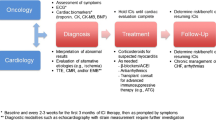
Abstract
Purpose of Review
Chimeric antigen receptor T cell therapy is gaining clinical use in the management of B cell lymphomas. As the use of this unique treatment option increases, its associated toxicities will require recognition and treatment. In this review, we aim to discuss the cardiovascular toxicities of chimeric antigen receptor T cell therapy and our approach to their clinical management.
Recent Findings
Cardiotoxicity may be due to direct or indirect effects of infused chimeric antigen receptor T cells. The cytokine release syndrome has been described extensively in the literature. Studies have also reported cardiovascular dysfunction including hypotension, left ventricular dysfunction, heart failure, and cardiogenic shock in the setting of cytokine release syndrome.
Summary
While there are no standardized guidelines for the treatment of cytokine release syndrome or associated cardiotoxicity, we present our current clinical practices. Further research is indicated into the pathophysiology of therapy-associated cardiac dysfunction and effective management strategies to optimize patient outcomes.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
• Brudno JN, Kochenderfer JN. Toxicities of chimeric antigen receptor T cells: recognition and management. Blood. 2016;127(26):3321–30. https://doi.org/10.1182/blood-2016-04-703751. The authors at City of Hope National Medical Center base many clinical practices on supportive care guidelines published by the National Cancer Institute Experimental Transplantation and Immunology Branch.
Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR, et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med. 2013;368(16):1509–18. https://doi.org/10.1056/NEJMoa1215134.
Linette GP, Stadtmauer EA, Maus MV, Rapoport AP, Levine BL, Emery L, et al. Cardiovascular toxicity and titin cross-reactivity of affinity-enhanced T cells in myeloma and melanoma. Blood. 2013;122:863–71. https://doi.org/10.1182/blood-2013-03-490,565.
Burstein DS, Maude S, Grupp S, Griffis H, Rossano J, Lin K. Cardiac profile of chimeric antigen receptor T cell therapy in children: a single-institution experience. Biol Blood and Marrow Transpl. 2018;24(8):1590–5. https://doi.org/10.1016/j.bbmt.2018.05.014.
Pathan N, Hemingway CA, Alizadeh AA, Stephens AC, Boldrick JC, Oragui EE, et al. Role of interleukin 6 in myocardial dysfunction of meningococcal septic shock. Lancet. 2004;363:203–9. https://doi.org/10.1016/S0140-6736(03)15326-3.
Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA, et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 2015;385(9967):517–28. https://doi.org/10.1016/S0140-6736(14)61403-3.
• Ganatra S, Carver JR, Hayek SS, Ky B, Leja MJ, Lenihan DJ, et al. Chimeric antigen receptor T-cell therapy for cancer and heart: JACC Council Perspectives. J Am Coll Cardiol. 2019;74(25):3153–63. https://doi.org/10.1016/j.jacc.2019.10.049. The authors have incorporated expert consensus perspectives from the Cardio-Oncology Section of the American College of Cardiology into clinical practice when managing patients undergoing chimeric antigen receptor T cell therapy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on T-Cell and Other Lymphoproliferative Malignancies
Rights and permissions
About this article
Cite this article
Jamal, F.A., Khaled, S.K. The Cardiovascular Complications of Chimeric Antigen Receptor T Cell Therapy. Curr Hematol Malig Rep 15, 130–132 (2020). https://doi.org/10.1007/s11899-020-00567-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-020-00567-4