Abstract
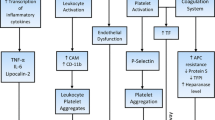
The clinical course of the classic myeloproliferative neoplasms (MPNs) is burdened by an increased rate of cardiovascular events, which are the major cause of mortality. Age and history of thrombosis are the criteria used to stratify patients to the most appropriate therapeutic options. However, the mechanisms ultimately responsible for the increased thrombotic tendency have not yet been elucidated; abnormalities of blood cell count, neutrophil and platelet activation, and a state of hypercoagulability can all occur. Recurrent mutations in JAK2 or MPL have been described in MPNs and serve as disease markers. There is also evidence that a JAK2V617F mutant state represents an independent factor associated with thrombosis, and abnormalities of cell function attributable to JAK2V617F have been characterized. It is hoped that elucidation of the role mutant JAK2 plays in MPNs will improve our understanding of the pathophysiology of thrombosis and eventually result in improved patient treatment using molecularly targeted drugs.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance•• Of major importance
Vannucchi AM, Guglielmelli P, Tefferi A: Advances in understanding and management of myeloproliferative neoplasms. CA Cancer J Clin 2009, 59:171–191.
•• Tefferi A, Thiele J, Orazi A, et al.: Proposals and rationale for revision of the World Health Organization diagnostic criteria for polycythemia vera, essential thrombocythemia, and primary myelofibrosis: recommendations from an ad hoc international expert panel. Blood 2007, 110:1092–1097. The myeloproliferative disorders have been renamed as “myeloproliferative neoplasms” to better signify their neoplastic nature, and the presence of JAK2 or MPL mutation is included as a major diagnostic criterion. This study prompted the final draft of the 2008 World Health Organization Classification of Tumors of Hematopoietic and Lymphoid Tissues.
Tefferi A: The history of myeloproliferative disorders: before and after Dameshek. Leukemia 2008, 22:3–13.
Rollison DE, Howlader N, Smith MT, et al.: Epidemiology of myelodysplastic syndromes and chronic myeloproliferative disorders in the United States, 2001–2004: utilizing data from the NAACCR and SEER Programs. Blood 2008, 112:45–52.
Campbell PJ: Somatic and germline genetics at the JAK2 locus. Nat Genet 2009, 41:385–386.
Vainchenker W, Constantinescu SN: A unique activating mutation in JAK2 (V617F) is at the origin of polycythemia vera and allows a new classification of myeloproliferative diseases. Hematology Am Soc Hematol Educ Program 2005, 195–200.
Levine RL, Pardanani A, Tefferi A, Gilliland DG: Role of JAK2 in the pathogenesis and therapy of myeloproliferative disorders. Nat Rev Cancer 2007, 7:673–683.
Kralovics R, Passamonti F, Buser AS, et al.: A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 2005, 352:1779–1790.
Vannucchi AM, Antonioli E, Guglielmelli P, et al.: Clinical correlates of JAK2V617F presence or allele burden in myeloproliferative neoplasms: a critical reappraisal. Leukemia 2008, 22:1299–1307.
Scott LM, Tong W, Levine RL, et al.: JAK2 exon 12 mutations in polycythemia vera and idiopathic erythrocytosis. N Engl J Med 2007, 356:459–468.
Pikman Y, Lee BH, Mercher T, et al.: MPLW515L is a novel somatic activating mutation in myelofibrosis with myeloid metaplasia. PLoS Med 2006, 3:e270.
Campbell PJ, Green AR: The myeloproliferative disorders. N Engl J Med 2006, 355:2452–2466.
•• Delhommeau F, Dupont S, Della Valle V, et al.: Mutation in TET2 in myeloid cancers. N Engl J Med 2009, 360:2289–2301. Deletions or mutations in TET2, a novel candidate tumor-suppressor gene, were identified in 19% of patients with myelodysplastic syndromes, 12% with myeloproliferative disorders, and 24% with acute myeloid leukemia. TET2 mutations were found in hematopoietic stem cells and preceded the JAK2V617F mutation, pointing to TET2 as a pre-JAK2 molecular lesion in myeloproliferative disorders.
Vannucchi AM, Guglielmelli P: Molecular pathophysiology of Philadelphia-negative myeloproliferative disorders: beyond JAK2 and MPL mutations. Haematologica 2008, 93:972–976.
Cervantes F, Passamonti F, Barosi G: Life expectancy and prognostic factors in the classic BCR/ABL-negative myeloproliferative disorders. Leukemia 2008, 22:905–914.
