Abstract
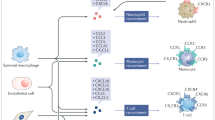
Chemokines are small proteins that can act on cells that express matching receptors. They are best known for their role in migration of cells, especially immune cells. Chemo-kine/ chemokine-receptor pairs are often functionally cate-gorized into three groups: inflammatory, homeostatic, and angiogenic/angiostatic, although functions sometimes over-lap. Interfering with the interaction between chemokines and their receptors is currently under investigation as a therapeutic strategy in rheumatoid arthritis.
Similar content being viewed by others
References and Recommended Reading
Firestein GS: Evolving concepts of rheumatoid arthritis. Nature 2003, 423:356–361.
Haringman JJ, Kraan MC, Smeets TJ, et al.: Chemokine block-ade and chronic inflammatory disease: proof of concept in patients with rheumatoid arthritis. Ann Rheum Dis 2003, 62:715–721. Reports of the first clinical trial in 16 patients with active RA showing decreased synovial inflammation after treatment with an orally administered CCR1 blocking agent.
Woodruff JJ, Clarke LM, Chin YH: Specific cell-adhesion mech-anisms determining migration pathways of recirculating lymphocytes. Annu Rev Immunol 1987, 5:201–222.
Gerard C, Gerard NP: C5A anaphylatoxin and its seven trans-membrane-segment receptor. Annu Rev Immunol 1994, 12:775–808.
Rudolph EH, Woods JM: Chemokine expression and regula-tion of angiogenesis in rheumatoid arthritis. Curr Pharm Des 2005, 11:613–631.Detailed and comprehensible review exploring the role of chemo-kines in angiogenesis in general and in RA in particular.
Haringman JJ, Ludikhuize J, Tak PP: Chemokines in joint disease: the key to inflammation? Ann Rheum Dis 2004, 63:1186–1194.
Sanchez-Madrid F, del Pozo MA: Leukocyte polarization in cell migration and immune interactions. EMBO J 1999, 18:501–511. Explains the mechanism of migration upon chemokine-receptor activation on the cellular level.
Butcher EC: Specificity of leukocyte-endothelial interactions and diapedesis: physiologic and therapeutic implications of an active decision process. Res Immunol 1993, 144:695–698. Explains the process of cell migration from the circulation into tissues.
Fong AM, Robinson LA, Steeber DA, et al.: Fractalkine and CX3CR1 mediate a novel mechanism of leukocyte capture, firm adhesion, and activation under physiologic flow. J Exp Med 1998, 188:1413–1419.
Tak PP: Examination of the synovium and synovial fluid. In Rheumatoid Arthritis: Frontiers in Pathogenesis and Ttreatment. Edited by Wollheim FA, Firestein GS, Panayi GS. Oxford: Oxford University Press; 2000:55–68.
Tak PP: Is early rheumatoid arthritis the same disease process as late rheumatoid arthritis? Best Pract Res Clin Rheumatol 2001, 15:17–26.
Ruth JH, Shahrara S, Park CC, et al.: Role of macrophage inflammatory protein-3alpha and its ligand CCR6 in rheum-atoid arthritis. Lab Invest 2003, 83:579–588.
Endo H, Akahoshi T, Takagishi K, et al.: Elevation of interleu-kin-8 (IL-8) levels in joint fluids of patients with rheumatoid arthritis and the induction by IL-8 of leukocyte infiltration and synovitis in rabbit joints. Lymphokine Cytokine Res 1991, 10:245–252.
Koch AE, Kunkel SL, Burrows JC, et al.: Synovial tissue macro-phage as a source of the chemotactic cytokine IL-8. J Immunol 1991, 147:2187–2195.
Kraan MC, Patel DD, Haringman JJ, et al.: The development of clinical signs of rheumatoid synovial inflammation is associ-ated with increased synthesis of the chemokine CXCL8 (interleukin-8). Arthritis Res 2001, 3:65–71.
Randolph GJ, Furie MB: A soluble gradient of endogenous monocyte chemoattractant protein-1 promotes the trans-endothelial migration of monocytes in vitro. J Immunol 1995, 155:3610–3618.
Koch AE, Kunkel SL, Harlow LA, et al.: Enhanced production of monocyte chemoattractant protein-1 in rheumatoid arthritis. J Clin Invest 1992, 90:772–779.
