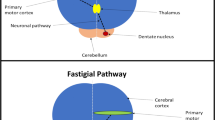
Abstract
Purpose of Review
First, an anatomical and functional review of these cortical areas and subcortical connections with T-fMRI and tractography techniques; second, to demonstrate the value of this approach in neurosurgical planning in a series of patients with tumors close to the SMA.
Recent Findings
Implications in language and cognitive networks with a clear hemispheric lateralization of these SMA/pre-SMA. The recommendation of the use of the advanced neuroimaging studies for surgical planning and preservation of these areas.
Summary
The SMA/pre-SMA and their subcortical connections are functional areas to be taken into consideration in neurosurgical planning. These areas would be involved in the control/inhibition of movement, in verbal expression and fluency and in tasks of cognitive control capacity. Its preservation is key to the patient’s postsurgical cognitive and functional evolution.






Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bizzi A, Blasi V, Falini A, Ferroli P, Cadioli M, Danesi U, et al. Presurgical functional MR imaging of language and motor functions: validation with intraoperative electrocortical mapping. Radiology. 2008;248:579–89.
Talos I, O’Donnell L, Westin C, Warfield S, Wells W, Yoo S et al. Diffusion tensor and functional MRI fusion with anatomical MRI for image-guided neurosurgery. In: Proceedings of the Sixth International Conference on Medical Image Computing and Computer-Assisted Intervention MICCAI’03: Lecture Notes in Computer Science, Montréal 2003; 2878:407–415.
Castellano A, Cirillo S, Bello L, Riva M, Falini A. Functional MRI for surgery of gliomas. Curr Treat Options Neurol. 2017;19:34.
Kleiser R, Staempfli P, Valavanis A, Boesiger P, Kollia S. Impact of fMRI-guided advanced DTI fiber tracking techniques on their clinical applications in patients with brain tumors. Neuroradiology. 2010;52:37–46.
• Essayed W, Zhang F, Unadkat P, Cosgrove G, Golby A, O’Donnell L. White matter tractography for neurosurgical planning: a topography-based review of the current state of the art. Neuroimage Clinl. 2017;15:659–72 A review of the literature in the field of tractography in neurosurgery where the authors discuss its multiple applications and its implications in clinical care.
• Wang H, Tong E, Zaharchuk W, Zeineh M, Goldstein-Piekarski BA, Liao C, et al. Resting-state functional MRI: everything that nonexperts have always wanted to know. AJNR Am J Neuroradiol. 2018;39:1390–9 An article for familiarizing the neuroradiologist with Resting State fMRI, facilitating the use in neurologic and psychiatric conditions.
Barnett A, Audrain S, McAndrews M. Applications of resting-state functional MR imaging to epilepsy. Neuroimaging Clin N Am. 2017;27:697–708.
Agarwal S, Sai H, Pillai J. Limitations of resting-state functional MR imaging in the setting of focal brain lesions. Neuroimaging Clin N Am. 2017;27:645–61.
Billings J, Eder M, Flood W, Singh Dhami D, Natarajan S, Whitlow C. Machine learning applications to resting-state functional MR imaging analysis. Neuroimaging Clin N Am. 2017;27:609–20.
Gil-Robles S, Carvallo A, Jimenez M, Gomez Caicoya A, Martinez R, Ruiz-Ocaña C, et al. Double dissociation between visual recognition and picture naming: a study of the visual language connectivity using tractography and brain stimulation. Neurosurgery. 2013;72(4):678–86.
Jiménez Peña MM, Gil Robles S, Cano Alonso R, Recio Rodríguez M, Carrascoso Arranz J, Ruiz-Ocaña C, et al. Essential subcortical tracts in language and reading. 3D-tractography for clinical practice and anatomic correlation with intraoperative subcortical electrostimulation. Clin Neuroradiol. 2015;27:81–9.
Rostomily RC, Berger MS, Ojemann GA, Lettich E. Postoperative deficits and functional recovery following removal of tumors involving the dominant hemisphere supplementary motor area. J Neurosurg. 1991;75:62–8.
Krainik A, Lehéricy S, Duffau H, et al. Role of the supplementary motor area in motor deficit following medial frontal lobe surgery. Neurology. 2001;57:871–8.
Martino J, Gabarrós A, Deus J, Juncadella M, Acebes JJ, Torres A, et al. Intrasurgical mapping of complex motor function in the superior frontal gyrus. Neuroscience. 2011;179:131–42.
Ford A, McGregor KM, Case K, et al. Structural connectivity of Broca’s area and medial frontal cortex. Neuroimage. 2010;52:1230–7.
Bizzi A, Nava S, Ferré F, et al. Aphasia induced by gliomas growing in the ventrolateral frontal region: assessment with diffusion MR tractography, functional MR imaging and neuropsychology. Cortex. 2012;48:255–72.
