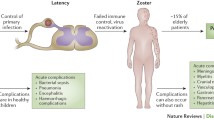
Abstract
Varicella zoster virus (VZV) is an exclusively human neurotropic alphaherpesvirus. Primary infection causes varicella (chickenpox), after which the virus becomes latent in ganglionic neurons along the entire neuraxis. With advancing age or immunosuppression, cell-mediated immunity to VZV declines, and the virus reactivates to cause zoster (shingles), dermatomal distribution, pain, and rash. Zoster is often followed by chronic pain (postherpetic neuralgia), cranial nerve palsies, zoster paresis, vasculopathy, meningoencephalitis, and multiple ocular disorders. This review covers clinical, laboratory, and pathological features of neurological complications of VZV reactivation, including diagnostic testing to verify active VZV infection in the nervous system. Additional perspectives are provided by discussions of VZV latency, animal models to study varicella pathogenesis and immunity, and of the value of vaccination of elderly individuals to boost cell-mediated immunity to VZV and prevent VZV reactivation.




Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as:• Of importance •• Of major importance
Gilden DH, Dueland AN, Cohrs R, et al. Preherpetic neuralgia. Neurology. 1991;41:1215–18.
Harnisch JP. Zoster in the elderly: clinical, immunologic and therapeutic considerations. J Am Geriatr Soc. 1984;32:789–93.
Gilden DH, Cohrs RJ, Mahalingam R. Clinical and molecular pathogenesis of varicella virus infection. Viral Immunol. 2003;16:243–58.
Tyndall MW, Nasio J, Agoki E, et al. Herpes zoster as the initial presentation of human immunodeficiency virus type 1 infection in Kenya. Clin Infect Dis. 1995;21:1035–7.
Lapresle J, Lasjaunias P. Cranial nerve ischaemic arterial syndromes. Brain. 1986;109:207–15.
•• Hevner R, Vilela M, Rostomily R, et al. An unusual cause of trigeminal-distribution pain and tumor. Lancet Neurol. 2003;2:567–72. A remarkable case of an inflammatory mass that produced prolonged dermatomal- distribution pain caused by VZV in the absence of rash.
•• Head H, Campbell AW. The pathology of herpes zoster and its bearing on sensory localization. Brain. 1900;23:353–523. A classic paper that lead to identification of dermatomes served by specific ganglia.
• Watson CP, Deck JH, Morshead C, Van der Kooy D, Evans RJ. Postherpetic neuralgia: further post-mortem studies of cases with and without pain. Pain. 1991;44:105–17. Correlation of PHN with pathological changes in ganglia.
Habib AA, Gilden D, Schmid DS, Safdieh JE. Varicella zoster virus meningitis with hypoglycorrhachia in the absence of rash and in an immunocompetent woman. J Neurovirol. 2009;15:206–8.
Gunson RN, Aitken C, Gilden D. A woman with acute headache and sacral dermatomal numbness. J Clin Virol. 2011;50:191–3.
Moses H, Nagel MA, Gilden DH. Acute cerebellar ataxia in a 41 year old woman. Lancet Neurol. 2006;5:984–8.
Gilden DH, Beinlich BR, Rubinstien EM, et al. Varicella zoster virus myelitis: an expanding spectrum. Neurology. 1994;44:1818–23.
Kleinschmidt-DeMasters BK, Gilden DH. Varicella zoster virus infections of the nervous system: clinical and pathologic correlates. Arch Pathol Lab Med. 2001;125:770–80.
de Silva SM, Mark AS, Gilden DH, et al. Zoster myelitis: improvement with antiviral therapy in two cases. Neurology. 1996;47:929–31.
Orme HT, Smith AG, Nagel MA, et al. VZV spinal cord infarction identified by diffusion-weighted magnetic resonance imaging (DWI). Neurology. 2007;69:398–400.
Kang JH, Ho JD, Chen YH, Lin HC. Increased risk of stroke after a herpes zoster attack: a population-based follow-up study. Stroke. 2010;40:3443–8.
Lin HC, Chien CW, Ho JD. Herpes zoster ophthalmicus and the risk of stroke: a population-based follow-up study. Neurology. 2010;74:792–7.
•• Nagel MA, Cohrs RJ, Mahalingam R, et al. The varicella zoster virus vasculopathies: clinical, CSF, imaging and virological features. Neurology. 2008;70:853–60. Comprehensive study of the VZV vasculopathies.
