Abstract
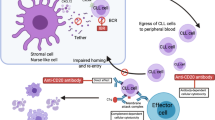
Ibrutinib is a potent covalent kinase inhibitor that targets BTK. BTK, or Bruton’s tyrosine kinase, is an obvious target for therapy of B cell diseases because inactivating mutations lead to B cell aplasia in humans and the disease X-linked agammaglobulinemia. Ibrutinib has modest cytotoxicity against CLL cells in vitro but also blocks trophic stimuli from the microenvironment. As with other inhibitors of the BCR pathway, ibrutinib causes rapid nodal reduction and response associated with rapid increase in lymphocytosis, which then returns to baseline over time. The ORR of ibrutinib in relapsed refractory CLL is 67 % with PFS 88 % at 15 months. In a cohort of untreated patients 65 years and over, the estimated 15 month PFS is 96 %. Registration trials have been initiated, and the difficult task that remains is to determine where in the course of CLL therapy this drug will have the greatest impact and benefit for patients.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Abrisqueta P, Pereira A, Rozman C, et al. Improving survival in patients with chronic lymphocytic leukemia (1980–2008): the Hospital Clinic of Barcelona experience. Blood. 2009;114(10):2044–50.
Byrd JC, Rai K, Peterson BL, et al. Addition of rituximab to fludarabine may prolong progression-free survival and overall survival in patients with previously untreated chronic lymphocytic leukemia: an updated retrospective comparative analysis of CALGB 9712 and CALGB 9011. Blood. 2005;105(1):49–53.
•• Hallek, M., Fischer, K., Fingerle-Rowson, G., et al.: Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet 2010, 376(9747): 1164–74. This study has established the standard of care therapy for previously untreated CLL patients.
Keating MJ, O’Brien S, Albitar M, et al. Early results of a chemoimmunotherapy regimen of fludarabine, cyclophosphamide, and rituximab as initial therapy for chronic lymphocytic leukemia. J Clin Oncol. 2005;23(18):4079–88.
Brown JR. The treatment of relapsed refractory chronic lymphocytic leukemia. Hematology Am Soc Hematol Educ Program. 2011;1:110–118.
• Davids, MS and Brown JR: Targeting the B Cell Receptor Pathway in Chronic Lymphocytic Leukemia. Leuk Lymphoma. 2012;53(12):2362–70. This review covers the range of inhibitors of the BCR pathway which have been studied in CLL thus far, and discusses ongoing issues with their development.
Hendriks RW, Bredius RG, Pike-Overzet K, Staal FJ. Biology and novel treatment options for XLA, the most common monogenetic immunodeficiency in man. Expert Opin Ther Targets. 2011;15(8):1003–21.
Wiestner, A.: Emerging role of kinase targeted strategies in chronic lymphocytic leukemia. Blood. 2012;120(24):4684–91.
Honigberg LA, Smith AM, Sirisawad M, et al. The Bruton tyrosine kinase inhibitor PCI-32765 blocks B-cell activation and is efficacious in models of autoimmune disease and B-cell malignancy. Proc Natl Acad Sci USA. 2010;107(29):13075–80.
Herman SE, Gordon AL, Hertlein E, et al. Bruton tyrosine kinase represents a promising therapeutic target for treatment of chronic lymphocytic leukemia and is effectively targeted by PCI-32765. Blood. 2011;117(23):6287–96.
de Rooij MF, Kuil A, Geest CR, et al. The clinically active BTK inhibitor PCI-32765 targets B-cell receptor- and chemokine-controlled adhesion and migration in chronic lymphocytic leukemia. Blood. 2012;119(11):2590–4.
Ponader S, Chen SS, Buggy JJ, et al.: Bruton’s tyrosine kinase inhibitor PCI-32765 thwarts chronic lymphocytic leukemia cell survival and tissue homing in vitro and in vivo. Blood. 2012;119(5):1182–1189.
