Abstract
This review presents inherited and acquired forms of mitochondrial dysfunction associated with oligodendrocytopathy and neurodegeneration in order to better understand the degenerative features of inflammatory demyelination. The recognition that various mitochondrial mechanisms are involved in the pathogenesis of multiple sclerosis leads to therapeutic considerations, re-emphasizing the importance of early neuroprotection in combination with the approved means of immune modulation.
Similar content being viewed by others
References and Recommended Reading
Trapp BD, Peterson J, Ransohoff RM, et al.: Axonal transection in the lesions of multiple sclerosis. N Engl J Med 1998, 338:278–285.
Bjartmar C, Trapp BD: Axonal and neuronal degeneration in multiple sclerosis: mechanisms and functional consequences. Curr Opin Neurol 2001, 14:271–278.
Husted CA, Goodin DS, Hugg JW, et al.: Biochemical alterations in multiple sclerosis lesions and normalappearing white matter detected by in vivo 31P and 1H spectroscopic imaging. Ann Neurol 1994, 36:157–165.
De Stefano N, Matthews PM, Antel JP, et al.: Chemical pathology of acute demyelinating lesions and its correlation with disability. Ann Neurol 1995, 38:901–909.
Clark JB: N-acetyl aspartate: a marker for neuronal loss or mitochondrial dysfunction. Dev Neurosci 1998, 20:271–276.
DiMauro S, Hirano M: Mitochondrial encephalomyopathies: an update [review]. Neuromusc Disord 2005, 15:276–286.
Zamzami N, Kroemer G: The mitochondrion in apoptosis: how Pandora’s box opens. Nat Rev Mol Cell Biol 2001, 2:67–71.
Schapira LJ: Mitochondrial respiratory chain disorders II: neurodegenerative disorders and nuclear defects. Lancet 2000, 355:389–394.
Kalman B, Leist TP: A mitochondrial component of neurodegeneration in multiple sclerosis. Neuromol Med 2003, 3:147–158.
Anderson S, Bankier AT, Barrell BG, et al.: Sequence and organization of the human mitochondrial genome. Nature 1981, 290:457–465.
Triepels RH, Van Den Heuvel LP, Trijbels JM, Smeitink JA: Respiratory chain complex I deficiency. Am J Med Genet 2001, 106:37–45.
Tiranti V, Hoertnagel K, Carrozzo R, et al.: Mutations of SURF-1 in Leigh disease associated with cytochrome c oxidase deficiency. Am J Hum Genet 1998, 63:1609–1621.
Nishino I, Spinazzola A, Hirano M: Thymidine phosphorylase gene mutations in MNGIE, a human mitochondrial disorder. Science 1999, 283:689–692.
Ogasahara S, Engel AG, Frens D, Mack D: Muscle coenzyme Q deficiency in familial mitochondrial encephalomyopathy. Proc Natl Acad Sci U S A 1989, 86:2379–2382.
Musumeci O, Naini A, Slonim AE, et al.: Familial cerebellar ataxia with muscle coenzyme Q10 deficiency. Neurology 2001, 56:849–855.
Rahman S, Hargreaves I, Clayton P, Heales S: Neonatal presentation of coenzyme q10 deficiency. J Pediatr 2001, 139:456–458.
Vreken P, Valianpour F, Nijtmans LG, et al.: Defective remodeling of cardiolipin and phosphatidylglycerol in Barth syndrome. Biochem Biophys Res Commun 2000, 279:378–382.
Kalman B, Lublin FD, Alder H: Impairment of central and peripheral myelin in mitochondrial diseases. Mult Scler 1997, 2:267–278.
Sadovnick AD, Bulman D, Ebers GC: Parent-child concordance in multiple sclerosis. Ann Neurol 1991, 29:252–255.
Lees F, MacDonald AM, Aldren Turner JW: Leber’s disease with symptoms resembling disseminated sclerosis. J Neurol Neurosurg Psychiatry 1964, 27:415–421.
Wallace DC, Singh G, Lott MT, et al.: Mitochondrial DNA mutation associated with Leber’s hereditary optic neuropathy. Science 1988, 242:1427–1430.
Johns DR, Neufeld MJ, Park RD: An ND-6 mitochondrial DNA mutation associated with Leber hereditary optic neuropathy. Biochem Biophys Res Comm 1992, 187:1551–1557.
Mackey D, Howell N: A variant of Leber hereditary optic neuropathy characterized by recovery of vision and by an unusual mitochondrial genetic etiology. Am J Hum Genet 1992, 51:1218–1228.
Harding AE, Sweeney MG, Miller DH, et al.: Occurrence of a multiple sclerosis-like illness in women who have a Leber’s hereditary optic neuropathy mitochondrial DNA mutation. Brain 1992, 115:979–989.
Flanigan KM, Johns DR: Association of the 11778 mitochondrial DNA mutation and demyelinating disease. Neurology 1993, 43:2720–2722.
Kellar-Wood H, Robertson N, Govan GG, et al.: Leber’s hereditary optic neuropathy mitochondrial DNA mutations in multiple sclerosis. Ann Neurol 1994, 36:109–112.
Horvath R, Abicht A, Shoubridge EA, et al.: Leber’s hereditary optic neuropathy presenting as multiple sclerosis-like disease of the CNS. J Neurol 2000, 247:65–67.
Nishimura M, Obayashi H, Ohta M, et al.: No association of the 11778 mitochondrial DNA mutation and multiple sclerosis in Japan. Neurology 1995, 45:1333–1334.