Nagel MA, Forghani B, Mahalingam R, et al. The value of detecting anti-VZV IgG antibody in CSF to diagnose VZV vasculopathy. Neurology. 2007;68:1069–73.
• Nagel MA, Traktinskiy I, Azarkh Y, et al. Varicella zoster virus vasculopathy: analysis of virus-infected arteries. Neurology. 2011;77:364–70. Correlative analysis of viral and muscle cell markers in cerebral arteries from patients with VZV vasculopathy.
• Nagel MA, Traktinskiy I, Stenmark KR, et al. Varicella zoster virus vasculopathy: Immune characteristics of virus-infected arteries. Neurology. 2013;80:62–8. Analysis of the immune repertoire in arteries of patients with VZV vasculopathy.
•• Nagel MA, Bennett JL, Khmeleva N, et al. Multifocal VZV vasculopathy with temporal artery infection mimics giant cell arteritis. Neurology. 2013;80:17–21. Exciting new study indicating that the clinical features of multifocal VZV vasculopathy with temporal artery infection can be the same as seen in classic GCA.
Lewis GW. Zoster sine herpete. Br Med J. 1958;2:418–21.
• Gilden DH, Wright RR, Schneck SA, Gwaltney Jr JM, Mahalingam R. Zoster sine herpete, a clinical variant. Ann Neurol. 1994;35:530–3. Correlative clinical-virologic analysis proving that zoster sine herpete is produced by VZV.
Amlie-Lefond C, Mackin GA, Ferguson M, et al. Another case of virologically confirmed zoster sine herpete with electrophysiologic correlation. J Neurovirol. 1996;2:136–8.
Blumenthal DT, Shacham-Shmueli E, Bokstein F, et al. Zoster sine herpete: virological verification by detection of anti-VZV IgG antibody in CSF. Neurology. 2011;76:484–5.
Franco-Paredes C, Bellehemeur T, Merchant A, et al. Aseptic meningitis and optic neuritis preceding varicella zoster progressive outer retinal necrosis in a patient with AIDS. AIDS. 2002;16:1045–9.
Menerath JM, Gerard M, Laurichesse H, et al. Bilateral acute retinal necrosis in a patient with acquired immunodeficiency syndrome. J Fr Ophtalmol. 1995;18:625–33.
Haug A, Mahalingam R, Cohrs RJ, et al. Recurrent polymorphonuclear pleocytosis with increased red blood cells caused by varicella zoster virus infection of the central nervous system. J Neurol Sci. 2010;292:85–8.
• Mahalingam R, Wellish M, Soike K, et al. Simian varicella virus infects ganglia before rash in experimentally infected monkeys. Virology. 2001;279:339–42. The first study to show that during primary varicella infection, ganglia are infected hematogenously.
Davison AJ, Scott JE. The complete DNA sequence of varicella zoster virus. J Gen Virol. 1986;66:1759–816.
Cohrs RJ, Hurley MP, Gilden DH. Array analysis of viral gene transcription during lytic infection of cells in tissue culture with Varicella zoster virus. J Virol. 2003;77:11718–32.
Brazeau E, Mahalingam R, Gilden D, et al. Varicella zoster virus–induced apoptosis in MeWo cells is accompanied by down-regulation of Bcl-2 expression. J Neurovirol. 2010;16:133–40.
Furuta Y, Takasu T, Fukuda S, et al. Detection of varicella zoster virus DNA in human geniculate ganglia by polymerase chain reaction. J Infect Dis. 1992;166:1157–9.
•• Gilden DH, Vafai A, Shtram Y, et al. Varicella zoster virus DNA in human sensory ganglia. Nature. 1983;306:478–80. A classic paper that first proved that VZV DNA is latent in human ganglia.
Gilden DH, Gesser R, Smith J, et al. Presence of VZV and HSV-1 DNA in human nodose and celiac ganglia. Virus Genes. 2001;23:145–7.
Mahalingam R, Wellish MC, Dueland AN, Cohrs RJ, Gilden DH. Localization of herpes simplex virus and varicella zoster virus DNA in human ganglia. Ann Neurol. 1992;31:444–8.
Clarke P, Beer T, Cohrs R, Gilden DH. Configuration of latent varicella zoster virus DNA. J Virol. 1995;69:8151–4.
