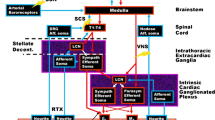
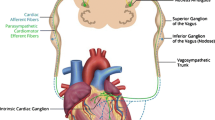
Abstract
Heart failure is the final common pathway in many forms of heart disease, and is associated with excessive morbidity and mortality. Pathophysiologic alterations in the interaction between the heart and the autonomic nervous system in advanced heart failure have been noted for decades. Over the last decade, great advances have been made in the medical and surgical treatment of heart failure – and some of these modalities target the neuro-cardiac axis. Despite these advances, many patients progress to end-stage heart failure and death. Recently, device-based therapy targeting the neuro-cardiac axis with various forms of neuromodulatory stimuli has been shown to improve heart function in experimental heart failure models. These include spinal cord stimulation, vagal nerve stimulation, and baroreflex modulation. Human trials are now underway to evaluate the safety and efficacy of these device-based neuromodulatory modalities in the heart failure population.



Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lloyd-Jones D, Adams RJ, Brown TM, et al. Heart disease and stroke statistics-2010 update. A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2010;121:e1–170.
Cohn JN, Levine TB, Olivari MT, et al. Plasma norepinephrine as a guide to prognosis in patients with chronic congestive heart failure. N Engl J Med. 1984;311:819–23.
Pepper GS, Lee RW. Sympathetic activation in heart failure and its treatment with beta-blockade. Arch Intern Med. 1999;159:225–34.
Regitz V, Leuchs B, Bossaller C, et al. Myocardial catecholamine concentrations in dilated cardiomyopathy and heart failure of different origins. Eur Heart J. 1991;12(Suppl):S171–4.
Piacentino III V, Weber CR, Chen X, et al. Cellular basis of abnormal calcium transients of failing human ventricular myocytes. Circ Res. 2003;92:651–8.
Olivetti G, Abbi R, Quaini F, et al. Apoptosis in the failing human heart. N Engl J Med. 1997;336:1131–41.
Communal C, Singh K, Pimentel DR, et al. Norepinephrine stimulates apoptosis in adult rat ventricular myocytes by activation of the beta-adrenergic pathway. Circulation. 1998;98:1329–34.
Watson AM, Hood SG, May CN. Mechanisms of sympathetic activation in heart failure. Clin Exp Pharmacol Physiol. 2006;33:1269–74.
Boogers MJ, Borleffs CJ, Henneman MM, et al. Cardiac sympathetic denervation assessed with 123-iodine metaiodobenzylguanidine imaging predicts ventricular arrhythmias in implantable cardioverter-defibrillator patients. J Am Coll Cardiol. 2010;55:2769–77.
Fishman GI, Chugh SS, DiMarco J, et al. Sudden cardiac death prediction and prevention: report from a national heart, lung, and blood institute and heart rhythm society workshop. Circulation. 2010;122:2335–48.
Newton GE, Parker AB, Landzberg JS, et al. Muscarinic receptor modulation of basal and beta-adrenergic stimulated function of the failing human left ventricle. J Clin Invest. 1996;98:2756–63.
Fox K, Borer JS, Camm AJ, et al. Resting heart rate in cardiovascular disease. J Am Coll Cardiol. 2007;50:823–30.
Kinugawa T, Dibner-Dunlap ME. Altered vagal and sympathetic control of heart rate in left ventricular dysfunction and heart failure. Am J Physiol. 1995;268:R317–23.
Damy T, Ratajczak P, Shah AM, et al. Increased neuronal nitric oxide synthase-derived no production in the failing human heart. Lancet. 2004;363:1365–7.
Tracey KJ. Physiology and immunology of the cholinergic anti-inflammatory pathway. J Clin Invest. 2007;117:289–96.
Dargie HJ, Lechat P. The cardiac insufficiency bisoprolol study (CIBIS-II): a randomized trial. Lancet. 1999;353:9–13.
Merit-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure: metoprolol CR/XL randomized intervention trial in congestive heart failure (MERIT-HF). Lancet. 1999;353:2001–7.
Eichorn E, Domanski M, Krause-Steinrauf H, Anderson J, Boardman K, Bristow M. The beta-blocker evaluation of survival trial investigators. A trial of the beta–blocker bucindolol in patients with advanced chronic heart failure. N Engl J Med. 2001;344:1659–67.
