Abstract
Pure menstrual migraine (PMM) and menstrually related migraine (MRM) are difficult challenges in migraine management. Triptans are a class of highly selective serotonin receptor agonists, which interfere with the pathogenesis of migraine and are effective in relieving the associated neurovegetative symptoms. In recent years triptans have been extensively proposed for the treatment of severe, disabling, and recurrent perimenstrual migraine attacks. This review summarizes the different levels of recommendations for the use of triptans in the treatment of perimenstrual migraine. This review is also intended to offer an updated reasonable guide to physicians treating perimenstrual migraine in daily practice.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as:•• Of major importance
Steiner TJ, Stovner LJ, Dua T, et al. Time to act on headache disorders. J Headache Pain. 2011;12:501–3.
Steiner TJ, Birbeck GL, Jensen R, et al. The Global Campaign, World Health Organization and Lifting The Burden: collaboration in action. J Headache Pain. 2011;12:273–4.
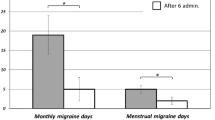
Silberstein SD, Massiou H, Le Jeunne C, et al. Rizatriptan in the treatment of menstrual migraine. Obstet Gynecol. 2000;96:237–42.
•• Vetvik KG, MacGregor EA, Lundqvist C et al. Self-reported menstrual migraine in the general population. J Headache Pain 2010;11:87–92. In this large survey, the authors reported the high prevalence of PMM and MRM in the general population in Norway.
Kvisvik EV, Stovner LJ, Helde G, et al. Headache and migraine during pregnancy and puerperium: the MIGRA-study. J Headache Pain. 2011;12:443–51.
•• Sacco S, Ricci S, Degan D, et al. Migraine in women: the role of hormones and their impact on vascular diseases. J Headache Pain 2012;13:177–189. This recent review highlights the importance of hormones in developing mechanisms and worsening risk factors of migraine in women.
Sulak PJ, Scow RD, Preece C, et al. Hormone withdrawal symptoms in oral contraceptive users. Obstet Gynecol. 2000;95:261–6.
•• Evers S, Afra J, Frese A, et al. EFNS guideline on the drug treatment of migraine—revised report of an EFNS task force. Eur J Neurol. 2009;16:968–81. This is a cornerstone paper in the management of all forms of migraine.
Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders, 2nd edition. Cephalalgia. 2004;24:9–160.
Calhoun AH. Menstrual migraine: update on pathophysiology and approach to therapy and management. Curr Treat Options Neurol. 2012;14:1–14.
Allais G, Castagnoli Gabellari I, Rolando S, et al. Evaluation of the use of sumatriptan-naproxen sodium for menstrual migraine and dysmenorrhea. Expert Rev Neurother. 2011;11(10):1383–7.
Granella F, Sances G, Allais G, et al. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia. 2004;24:707–16.
Couturier EG, Bomhof MA, Neven AK, et al. Menstrual migraine in a representative Dutch population sample: prevalence, disability and treatment. Cephalalgia. 2003;23(4):302–8.
Stewart WF, Lipton RB, Chee E, et al. Menstrual cycle and headache in a population sample of migraineurs. Neurology. 2000;55(10):1517–23.
Monteith TS, Goadsby PJ. Acute migraine therapy: new drugs and new approaches. Curr Treat Options Neurol. 2011;13:1–14.
MacGregor EA, Frith A, Ellis J, Aspinall L, et al. Incidence of migraine relative to menstrual cycle phases of rising and falling estrogen. Neurology. 2006;67:2154–8.
Silberstein S, Merriam G. Sex hormones and headache 1999 (menstrual migraine). Neurology. 1999;53(4 Suppl 1):S3–13.
Moskowitz MA. The neurobiology of vascular head pain. Ann Neurol. 1984;16:157–68.
Taiwo YO, Levine JD. Prostaglandins inhibit endogenous pain control mechanisms by blocking transmission at spinal noradrenergic synapses. J Neurosci. 1988;8:1346–9.