Catani M, Mesulam MM, Jakobsen E, Malik F, Martersteck A, Wieneke C, et al. A novel frontal pathway underlies verbal fluency in primary progressive aphasia. Brain. 2013;136:2619–28.
• Puglisi G, Sciortino T, Rossi M, Leonetti A, Fornia L, Conti Nibali M, et al. Preserving executive functions in non-dominant frontal lobe glioma surgery: an intraoperative tool. J Neurosurg. 2018;28:1–7 The authors evaluated the accuracy of the intraoperative version of the Stroop task to reduce the postoperative deficits in the executive functions, of right frontal gliomas.
Rossi M, Fornia L, Puglisi G, Leonetti A, Zuccon G, Fava E, et al. Assessment of the praxis circuit in glioma surgery to reduce the incidence of postoperative and long-term apraxia: a new intraoperative test. J Neurosurg. 2018;23:1–11.
Thiebaut de Schotten M, Dell’Acqua F, Forkel SJ, Simmons A, Vergani F, Murphy DGM, et al. A lateralized brain network for visuospatial attention. Nat Neurosci. 2011;14:1245–6.
Aron AR, Herz DM, Brown P, Forstmann BU, Zaghloul K. Frontosubthalamic circuits for control of action and cognition. J Neurosci. 2016;36:11489–95.
Martino J, de Lucas EM, Ibáñez-Plágaro FJ, Valle-Folgueral JM, Vázquez-Barquero A. Foix-Chavany-Marie syndrome caused by a disconnection between the right pars opercularis of the inferior frontal gyrus and the supplementary motor area. J Neurosurg. 2012;117:844–50.
• Black D, Vachha B, Mian A, Faro S, Maheshwari M, Sair H, et al. American society of functional neuroradiology recommended fMRI paradigm algorithms for presurgical language assessment. Am J Neuroradiol. 2017;38:65–73 The ASFNR proposes fMRI languages paradigm algorithms as standard clinical practice, that provide a common information to compare across the institutions the fMRI data.
Zentner J, Hufnagel A, Pechstein U, Wolf HK, Schramm J. Functional results after resective procedures involving the supplementary motor area. J Neurosurg. 1996;85:542–9.
Pai MC. Supplementary motor area aphasia: a case report. Clin Neurol Neurosurg. 1999;101:29–32.
Chung G, Han Y, Jeong S, Clifford R. Functional heterogeneity of the supplementary motor area. AJNR Am J Neuroradiol. 2005;26:1819–23.
•• Wongsripuemtet J, Tyan A, Carass A, Agarwal S, Gujar S, Pillai J, et al. Preoperative mapping of the supplementary motor area in patients with brain tumor using resting-state fMRI with seed-based analysis. AJNR Am J Neuroradiol. 2018;39:1493–8 Authors demonstrate that the use of the resting state fMRI in the surgical planning of glioma of the SMA is an effective and reliable method.
Sanvito F, Caverzasi E, Riva M, Jordan KM, Blasi V, Scifo P, et al. F-MRI-targeted high angular resolution diffusion MR-Tractography to identify functional language tracts in healthy controls and glioma patients. Front Neurosci. 2020;31:14–225.
Bizzi A. Presurgical mapping of verbal language in brain tumors with functional MR imaging and MR tractography. Neuroimaging Clin N Am. 2009;19:573–96.
Northoff G, Richter A, Gessner M, Schlagenhauf F, Fell J, Baumgart F, et al. Functional dissociation between medial and lateral prefrontal cortical spatiotemporal activation in negative and positive emotions: a combined fMRI/MEG study. Cereb Cortex. 2000;10:93–107.
Oliveri M, Babiloni C, Filippi MM, Caltagirone C, Babiloni F, Cicinelli P, et al. Influence of the supplementary motor area on primary motor cortex excitability during movements triggered by neutral or emotionally unpleasant visual cues. Exp Brain Res. 2003;149:214–2.
•• Dick A, Garic D, Graziano P, Tremblay P. The frontal aslant tract and its role in speech, language and executive function. Cortex. 2019;111:148–63 This article provide an exquisite review of the anatomy and laterality function of the frontal aslant tract.
Catani M, Dell’acqua F, Vergani F, Malik F, Hodge H, Roy P, et al. Short frontal lobe connections of the human brain. Cortex. 2012;48:273–91.
Vergani F, Lacerda L, Martino J, Attems J, Morris C, Mitchell P, et al. White matter connections of the supplementary motor area in humans. J Neurol Neurosurg Psychiatry. 2014;85:1377–85.
Martino J, De Lucas E. Subcortical anatomy of the lateral association fascicles of the brain: a review. Clin Anat. 2014;27:563–9.