Luster AD: Chemokines-chemotactic cytokines that mediate inflammation. N Engl J Med 1998, 338:436–445.
Bruhl H, Cihak J, Schneider MA, et al.: Dual role of CCR2 during initiation and progression of collagen-induced arthritis: evi-dence for regulatory activity of CCR2+ T cells. J Immunol 2004, 172:890–898.
Quinones MP, Ahuja SK, Jimenez F, et al.: Experimental arthritis in CC chemokine receptor 2-null mice closely mimics severe human rheumatoid arthritis. J Clin Invest 2004, 113:856–866.
Haringman JJ, Gerlag DM, Smeets TJ, et al.: A randomized placebo controlled trial with an anti-MCP-1(CCL2) mono-clonal antibody in patients with rheumatoid arthritis [abstract]. Arthritis Rheum 2004.
Barnes DA, Tse J, Kaufhold M, et al.: Polyclonal antibody directed against human RANTES ameliorates disease in the Lewis rat adjuvant-induced arthritis model. J Clin Invest 1998, 101:2910–2919.
Toh K, Kukita T, Wu Z, et al.: Possible involvement of MIP-1alpha in the recruitment of osteoclast progenitors to the distal tibia in rats with adjuvant-induced arthritis. Lab Invest 2004, 84:1092–1102.
Zapico I, Coto E, Rodriguez A, et al.: CCR5 (chemokine recep-tor-5) DNA-polymorphism influences the severity of rheu-matoid arthritis. Genes Immun 2000, 1:288–289.
John S, Smith S, Morrison JF, et al.: Genetic variation in CCR5 does not predict clinical outcome in inflammatory arthritis. Arthritis Rheum 2003, 48:3615–3616.
Vierboom MP, Zavodny PJ, Chou CC, et al.: Inhibition of the development of collagen-induced arthritis in rhesus monkeys by a small molecular weight antagonist of CCR5. Arthritis Rheum 2005, 52:627–636.
Shahrara S, Proudfoot AE, Woods JM, et al.: Amelioration of rat adjuvant-induced arthritis by Met-RANTES. Arthritis Rheum 2005, 52:1907–1919.
Cupedo T, Mebius RE: Cellular interactions in lymph node development. J Immunol 2005, 174:21–25. Reviews the embryogenesis of lymph nodes and describes several inflammatory disorders in which ectopic lymphoid tissue formation is seen.
Nishikawa SI, Hashi H, Honda K, et al.: Inflammation, a proto-type for organogenesis of the lymphopoietic/hematopoietic system. Curr Opin Immunol 2000, 12:342–345.
Braun A, Takemura S, Vallejo AN, et al.: Lymphotoxin beta-mediated stimulation of synoviocytes in rheumatoid arthritis. Arthritis Rheum 2004, 50:2140–2150.
Bradfield PF, Amft N, Vernon-Wilson E, et al.: Rheumatoid fibro-blast-like synoviocytes overexpress the chemokine stromal cell-derived factor 1 (CXCL12), which supports distinct patterns and rates of CD4+ and CD8+ T cell migration within synovial tissue. Arthritis Rheum 2003, 48:2472–2482.
Manzo A, Paoletti S, Carulli M, et al.: Systematic microanatom-ical analysis of CXCL13 and CCL21 in situ production and progressive lymphoid organization in rheumatoid synovitis. Eur J Immunol 2005, 35:1347–1359. Recent study, supporting the role of chemokines in the formation of ectopic lymphoid tissue in RA synovium.
Tak PP, Smeets TJ, Daha MR, et al.: Analysis of the synovial cell infiltrate in early rheumatoid synovial tissue in relation to local disease activity 2. Arthritis Rheum 1997, 40:217–225.
Takemura S, Braun A, Crowson C, et al.: Lymphoid neogenesis in rheumatoid synovitis. J Immunol 2001, 167:1072–1080.
Shi K, Hayashida K, Kaneko M, et al.: Lymphoid chemokine B cell-attracting chemokine-1 (CXCL13) is expressed in germi-nal center of ectopic lymphoid follicles within the synovium of chronic arthritis patients. J Immunol 2001, 166:650–655.