•• Cervantes F, Dupriez B, Pereira A, et al.: New prognostic scoring system for primary myelofibrosis based on a study of the International Working Group for Myelofibrosis Research and Treatment. Blood 2009, 113:2895–2901. In this international multicenter study, 1054 patients with PMF were studied with the aim of identifying clinical and hematologic variables associated with survival. Older age, constitutional symptoms, lower hemoglobin, higher leukocyte count, and circulating blast cells (≥ 1%) were identified in multivariate analysis as predictors of shortened survival. By using these variables, four classes of patients with no overlapping in their survival curves were delineated. This scoring system will be pivotal in therapeutic decisions and for inclusion of PMF patients in clinical trials.
Cortelazzo S, Finazzi G, Ruggeri M, et al.: Hydroxyurea for patients with essential thrombocythemia and a high risk of thrombosis. N Engl J Med 1995, 332:1132–1136.
Landolfi R, Marchioli R, Kutti J, et al.: Efficacy and safety of low-dose aspirin in polycythemia vera. N Engl J Med 2004, 350:114–124.
Marchioli R, Finazzi G, Landolfi R, et al.: Vascular and neoplastic risk in a large cohort of patients with polycythemia vera. J Clin Oncol 2005, 23:2224–2232.
Harrison CN, Campbell PJ, Buck G, et al.: Hydroxyurea compared with anagrelide in high-risk essential thrombocythemia. N Engl J Med 2005, 353:33–45.
Cervantes F, Alvarez-Larran A, Arellano-Rodrigo E, et al.: Frequency and risk factors for thrombosis in idiopathic myelofibrosis: analysis in a series of 155 patients from a single institution. Leukemia 2006, 20:55–60.
• De Stefano V, Za T, Rossi E, et al.: Recurrent thrombosis in patients with polycythemia vera and essential thrombocythemia: incidence, risk factors, and effect of treatments. Haematologica 2008, 93:372–380. The rate of recurrence was estimated in a multicenter cohort of 494 patients with ET or PV with previous arterial thrombosis (67.6%), venous thrombosis (31%), or both (1.4%). Thrombosis recurred in 166 patients (33.6%), with an incidence of 7.6% per patient-year. Age greater than 60 years and increased leukocyte count at the time of first thrombosis in patients less than 60 years old predicted for recurrence. Cytoreduction halved the risk in the overall cohort, and a combination regimen with antiplatelet agents or oral anticoagulants proved more effective than administration of a single drug.
• Kiladjian JJ, Cervantes F, Leebeek FW, et al.: The impact of JAK2 and MPL mutations on diagnosis and prognosis of splanchnic vein thrombosis: a report on 241 cases. Blood 2008, 111:4922–4929. The prognostic value of JAK2 and MPL515 mutations was assessed in 241 patients with SVT. JAK2V617F was found in almost all patients with bone marrow or progenitor-cell growth abnormalities specific for a myeloproliferative disorder but also in more than half of those with only one feature and in 7% of those with neither feature. This paper supports the utility of routine JAK2 genotyping in patients with SVT, even in the absence of clinical suspicion of MPN.
Finazzi G, Barbui T: Risk-adapted therapy in essential thrombocythemia and polycythemia vera. Blood Rev 2005, 19:243–252.
Marchetti M, Falanga A: Leukocytosis, JAK2V617F mutation, and hemostasis in myeloproliferative disorders. Pathophysiol Haemost Thromb 2008, 36:148–159.
Di Nisio M, Barbui T, Di Gennaro L, et al.: The haematocrit and platelet target in polycythemia vera. Br J Haematol 2007, 136:249–259.
Pearson TC, Wetherley-Mein G: Vascular occlusive episodes and venous haematocrit in primary proliferative polycythaemia. Lancet 1978, 2:1219–1222.
Tefferi A, Gangat N, Wolanskyj AP: Management of extreme thrombocytosis in otherwise low-risk essential thrombocythemia; does number matter? [letter]. Blood 2006, 108:2493–2494.
•• Barbui T, Carobbio A, Rambaldi A, Finazzi G: Perspectives on thrombosis in essential thrombocythemia and polycythemia vera: Is leukocytosis a causative factor? Blood 2009, 114:759–763. This illuminating critical reappraisal of recent data and of pathophysiologic mechanisms supports the relevance of leukocytosis as a risk factor for thrombosis in MPNs.
Carobbio A, Finazzi G, Antonioli E, et al.: Thrombocytosis and leukocytosis interaction in vascular complications of essential thrombocythemia. Blood 2008, 112:3135–3137.
Falanga A, Marchetti M, Evangelista V, et al.: Polymorphonuclear leukocyte activation and hemostasis in patients with essential thrombocythemia and polycythemia vera. Blood 2000, 96:4261–4266.
• Marchetti M, Castoldi E, Spronk HM, et al.: Thrombin generation and activated protein C resistance in patients with essential thrombocythemia and polycythemia vera. Blood 2008, 112:4061–4068. The aim of this study was to define the relationships of a hypercoagulable state with JAK2V617F mutational status in ET and PV. The occurrence of activated protein C (APC) resistance was demonstrated to correlate with a JAK2V617F mutant genotype and the burden of mutant allele. APC resistance was probably due to a reduction in free protein-S levels. These data indicate that the APC-resistant phenotype is influenced by the JAK2V617F mutational load.