Ingham R, Ingham J, Euler H, Neumann K. Stuttering treatment and brain research in adults: a still unfolding relationship. J Fluen Disord. 2017. https://doi.org/10.1016/j.jfludis.2017.02.003.
Tremblay P, Gracco VL. Contribution of the pre-SMA to the production of words and non-speech oral motor gestures, as revealed by repetitive transcranial magnetic stimulation (rTMS). Brain Res. 2009;1268:112–24.
Tremblay P, Gracco VL. On the selection of words and oral motor responses: evidence of a response-independent fronto-parietal network. Cortex. 2010;46:15–28.
Aron A, Monsell S, Sahakian B, Robbins T. A componential analysis of task-switching deficits associated with lesions of left and right frontal cortex. Brain. 2004;127:1561–73.
Aron A, Robbins T, Poldrack R. Inhibition and the right inferior frontal cortex: one decade on. Trends Cogn Sci. 2014;18:177–85.
Van Wouwe NC, Pallavaram S, Phibbs FT, Martinez-Ramirez D, Neimat JS, Dawant BM, et al. Focused stimulation of dorsal subthalamic nucleus improves reactive inhibitory control of action impulses. Neuropsychologia. 2017;99:37–47.
Baker CM, Burks JD, Briggs RG, Smitherman AD, Glenn CA, Conner AK, et al. Crossed frontal aslant tract: a possible pathway involved in the recovery of supplementary motor area syndrome. Brain Behav. 2018. https://doi.org/10.1002/brb3.926.
Lehéricy S, Ducros M, Krainik A, Francois C, van de Moortele P, Ugurbil K, et al. 3-D diffusion tensor axonal tracking shows distinct SMA and pre-SMA projections to the human striatum. Cereb Cortex. 2004;14:1302–9.
Liston C, Watts R, Tottenham N, Davidson M, Niogi S, Ulug A, et al. Frontostriatal microstructure modulates efficient recruitment of cognitive control. Cereb Cortex. 2006;16:553–60.
Laplane D, Talairach J, Meininger V, Bancaud J, Orgogozo J. Clinical consequences of corticectomies involving the supplementary motor area in man. J Neurol Sci. 1977;34:301–14.
Fontaine D, Capelle L, Duffau H. Somatotopy of the supplementary motor area: evidence from correlation of the extent of surgical resection with the clinical patterns of deficit. Neurosurgery. 2002;50:297–303.
Russell S, Kelly P. Incidence and clinical evolution of postoperative deficits after volumetric stereotactic resection of glial neoplasms involving the supplementary motor area. Neurosurgery. 2003;52:506–16.
Rosenberg K, Nossek E, Liebling R, Fried I, Shapira-Lichter I, Hendler T, et al. Prediction of neurological deficits and recovery after surgery in the supplementary motor area: a prospective study in 26 patients. J Neurosurg. 2010;113:1152–63.
Mendez M. Aphemia-like syndrome from a right supplementary motor area lesion. Clin Neurol Neurosurg. 2004;106:337–9.
Swanberg M, Nasreddine Z, Mendez M, Cummings J. Speech and language. In: Goetz C, editor. Text book of clinical neurology. Philadelphia: Saunders Elsevier; 2007.
Puglisi G, Howells H, Sciortino T, Leonetti A, Rossi M, Conti Nibali M, et al. Frontal pathways in cognitive control: direct evidence from intraoperative stimulation and diffusion tractography. Brain. 2019;142:2451–65.
Nambu A, Tokuno H, Takada M. Functional significance of the cortico-subthalamo-pallidal ‘hyperdirect’ pathway. Neurosci Res. 2002;43:111–7.
Gerloff C, Bushara K, Sailer A, Wassermann E, Chen R, Matsuoka T, et al. Multimodal imaging of brain reorganization in motor areas of the contralesional hemisphere of well recovered patients after capsular stroke. Brain. 2006;129:791–808.
Park C, Chang W, Ohn S, Kim S, Bang O, et al. Longitudinal changes of resting-state functional connectivity during motor recovery after stroke. Stroke. 2011;42:1357–62.
Rehme A, Fink G, von Cramon D, Grefkes C. The role of the contralesional motor cortex for motor recovery in the early days after stroke assessed with longitudinal FMRI. Cereb Cortex. 2011;21:756–68.
Sharma N, Baron J, Rowe J. Motor imagery after stroke: relating outcome to motor network connectivity. Ann Neurol. 2009;66:604–16.
Grefkes C, Nowak D, Eickhoff S, Dafotakis M, Kust J, Karbe H, et al. Cortical connectivity after subcortical stroke assessed with functional magnetic resonance imaging. Ann Neurol. 2008;63:236–46.
Lotze M, Beutling W, Loibl M, Domin M, Platz T, Schminke U, et al. Contralesional motor cortex activation depends on ipsilesional corticospinal tract integrity in well-recovered subcortical stroke patients. Neurorehabil Neural Repair. 2011;26:594–603.