Advani RH, Buggy JJ, Sharman JP, et al. The BTK Inhibitor PCI-32765 is highly active and well tolerated in patients (pts) with relapsed/refractory B cell malignancies: Final Results from a Phase 1 Study. J Clin Oncol. doi:10.1200/JCO.2012.44.4281
Burger JA, O’Brien S, Fowler N, et al. The Bruton’s Tyrosine Kinase Inhibitor, PCI-32765, is well tolerated and demonstrates promising clinical activity in chronic lymphocytic leukemia (CLL) and Small Lymphocytic Lymphoma (SLL): an update on ongoing phase 1 studies. ASH Annual Meeting Abstracts. 2010;116(21):57.
Hallek M, Cheson BD, Catovsky D, et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood. 2008;111(12):5446–56.
• Cheson BD, Byrd JC, Rai KR, et al.: Novel targeted agents and the need to refine clinical end points in chronic lymphocytic leukemia. J Clin Oncol 2012, 30(23):2820–2. This article discusses the adaptation to the CLL response criteria which is required for the evaluation of the BCR pathway inhibitors.
O’Brien S, Burger J, Blum K, et al. The Bruton’s Tyrosine Kinase (BTK) Inhibitor PCI-32765 Induces Durable Responses in Relapsed or Refractory (R/R) Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (CLL/SLL): follow-up of a phase Ib/II study. ASH Annual Meeting Abstracts. 2011;118(21):983.
Byrd JC, Furman R, Coutre S, et al. The Bruton’s tyrosine kinase (BTK) inhibitor PCI-32765 (P) in treatment-naive (TN) chronic lymphocytic leukemia (CLL) patients (pts): interim results of a phase Ib/II study. ASCO Meeting Abstracts. 2012;30(15_suppl):6507.
Wierda WG, et al. Ofatumumab as single-agent CD20 immunotherapy in fludarabine-refractory chronic lymphocytic leukemia. J Clin Oncol. 2010;28(10):1749–55.
Wierda WG, Kipps TJ, Mayer J, et al. Ofatumumab is active in patients with fludarabine-refractory CLL irrespective of prior rituximab: results from the phase 2 international study. Blood. 2011;118(19):5126–9.
Jaglowski SM, Jones J, Flynn J, et al. A phase Ib/II study evaluating activity and tolerability of BTK inhibitor PCI-32765 and ofatumumab in patients with chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) and related diseases. ASCO Meeting Abstracts. 2012;30(15_suppl):6508.
O’Brien SM, Barrientos J, Flinn I, et al. Combination of the Bruton’s tyrosine kinase (BTK) inhibitor PCI-32765 with bendamustine (B)/rituximab (R) (BR) in patients (pts) with relapsed/refractory (R/R) chronic lymphocytic leukemia (CLL): Interim results of a phase Ib/II study. ASCO Meeting Abstracts. 2012;30(15_suppl):6515.
Brown JR, Barrientos J, Flinn I, et al.: The Bruton’s Tyrosine Kinase (BTK) inhibitor ibrutinib combined with bendamustine and rituximab is active and tolerable in patients with relapsed/refractory CLL, interim results of a phase Ib/II study. EHA Meeting Abstracts 2012, 97.
Fischer K, Cramer P, Busch R, et al. Bendamustine combined with rituximab in patients with relapsed and/or refractory chronic lymphocytic leukemia: a multicenter phase II trial of the German Chronic Lymphocytic Leukemia Study Group. J Clin Oncol. 2011;29(26):3559–66.
Disclosure
J. Brown: consultant for Avila, Novartis, Emergent, Celgene, Genentech, Pharmacyclics, Sanofi; received funding for investigator initiated clinical trial from Genzyme (now Sanofi); travel/accommodation expenses covered or reimbursed by Pharmacyclics.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brown, J.R. Ibrutinib (PCI-32765), the First BTK (Bruton’s Tyrosine Kinase) Inhibitor in Clinical Trials. Curr Hematol Malig Rep 8, 1–6 (2013). https://doi.org/10.1007/s11899-012-0147-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-012-0147-9