded by T cells. Ann Neurol 1984, 16:193–208.
Siegal FP, Kadowaki N, Shodell M, et al.: The nature of the principal type 1 interferon-producing cells in human blood. Science 1999, 284:1835–1837.
Greenberg SA, Pinkus GS, Amato AA, Pinkus JL: Myeloid dendritic cells in inclusion-body myositis and polymyositis. Muscle Nerve 2007, 35:17–23.
Fyhr IM, Moslemi AR, Lindberg C, Oldfors A: T cell receptor beta-chain repertoire in inclusion body myositis. J Neuroimmunol 1998, 91:129–134.
Mantegazza R, Andreetta F, Bernasconi P, et al.: Analysis of T cell receptor repertoire of muscle-infiltrating T lymphocytes in polymyositis: restricted V alpha/beta rearrangements may indicate antigen-driven selection. J Clin Invest 1993, 91:2880–2886.
O’Hanlon TP, Dalakas MC, Plotz PH, Miller FW: Predominant TCR-alpha beta variable and joining gene expression by muscle-infiltrating lymphocytes in the idiopathic inflammatory myopathies. J Immunol 1994, 152:2569–2576.
van der Meulen MF, van Wichen DF, van Blokland WT, et al.: Evidence for heterogeneity of T cell expansion in polymyositis and inclusion body myositis. J Neuroimmunol 2002, 133:198–204.
Dimitri D, Benveniste O, Dubourg T, et al.: Shared blood and muscle CD8+ T-cell expansions in inclusion body myositis. Brain 2006, 129:986–995.
Bender A, Ernst N, Iglesias A, et al.: T cell receptor repertoire in polymyositis: clonal expansion of autoaggressive CD8+ T cells. J Exp Med 1995, 181:1863–1868.
Hofbauer M, Wiesener S, Babbe H, et al.: Clonal tracking of autoaggressive T cells in polymyositis by combining laser microdissection, single-cell PCR, and CDR3-spectratype analysis. Proc Natl Acad Sci USA 2003, 100:4090–4095.
Amemiya K, Granger RP, Dalakas MC: Clonal restriction of T-cell receptor expression by infiltrating lymphocytes in inclusion body myositis persists over time: studies in repeated muscle biopsies. Brain 2000, 123:2030–2039.
Muntzing K, Lindberg C, Moslemi AR, Oldfors A: Inclusion body myositis: clonal expansions of muscle-infiltrating T cells persists over time. Scan J Immunol 2003, 58:195–200.
Benveniste O, Herson S, Salomon B, et al.: Long-term persistence of clonally expanded T cells in patients with polymyositis. Ann Neurol 2004, 56:867–872.
Love LA, Leff RL, Fraser DD, et al.: A new approach to the classification of idiopathic inflammatory myopathy: myositis-specific autoantibodies define useful homogeneous patient groups. Medicine (Baltimore) 1991, 70:360–374.
Greenberg SA, Sanoudou D, Haslett JN, et al.: Molecular profiles of inflammatory myopathies. Neurology 2002, 59:1170–1182.
Greenberg SA, Bradshaw EM, Pinkus JL, et al.: Plasma cells in muscle in inclusion body myositis and polymyositis. Neurology 2005, 65:1782–1787.
Bradshaw EM, Orihuela A, McArdel SL, et al.: A local antigen-driven humoral response is present in the inflammatory myopathies. J Immunol 2007, 178:547–556.
Miller FW: Myositis-specific autoantibodies. Touchstones for understanding the inflammatory myopathies. JAMA 1993, 270:1846–1849.
Casciola-Rosen L, Nagaraju K, Plotz P, et al.: Enhanced autoantigen expression in regenerating muscle cells in idiopathic inflammatory myopathy. J Exp Med 2005, 201:591–601.
DeVere R, Bradley WG: Polymyositis: its presentation, morbidity and mortality. Brain 1975, 98:637–666.
Emslie-Smith AM, Arahata K, Engel AG: Major histocompatibility complex class I antigen expression, immunolocalization of interferon subtypes, and T cell-mediated cytotoxicity in myopathies. Hum Pathol 1989, 20:224–231.
Englund P, Nennesmo I, Klareskog L, Lundberg IE: Interleukin-1alpha expression in capillaries and major histocompatibility complex class I expression in type II muscle fibers from polymyositis and dermatomyositis patients: important pathogenic features independent of inflammatory cell clusters in muscle tissue. Arthritis Rheum 2002, 46:1044–1055.
Plotz PH, Dalakas M, Leff RL, et al.: Current concepts in the idiopathic inflammatory myopathies: polymyositis, dermatomyositis, and related disorders. Ann Intern Med 1989, 111:143–157.
Tajima Y, Moriwaka F, Tashiro K: Temporal alterations of immunohistochemical findings in polymyositis. Intern Med 1994, 33:263–270.
Bartoccioni E, Gallucci S, Scuderi F, et al.: MHC class I, MHC class II and intracellular adhesion molecule-I (ICAM-I) expression in inflammatory myopathies. Clin Exp Immunol 1994, 95:166–172.
Nagaraju K, Raben N, Villalba ML, et al.: Costimulatory markers in muscle of patients with idiopathic inflammatory myopathies and in cultured muscle cells. Clin Immunol 1999, 92:161–169.
Nyberg P, Wikman AL, Nennesmo I, Lundberg IE: Increased expression of interleukin 1alpha and MHC class I in muscle tissue of patients with chronic, inactive polymyositis and dermatomyositis. J Rheumatol 2000, 27:940–948.
Nagaraju K, Raben N, Loeffler L, et al.: Conditional up-regulation of MHC class I in skeletal muscle leads to self-sustaining autoimmune myositis and myositis-specific autoantibodies. Proc Natl Acad Sci U S A 2000, 97:9209–9214.
Pavlath GK: Regulation of class I MHC expression in skeletal muscle: deleterious effect of aberrant expression on myogenesis. J Neuroimmunol 2002, 125:42–50.
Kaufman RJ: Stress signaling from the lumen of the endoplasmic reticulum: coordination of gene transcriptional and translational controls. Genes Dev 1999, 13:1211–1233.
Mori K: Tripartite management of unfolded proteins in the endoplasmc reticulum. Cell 2000, 101:451–454.
Nagaraju K, Casciola-Rosen L, Lundberg I, et al.: Activation of the endoplasmic reticulum stress response in autoimmune myositis. Arthritis Rheum 2005, 52:1824–1835.
Guttridge DC, Mayo MW, Madrid LV, et al.: NF-kappaB-induced loss of MyoD messenger RNA: possible role in muscle damage and cachexia. Science 2000, 289:2363–2366.
Dorph C, Englund P, Nennesmo I, Lundberg IE: Signs of inflammation in both symptomatic and asymptomatic muscles from patients with polymyositis and dermatomyositis. Ann Rheum Dis 2006, 65:1565–1571.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Hengstman, G.J.D. Advances in the immunopathophysiology of the idiopathic inflammatory myopathies: Not as simple as suspected. Curr Rheumatol Rep 9, 280–285 (2007). https://doi.org/10.1007/s11926-007-0045-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11926-007-0045-7