Kalman B, Lublin FD, Alder H: Mitochondrial DNA mutations in multiple sclerosis. Mult Scler 1995, 1:32–36.
Kalman B, Rodriguez-Valdez JL, Bosch U, Lublin FD: Screening for Leber’s hereditary optic neuropathy associated mitochondrial DNA mutations in patients with prominent optic neuritis. Mult Scler 1997, 2:279–282.
Kalman B, Li S, Chatterjee D, et al.: Large scale screening of the mitochondrial DNA reveals no pathogenic mutations but a haplotype associated with multiple sclerosis in Caucasians. Acta Neurol Scand 1999, 99:16–25.
Hanefeld FA, Ernst BP, Wilichowski E, Christen HJ: Leber’s hereditary optic neuropathy mitochondrial DNA mutations in childhood multiple sclerosis. Neuropediatrics 1994, 25:331.
Kalman B, Lublin FD, Alder H: Characterization of the mitochondrial DNA in patients with multiple sclerosis. J Neurol Sci 1996, 140:75–84.
Kalman B, Mandler RN: Studies of mitochondrial DNA in Devic’s disease revealed no pathogenic mutations, but polymorphisms also found in association with multiple sclerosis. Ann Neurol 2002, 51:661–662.
Brown MD, Sun F, Wallace DC: Clustering of Caucasian Leber hereditary optic neuropathy patients containing the 11,778 or 14,484 mutations on an mtDNA lineage. Am J Hum Genet 1997, 60:381–387.
Qi X, Lewin AS, Hauswirth WW, Guy J: Suppression of complex I gene expression induces optic neuropathy. Ann Neurol 2003, 53:198–205.
Vyshkina T, Banisor I, Shugart YY, et al.: Genetic variants of Complex I in multiple sclerosis. J Neurol Sci 2005, 228:55–64.
Kovacs GG, Hoftberger R, Majtenyi K, et al.: Neuropathology of white matter disease in Leber’s hereditary optic neuropathy. Brain 2005, 128:35–41. This is the first autopsy report of a patient with the T14,484C primary LHON mutation and an MS-like disease. The pathology includes lesions with metabolic (cystic necrosis) and autoimmune (plaque) characteristics.
Baracca A, Solaini G, Sgarbi G, et al.: Severe impairment of Complex I-driven adenosine triphosphate synthesis in Leber Hereditary Optic Neuropathy Cybrids. Arch Neurol 2005, 62:730–736.
Smith PR, Cooper JM, Govan GG, et al.: Antibodies to human optic nerve in Leber’s hereditary optic neuropathy. J Neurol Sci 1995, 130:134–138.
Vladimirova O, O’Connor J, Cahill A, et al.: Oxidative damage to DNA in plaques of MS brains. Mult Scler 1998, 4:413–418.
Cross AH, Manning PT, Stern MK, Misko TP: Evidence for the production of peroxynitrite in inflammatory CNS demyelination. J Neuroimmunol 1997, 80:121–130.
Lu F, Selak M, O’Connor J, et al.: Oxidative damage to mitochondrial DNA and activity of mitochondrial enzymes in chronic active lesions of multiple sclerosis. J Neurol Sci 2000, 177:95–103.
Lucchinetti C, Bruck W, Parisi J, et al.: Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Ann Neurol 2000, 47:707–717.
Aboul-Enein F, Lassmann H: Mitochondrial damage and histotoxic hypoxia: a pathway of tissue injury in inflammatory brain disease? Acta Neuropathol 2005, 109:49–55.
Stadelmann C, Ludwin S, Tabira T, et al.: Tissue preconditioning may explain concentric lesions in Balo’s type of multiple sclerosis. Brain 2005, 128:979–987. This paper elegantly demonstrates that the concentric layering of normal and damaged myelin in Balo’s disease may be related to the upregulation of proteins involved in tissue preconditioning and neuroprotection at the edges of lesions, thereby providing resistance to further damage. At a certain distance, the protection, however, will be overcome by forces promoting inflammation and demyelination.
Andrews HE, Nichols PP, Bates D, Turnbull DM: Mitochondrial dysfunction plays a key role in progressive axonal loss in multiple sclerosis. Med Hypothesis 2005, 64:669–677.
Perieir O, Gregoire A: Electron microscopic features of multiple sclerosis lesions. Brain 1965, 88:937–952.
Griffiths I, Klugmann M, Anderson T, et al.: Axonal swellings and degeneration in mice lacking the major proteolipid of myelin. Science 1998, 280:1610–1613.
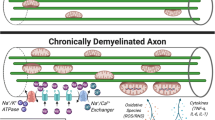
Craner MJ, Newcombe J, Black JA, et al.: Molecular changes in neurons in multiple sclerosis: altered axonal expression of Nav1.2 and Nav1.6 sodium channels and Na+/Ca2+ exchanger. Proc Natl Acad Sci U S A 2004, 101:8168–8173. This study identifies molecular subtypes of voltage-gaited sodium channels in demyelinated and degenerating axons in MS, and reveals the co-expression of Nav1.6 and Na+/Ca2+ exchanger in degenerating axons.
Dutta R, McDonough J, Yin X, et al.: Mitochondrial dysfunction as a cause of axonal degeneration in multiple sclerosis patients. Ann Neurol 2006, 59:478–489.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalman, B. Role of mitochondria in multiple sclerosis. Curr Neurol Neurosci Rep 6, 244–252 (2006). https://doi.org/10.1007/s11910-006-0012-0
Issue Date:
DOI: https://doi.org/10.1007/s11910-006-0012-0