Ferreira SH, Lorenzetti BB, Correa FM. Blockade of central and peripheral generation of prostaglandins explains the antialgic effect of aspirin like drugs. Pol J Pharmacol Pharm. 1978;30:133–40.
Behrman HR, Caldwell BV. Prostaglandins, thromboxanes, and leukotrienes. In: Yen SSC, Jaffe RB, editors. Reproductive endocrinology: physiology, pathophysiology and clinical management. Philadelphia: Saunders; 1986. p. 154–76.
Nattero G, Allais G, De Lorenzo C, et al. Relevance of prostaglandins in true menstrual migraine. Headache. 1989;29:233–8.
Papakostas Y, Daras M, Markianos M, et al. Increased prolactin response to thyrotropin releasing hormone during migraine attacks. J Neurol Neurosurg Psychiatry. 1987;50:927–8.
Yen SSC. Prolactin in human reproduction. In: Yen SSC, Jaffe RB, editors. Reproductive endocrinology, physiology, pathophysiology and clinical management. Philadelphia: Saunders; 1986. p. 237–63.
Nappi G, Martignoni E. Significance of hormonal changes in primary headache disorders. In: Olesen J, Edvinsson L, editors. Basic mechanisms of headache. New York: Elsevier; 1988. p. 277–98.
Schreiber CP, Cady RK. Diagnosis of menstrual headache and an open-label study among those with previously undiagnosed menstrually related migraine to evaluate the efficacy of sumatriptan 100 mg. Clin Ther. 2007;29:2511–9.
Landy S, Savani N, Shackelford S, et al. Efficacy and tolerability of sumatriptan tablets administered during the mild-pain phase of menstrually associated migraine. Int J Clin Pract. 2004;58:913–9.
Nett R, Landy S, Shackelford S, et al. Pain-free efficacy after treatment with sumatriptan in the mild pain phase of menstrually associated migraine. Obstet Gynecol. 2003;102:835–42.
Dowson AJ, Massiou H, Aurora SK. Managing migraine headaches experienced by patients who self-report with menstrually related migraine: a prospective, placebo-controlled study with oral sumatriptan. J Headache Pain. 2005;6:81–7.
Brainin M, Barnes M, Baron J-C, et al. Guidance for the preparation of neurological management guidelines by EFNS scientific task forces—revised recommendations 2004. Eur J Neurol. 2004;11:577–81.
Pringsheim T, Davenport WJ, Dodick D. Acute treatment and prevention of menstrually related migraine headache: evidence-based review. Neurology. 2008;70:1555–63.
Martin V, Cady R, Mauskop A, et al. Efficacy of rizatriptan for menstrual migraine in an early intervention model: a prospective subgroup analysis of the rizatriptan TAME (Treat A Migraine Early) studies. Headache. 2008;48:226–35.
Nett R, Mannix LK, Mueller L, et al. Rizatriptan efficacy in ICHD-II pure menstrual migraine and menstrually related migraine. Headache. 2008;48:1194–201.
Mannix LK, Loder E, Nett R, et al. Rizatriptan for the acute treatment of ICHD-II proposed menstrual migraine: two prospective, randomized, placebo-controlled, double-blind studies. Cephalalgia. 2007;27:414–21.
Silberstein SD, Massiou H, McCarroll KA, et al. Further evaluation of rizatriptan in menstrual migraine: retrospective analysis of long-term data. Headache. 2002;42:917–23.
Tuchman M, Hee A, Emeribe U, et al. Efficacy and tolerability of zolmitriptan oral tablet in the acute treatment of menstrual migraine. CNS Drugs. 2006;20:1019–26.
Allais G, Bussone G, D’Andrea G, et al. Almotriptan 12.5 mg in menstrually related migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2011;31:144–51.
Loder E, Silberstein SD, Abu-Shakra S, et al. Efficacy and tolerability of oral zolmitriptan in menstrually associated migraine: a randomized, prospective, parallel-group, double-blind, placebo-controlled study. Headache. 2004;44:120–30.