Antonioli E, Guglielmelli P, Pancrazzi A, et al.: Clinical implications of the JAK2 V617F mutation in essential thrombocythemia. Leukemia 2005, 19:1847–1849.
Antonioli E, Guglielmelli P, Poli G, et al.: Influence of JAK2V617F allele burden on phenotype in essential thrombocythemia. Haematologica 2008, 93:41–48.
Wolanskyj AP, Lasho TL, Schwager SM, et al.: JAK2 mutation in essential thrombocythaemia: clinical associations and long-term prognostic relevance. Br J Haematol 2005, 131:208–213.
• Vannucchi AM, Antonioli E, Guglielmelli P, et al.: Clinical profile of homozygous JAK2V617F mutation in patients with polycythemia vera or essential thrombocythemia. Blood 2007, 110:840–846. To evaluate the clinical relevance of a homozygous state of the JAK2V617F mutation, 118 homozygous patients (104 PV, 14 ET) were identified in a retrospective, multicentric study. JAK2V617F homozygosity was found to be associated with a distinct clinical phenotype and more frequent evolution into secondary myelofibrosis in PV and ET. In a multivariate analysis, homozygous ET patients displayed a significantly higher risk of cardiovascular events.
Vannucchi AM, Antonioli E, Guglielmelli P, et al.: Prospective identification of high-risk polycythemia vera patients based on JAK2(V617F) allele burden. Leukemia 2007, 21:1952–1959.
Ziakas PD: Effect of JAK2 V617F on thrombotic risk in patients with essential thrombocythemia: measuring the uncertain. Haematologica 2008, 93:1412–1414.
Dahabreh IJ, Zoi K, Giannouli S, et al.: Is JAK2 V617F mutation more than a diagnostic index? A meta-analysis of clinical outcomes in essential thrombocythemia. Leuk Res 2008, 33:67–73.
Lussana F, Caberlon S, Pagani C, et al.: Association of V617F Jak2 mutation with the risk of thrombosis among patients with essential thrombocythaemia or idiopathic myelofibrosis: a systematic review. Thromb Res 2009, 124:409–417.
De Stefano V, Za T, Rossi E, et al.: Increased risk of recurrent thrombosis in patients with essential thrombocythemia carrying the homozygous JAK2 V617F mutation. Ann Hematol 2009 Jul 7 (Epub ahead of print).
De Stefano V, Fiorini A, Rossi E, et al.: Incidence of the JAK2 V617F mutation among patients with splanchnic or cerebral venous thrombosis and without overt chronic myeloproliferative disorders. J Thromb Haemost 2007, 5:708–714.
Dentali F, Squizzato A, Brivio L, et al.: JAK2V617F mutation for the early diagnosis of Ph- myeloproliferative neoplasms in patients with venous thromboembolism: a meta-analysis. Blood 2009, 113:5617–5623.
Griesshammer M, Struve S, Barbui T: Management of Philadelphia negative chronic myeloproliferative disorders in pregnancy. Blood Rev 2008, 22:235–245.
Passamonti F, Randi ML, Rumi E, et al.: Increased risk of pregnancy complications in patients with essential thrombocythemia carrying the JAK2 (617 V > F) mutation. Blood 2007, 110:485–489.
Mercier E, Lissalde-Lavigne G, Gris JC: JAK2 V617F mutation in unexplained loss of first pregnancy. N Engl J Med 2007, 357:1984–1985.
Gangat N, Wolanskyj AP, Schwager S, Tefferi A: Predictors of pregnancy outcome in essential thrombocythemia: a single institution study of 63 pregnancies. Eur J Haematol 2009, 82:350–353.
Dahabreh IJ, Jones AV, Voulgarelis M, et al.: No evidence for increased prevalence of JAK2 V617F in women with a history of recurrent miscarriage. Br J Haematol 2009, 144:802–803.
Falanga A, Marchetti M, Vignoli A, et al.: V617F JAK-2 mutation in patients with essential thrombocythemia: relation to platelet, granulocyte, and plasma hemostatic and inflammatory molecules. Exp Hematol 2007, 35:702–711.
Pardanani A: JAK2 inhibitor therapy in myeloproliferative disorders: rationale, preclinical studies and ongoing clinical trials. Leukemia 2008, 22:23–30.
Acknowledgments
This work was supported by Istituto Toscano Tumori. The author apologizes to his many eminent colleagues whose work in the field could not be cited because of space limitations.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vannucchi, A.M. JAK2 Mutation and Thrombosis in the Myeloproliferative Neoplasms. Curr Hematol Malig Rep 5, 22–28 (2010). https://doi.org/10.1007/s11899-009-0038-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-009-0038-x