Carlsen HS, Baekkevold ES, Morton HC, et al.: Monocyte-like and mature macrophages produce CXCL13 (B cell-attracting chemokine 1) in inflammatory lesions with lymphoid neo-genesis. Blood 2004, 104:3021–3027. Recent study, supporting the role of chemokines in the formation of ectopic lymphoid tissue in RA synovium.
Edwards JC, Szczepanski L, Szechinski J, et al.: Efficacy of B-cell-targeted therapy with rituximab in patients with rheuma-toid arthritis. N Engl J Med 2004, 350:2572–2581.
Zheng B, Ozen Z, Zhang X, et al.: CXCL13 neutralization reduces the severity of collagen-induced arthritis. Arthritis Rheum 2005, 52:620–626.
Qin S, Rottman JB, Myers P, et al.: The chemokine receptors CXCR3 and CCR5 mark subsets of T cells associated with certain inflammatory reactions. J Clin Invest 1998, 101:746–754.
Burman A, Haworth O, Hardie DL, et al.: A chemokine-dependent stromal induction mechanism for aberrant lymphocyte accu-mulation and compromised lymphatic return in rheumatoid arthritis. J Immunol 2005, 174:1693–1700. Recent study, supporting the role of chemokines in the formation of ectopic lymphoid tissue in RA synovium.
Addison CL, Daniel TO, Burdick MD, et al.: The CXC chemo-kine receptor 2, CXCR2, is the putative receptor for ELR+ CXC chemokine-induced angiogenic activity. J Immunol 2000, 165:5269–5277.
Pablos JL, Santiago B, Galindo M, et al.: Synoviocyte-derived CXCL12 is displayed on endothelium and induces angiogenesis in rheumatoid arthritis. J Immunol 2003, 170:2147–2152.
Joven B, Gonzalez N, Aguilar F, et al.: Association between stromal cell-derived factor 1 chemokine gene variant and radiographic progression of rheumatoid arthritis. Arthritis Rheum 2005, 52:354–356.
Grassi F, Cristino S, Toneguzzi S, et al.: CXCL12 chemokine up-regulates bone resorption and MMP-9 release by human osteoclasts: CXCL12 levels are increased in synovial and bone tissue of rheumatoid arthritis patients. J Cell Physiol 2004, 199:244–251.
Koch AE, Volin MV, Woods JM, et al.: Regulation of angiogenesis by the C-X-C chemokines interleukin-8 and epithelial neutro-phil activating peptide 78 in the rheumatoid joint. Arthritis Rheum 2001, 44:31–40.
Van CE, Van A, Wuyts A, et al.: Tumor angiogenesis induced by granulocyte chemotactic protein-2 as a countercurrent principle. Am J Pathol 2001, 159:1405–1414.
Pierer M, Rethage J, Seibl R, et al.: Chemokine secretion of rheumatoid arthritis synovial fibroblasts stimulated by Toll-like receptor 2 ligands. J Immunol 2004, 172:1256–1265.
Pierer M, Rethage J, Seibl R, et al.: Chemokine secretion of rheumatoid arthritis synovial fibroblasts stimulated by Toll-like receptor 2 ligands. J Immunol 2004, 172:1256–1265.
Halloran MM, Woods JM, Strieter RM, et al.: The role of an epithelial neutrophil-activating peptide-78-like protein in rat adjuvant-induced arthritis. J Immunol 1999, 162:7492–7500.
Wooley PH, Schaefer C, Whalen JD, et al.: A peptide sequence from platelet factor 4 (CT-112) is effective in the treatment of type II collagen induced arthritis in mice. J Rheumatol 1997, 24:890–898.
Matthys P, Hatse S, Vermeire K, et al.: AMD3100, a potent and specific antagonist of the stromal cell-derived factor-1 chemo-kine receptor CXCR4, inhibits autoimmune joint inflamma-tion in IFN-gamma receptor-deficient mice. J Immunol 2001, 167:4686–4692.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vergunst, C.E., Tak, P.P. Chemokines: Their role in rheumatoid arthritis. Curr Rheumatol Rep 7, 382–388 (2005). https://doi.org/10.1007/s11926-005-0026-7
Issue Date:
DOI: https://doi.org/10.1007/s11926-005-0026-7