Silberstein SD, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology. 2004;63:261–9.
Allais G, Bussone G, Airola G, et al. Oral contraceptive-induced menstrual migraine. Clinical aspects and response to frovatriptan. Neurol Sci. 2008;29(Suppl 1):S186–90.
MacGregor EA, Pawsey SP, Campbell JC, et al. Safety and tolerability of frovatriptan in the acute treatment of migraine and prevention of menstrual migraine: results of a new analysis of data from five previously published studies. Gend Med. 2010;7(2):88–108.
Bartolini M, Giamberardino MA, Lisotto C, et al. Frovatriptan versus almotriptan for acute treatment of menstrual migraine: analysis of a double-blind, randomized, cross-over, multicenter, Italian, comparative study. J Headache Pain. 2012. doi:10.1007/s10194-012-0455-4.
Massiou H, Jamin C, Hinzelin G, et al. Efficacy of oral naratriptan in the treatment of menstrually related migraine. Eur J Neurol. 2005;12(10):774–81.
Diamond ML, Cady RK, Mao L, et al. Characteristics of migraine attacks and responses to almotriptan treatment: a comparison of menstrually related and nonmenstrually related migraines. Headache. 2008;48(2):248–58.
Lionetto L, Fiorillo M, Martelletti P. Efficacy and tolerability of almotriptan in menstrual migraine: a comment. Expert Opin Pharmacother. 2011;12:2279–81.
Massiou H, Pitei D, Poole P, et al. Efficacy of eletriptan for the treatment of migraine in women with menstrually associated migraine, and in women on contraceptives or hormone replacement therapy: meta-analyses of randomized clinical trials [abstract]. Cephalalgia. 2000;20:435.
MacGregor EA, Frith A, Ellis J, et al. Predicting menstrual migraine with a home-use fertility monitor. Neurology. 2005;64:561–3.
Brandes JL, Poole A, Kallela M, et al. Short-term frovatriptan for the prevention of difficult-to-treat menstrual migraine attacks. Cephalalgia. 2009;29:1133–48.
Mannix LK, Savani N, Landy S, et al. Efficacy and tolerability of naratriptan for short-term prevention of menstrually related migraine: data from two randomized, double-blind, placebo-controlled studies. Headache. 2007;47:1037–49.
Newman L, Mannix LK, Landy S, Silberstein S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized double-blind, placebo-controlled study. Headache. 2001;41:248–56.
Newman LC, Lipton RB, Lay CL, et al. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology. 1998;51:307–9.
Tuchman MM, Hee A, Emeribe U, et al. Oral zolmitriptan in the short-term prevention of menstrual migraine: a randomized, placebo-controlled study. CNS Drugs. 2008;22:877–86.
Marcus DA, Bernstein CD, Sullivan EA, et al. Perimenstrual eletriptan prevents menstrual migraine: an open-label study. Headache. 2010;50:551–56.
Negro A, Martelletti P. Chronic migraine plus medication overuse headache: two entities or not? J Headache Pain. 2011;12:593–601.
Disclosures
B. Casolla: none; L. Lionetto: none; S. Candela: none. Dr. Lidia D’Alonzo has received travel expense compensation from Pfizer. Dr. Andrea Negro has received travel expense compensation from Menarini and Pfizer. Dr. Maurizio Simmaco has received payment for the development of educational presentations from Diatech Pharmacogenomics. Dr. Paolo Martelletti is a member of an advisory board to Allergan, Inc.; has received grants, honoraria, and/or research support from ACRAF, Allergan, and Almirall; has received payment for the development of educational presentations from Allergan, ACRAF Angelini, Almirall, and Guidotti; and has received travel expense compensation from Nevro Corporation, Menarini, and the Lifting The Burden campaign.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Casolla, B., Lionetto, L., Candela, S. et al. Treatment of Perimenstrual Migraine with Triptans: an Update. Curr Pain Headache Rep 16, 445–451 (2012). https://doi.org/10.1007/s11916-012-0280-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11916-012-0280-0