Robles SG, Gatignol P, Lehericy S, Duffau H. Long-term brain plasticity allowing a multistage surgical approach to World Health Organization grade II gliomas in eloquent areas. J Neurosurg. 2008;109:615–24.
Rosenberg K, Liebling R, Avidan G, Perry D, Siman-Tov T, Andelman F, et al. Language related reorganization in adult brain with slow growing glioma: fMRI prospective case-study. Neurocase. 2008;14:465–73.
Tombari D, Loubinoux I, Pariente J, Gerdelat A, Albucher J, Tardy J, et al. A longitudinal fMRI study: in recovering and then in clinically stable subcortical stroke patients. Neuroimage. 2004;23:827–39.
Kielar A, Deschamps T, Jokel R, Meltzer J. Functional reorganization of language networks for semantics and syntax in chronic stroke: evidence from MEG. Hum Brain Mapp. 2016;37:2869–93.
Saur D, Lange R, Baumgaertner A, Schraknepper V, Willmes K, Rijntjes M, et al. Dynamics of language reorganization after stroke. Brain. 2006;129(6):1371–84.
Deverdun J, van Dokkum L, Le Bars E, et al. Language reorganization after resection of low-grade gliomas: an fMRI task based connectivity study. Brain Imaging Behav. 2019. https://doi.org/10.1007/s11682-019-00114-7.
Duffau H. Lessons from brain mapping in surgery for low-grade glioma: insights into associations between tumour and brain plasticity. Lancet Neurol. 2005;4:476–86.
Vassal M, Charroud C, Deverdun J. Recovery of functional connectivity of the sensorimotor network after surgery for diffuse low-grade gliomas involving the supplementary motor area. J Neurosurg. 2017;126:1181–90.
Ghinda C, Duffau H. Network plasticity and intraoperative mapping for personalized multimodal management of diffuse low-grade gliomas. Front Surg. 2017;31(4):3. https://doi.org/10.3389/fsurg.2017.00003.
• Liu H, Cai W, Xu L, Li W, Qin W. Differential reorganization of SMA subregions after stroke: a subregional level resting-state functional connectivity study. Front Hum Neurosci. 2020;13:468. https://doi.org/10.3389/fnhum.2019.00468The authors evidence the different cortical reorganization of the SMA proper and the pre-SMA with the use of resting state fMRI, suggesting that these two subregions exhibit completely different within the motor network.
• Chivukula S, Pikul B, Black K, Pouratian N, Bookheimer S. Contralateral functional reorganization of the speech supplementary motor area following neurosurgical tumor resection. Lang. 2018;183:41–6 Authors identify functional reorganization of the speech SMA observing a migration of speech SMA function from the dominant hemisphere to contralateral homologous region, parallel to the clinical recovery.
Acioly MA, Cunha AM, Parise M, Rodrigues E, Tovar-Moll F. Recruitment of contralateral supplementary motor area in functional recovery following medial frontal lobe surgery: an fMRI case study. J Neurol Surg Part A Central Eur Neurosurg. 2015;76:508–12.
Riecker A, Wildgruber D, Grodd W, Ackermann H. Reorganization of speech production at the motor cortex and cerebellum following capsular infarction: a follow-up functional magnetic resonance imaging study. Neurocase. 2002;8:417–23.
Winhuisen L, Thiel A, Schumacher B, Kessler J, Rudolf J, Haupt WF, et al. Role of the contralateral inferior frontal gyrus in recovery of language function in poststroke aphasia: a combined repetitive transcranial magnetic stimulation and positron emission tomography study. Stroke. 2005;36:1759–63.
Hung Y, Gaillard SL, Yarmak P, Arsalidou M. Dissociations of cognitive inhibition, response inhibition, and emotional interference: voxelwise ALE meta-analyses of fMRI studies. Hum Brain Mapp. 2018;39:4065–82.
Banich MT, Milham MP, Atchley R, Cohen NJ, Webb A, Wszalek T, et al. fMRI studies of stroop tasks reveal unique roles of anterior and posterior brain systems in attentional selection. J Cogn Neurosci. 2000;12:988–1000.
Acknowledgments
We wish to thank Caroline Coope for her support in proofreading this article and Pablo García-Polo and Juan Bachiller for their technical counselling.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of Topical Collection on Behavior
Rights and permissions
About this article
Cite this article
de la Peña, M.J., Gil-Robles, S., de Vega, V.M. et al. A Practical Approach to Imaging of the Supplementary Motor Area and Its Subcortical Connections. Curr Neurol Neurosci Rep 20, 50 (2020). https://doi.org/10.1007/s11910-020-01070-2
Published:
DOI: https://doi.org/10.1007/s11910-020-01070-2