Mahalingam R, Wellish M, Lederer D, et al. Quantitation of latent varicella zoster virus DNA in human trigeminal ganglia by polymerase chain reaction. J Virol. 1993;67:2381–4.
• Cohrs RJ, Randall J, Smith J, et al. Analysis of individual human trigeminal ganglia for latent herpes simplex virus type 1 and varicella zoster virus nucleic acids using real-time PCR. J Virol. 2000;74:11464–71. First real-time PCR analysis of alphaherpesvirus transcription in human ganglia.
Nagel MA, Gilden D, Shade T, Gao B, Cohrs RJ. Rapid and sensitive detection of 68 unique varicella zoster virus gene transcripts in five multiplex reverse transcription-polymerase chain reactions. J Virol Methods. 2009;157:62–8.
Cohrs RJ, Gilden DH. Prevalence and abundance of latently transcribed varicella zoster virus genes in human ganglia. J Virol. 2007;81:2950–6.
Gary L, Gilden DH, Cohrs RJ. Epigenetic regulation of varicella zoster virus open reading frames 62 and 63 in latently infected human trigeminal ganglia. J Virol. 2006;80:4921–6.
Ouwendijk W, Mahalingam R, Traina-Dorge V, et al. Simian varicella virus infection of Chinese rhesus macaques produces ganglionic infection in the absence of rash. J Neurovirol. 2012;18:91–9.
Vafai A, Wellish M, Gilden DH. Expression of varicella zoster virus in blood mononuclear cells of patients with postherpetic neuralgia. Proc Natl Acad Sci USA. 1988;85:2767–70.
Lu PJ, Euler GL, Harpaz R. Herpes zoster vaccination among adults aged 60 years and older in the US 2008. Am J Prev Med. 2011;40:e1–6.
Mahalingam R, Traina-Dorge V, Wellish M, et al. Simian varicella virus reactivation in cynomolgus monkeys. Virology. 2007;368:50–9.
Messaoudi I, Barron A, Wellish M, et al. Simian varicella virus infection of rhesus macaques recapitulates essential features of varicella zoster virus infection in humans. PLoS Pathog. 2009;5:e1000657.
•• Mahalingam R, Traina-Dorge V, Wellish M, et al. Latent simian varicella virus reactivates in monkeys treated with tacrolimus with or without exposure to irradiation. J Neurovirol. 2010;16:342–54. First study revealing that virus reactivates in experimentally immunosuppressed primates latently-infected with SVV.
Kolappaswamy K, Mahalingam R, Traina-Dorge V, et al. Disseminated simian varicella virus infection in an irradiated rhesus macaque (Macaca mulatta). J Virol. 2007;81:411–15.
Ouwendijk WJD, Abendroth A, Traina-Dorge V, et al. T-cell infiltration correlates with CXCL10 expression in ganglia of cynomolgus macaques with reactivated simian varicella virus. J Virol. 2013;87:2979–82.
Gilden D, Mahalingam R, Nagel MA, Pugazhenthi S, Cohrs RJ. The neurobiology of varicella zoster virus infection. Neuropathol Appl Neurobiol. 2011;37:441–63.
Gilden DH, Mahalingam R, Cohrs RJ, Kleinschmidt-DeMasters BK, Forghani B. The protean manifestations of varicella-zoster vasculopathy. J Neurovirol. 2002;8 Suppl 2:75–9.
•• Nagel MA, Choe A, Traktinskiy I, et al. Varicella zoster virus transcriptome in latently infected human ganglia. J Virol. 2011;85:2276–87. State-of-the-art analysis of the VZV transcriptome in latently infected human ganglia.
Acknowledgments
This work was supported by NIH grants AG006127 to DG, AG032958 to DG, RJC and RM, NS082228 to RJC, and NS067070 to MAN.
We thank Marina Hoffman for editorial assistance and Lori DePriest for help with the manuscript preparation.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
All authors report no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Infection
Rights and permissions
About this article
Cite this article
Gilden, D., Nagel, M.A., Cohrs, R.J. et al. The Variegate Neurological Manifestations of Varicella Zoster Virus Infection. Curr Neurol Neurosci Rep 13, 374 (2013). https://doi.org/10.1007/s11910-013-0374-z
Published:
DOI: https://doi.org/10.1007/s11910-013-0374-z