Packer M, Bristow MR, Cohn JN, et al. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. N Engl J Med. 1996;334:1349–55.
Packer M, Coats AJ, Fowler MB, et al. Effect on carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344:1651–8.
Cohn JN, Archibald DG, Ziesche S, et al. Effect of vasodilator therapy on mortality in chronic congestive heart failure. results of a Veterans Administration cooperative study. N Engl J Med. 1986;314:1547–52.
ALLHAT Collaborative Research Group. Major cardiovascular events in hypertensive patients randomized to doxazosin vs chlorthalidone: the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). JAMA. 2000;283:1967–75.
Giles TD, Thomas MG, Quiroz A, et al. Acute and short-term effects of clonidine in heart failure. Angiology. 1987;38:537–48.
Issa ZF, Ujhelyi MR, Hildebrand KR, et al. Intrathecal clonidine reduces the incidence of ischemia-provoked ventricular arrhythmias in a canine post infarction heart failure model. Heart Rhythm. 2005;2:1122–7.
Cohn JN, Pfeffer MA, Rouleau J, et al. on behalf of the MOXCON Investigators. Adverse mortality effect of central sympathetic inhibition with sustained–release moxonidine in patients with heart failure (MOXCON). Eur J Heart Fail. 2003;5:659–67.
Mahajan A, Moore J, Cesario DA. Use of thoracic epidural anesthesia for management of electrical storm: a case report. Heart Rhythm. 2005;2:1359–62.
Zipes DP, Festoff B, Schaal SF, et al. Treatment of ventricular arrhythmia by permanent atrial pacemaker and cardiac sympathectomy. Ann Intern Med. 1968;68:591–7.
Wilde AA, Bhuiyan A, Crotti L, et al. Left cardiac sympathetic denervation for catecholaminergic polymorphic ventricular tachycardia. N Engl J Med. 2008;358:2024–9.
Collura CA, Johnson JN, Moir C, et al. Left cardiac sympathetic denervation for the treatment of long QT syndrome and catecholaminergic polymorphic ventricular tachycardia using video-assisted thoracic surgery. Heart Rhythm. 2009;6:752–9.
Behling A, Moraes RS, Rohde LE, et al. Cholinergic stimulation with pyridostigmine reduces ventricular arrhythmias and enhances heart rate variability in heart failure. Am Heart J. 2003;146:494–500.
Binkley PF, Haas GS, Starling RC, et al. Sustained augmentation of parasympathetic tone with angiotensin–converting enzyme inhibition in patients with congestive heart failure. J Am Coll Cardiol. 1993;21:655–61.
Foreman R, Linderoth B, Ardelt J, et al. Modulation of intrinsic cardiac neurons by spinal cord stimulation: implications for its therapeutic use in angina pectoris. Cardiovasc Res. 2000;47:367–75.
Southerland EM, Milhorn DM, Foreman RD, et al. Preemptive, but not reactive, spinal cord stimulation mitigates transient ischemia-induced myocardial infarction via cardiac adrenergic neurons. Am J Physiol Heart Circ Physiol. 2007;292:H311–7.
• JE Olgin, Takahashi T, Wilson E, et al. Effects of thoracic spinal cord stimulation on cardiac autonomic regulation of the sinus and atrioventricular nodes. J Cardiovasc Electrophysiol. 2002;13:475–81. This work provided evidence of the vagomimetic actions of SCS.
•• Issa ZF, Zhou X, Ujhelyi MR, et al. Thoracic spinal cord stimulation reduces the risk of ischemic ventricular arrhythmias in a post-infarction heart failure canine model. Circulation. 2005;111:3217–20. This work provided evidence that SCS can reduce ischemic ventricular arrhythmias.
Odenstedt J, Linderoth B, Bergfeldt L, et al. Spinal cord stimulation effects on myocardial ischemia, infarct size, ventricular arrhythmia, and noninvasive electrophysiology in a porcine ischemia-reperfusion model. Heart Rhythm. 2011;8:892–8.
•• Lopshire JC, Zhou X, Dusa C, et al. Spinal cord stimulation improves ventricular function and reduces ventricular arrhythmias in a canine postinfarction heart failure model. Circulation. 2009;120:286–94. This work demonstrated that chronic SCS can improve LV function in heart failure.
Lopshire JC, Zhou X, Mullen T, et al. The site and intensity of spinal cord stimulation determines remodeling response in canine heart failure. Hear Rhythm. 2011;8:S78.
Liu Y, Yue WS, Liao SY, et al. Thoracic spinal cord stimulation improves cardiac contractile function and myocardial oxygen consumption in a porcine model of ischemic heart failure. J Cardiovasc Electrophysiol. 2012;23(5):534-540.
Vanoli E, De Ferrari GM, Stramba-Badiale M, et al. Vagal stimulation and prevention of sudden death in conscious dogs with a healed myocardial infarction. Circ Res. 1991;68:1471–81.
Li M, Zheng C, Sato T, Kawada T, et al. Vagal nerve stimulation markedly improves long-term survival after chronic heart failure in rats. Circulation. 2004;109:120–4.
Zheng C, Li M, Inagaki M, Kawada T, et al. Vagal stimulation markedly suppresses arrhythmias in conscious rats with chronic heart failure after myocardial infarction. Conf Proc IEEE Eng Med Biol Soc. 2004;7:7072–5.
•• Zhang Y, Popovic ZB, Bibevski S, et al. Chronic vagus nerve stimulation improves autonomic control and attenuates systemic inflammation and heart failure progression in a canine high–rate pacing model. Circulation. 2009;2:692–9. This work demonstrated that VNS can improve LV function.
• Sabbah HN, Ilsar I, Zaretsky A, et al. Vagus nerve stimulation in experimental heart failure. Heart Fail Rev. 2011;16:171–8. This work utilized a novel VNS system in HF and found improved LV function with therapy.
Filippone JD, Bisognano JD. Baroreflex stimulation in the treatment of hypertension. Curr Opin Nephrol Hypertens. 2007;16:403–8.
Heusser K, Tank J, Engeli S, et al. Carotid baroreceptor stimulation, sympathetic activity, baroreflex function, and blood pressure in hypertensive patients. Hypertension. 2010;55:619–26.
•• Sabbah HN, Gupta RC, Imai M, et al. Chronic electrical stimulation of the carotid sinus baroreflex improves left ventricular function and promotes reversal of ventricular remodeling in dogs with advanced heart failure. Circ Heart Fail. 2011;4(1):65–70. This work demonstrated a therapeutic benefit of baroreflex stimulation in experimental heart failure.
•• Determining the feasibility of spinal cord neuromodulation for the treatment of chronic heart failure (defeat-HF). Available at: http://clinicaltrials.gov/ct2/show/NCT01112579. This work is the first large human trial of SCS in heart failure patients.. Accessed 1 Apr 2012.
Cornelussen RN, Splett V, Klepfer RN, et al. Electrical modalities beyond pacing for the treatment of heart failure. Heart Fail Rev. 2011;16:315–25.
Spinal cord stimulation for heart failure (SCS HEART). Available at: http://clinicaltrials.gov/ct2/show/NCT01362725. Accessed 1 Apr 2012.
Neurostimulation of spinal nerves that affect the heart. Available at: http://clinicaltrials.gov/ct2/show/NCT01124136. Accessed 1 Apr 2012.
De Ferrari GM, Ezra OB, Ajmone-Marsan N, et al. Chronic vagal stimulation in patients with heart failure is feasible, safe and appears beneficial. Eur Heart J. 2006;27:330–7.
•• Schwartz PJ, Gaetano M, De Ferrari GM. Vagal stimulation for heart failure: Background and first in–man study. Heart Rhythm. 2009;6(11):S76–81. This study was the first report on the use of vagal stimulation in heart failure patients.
CardioFit™ for the treatment of heart failure. Available at: http://clinicaltrials.gov/ct2/show/NCT00461019. Accessed 1 Apr 2012.
•• De Ferrari GM, Crijns HJ, Borggrefe M, et al. Chronic vagus nerve stimulation; a new and promising therapeutic approach for chronic heart failure. Eur Heart J. 2011;32(7):847–55. This paper reported on significant early trials of vagus nerve stimulation in heart failure.
Increase of vagal tone in chf (inovate–hf). Available at: http://clinicaltrials.gov/ct2/show/NCT01303718. Accessed 1 Apr 2012.
Neural cardiac therapy for heart failure study (NECTAR–HF). Available at: http://clinicaltrials.gov/ct2/show/NCT01385176. Accessed 1 Apr 2012.
Rheos® diastolic heart failure trial. Available at: http://clinicaltrials.gov/ct2/show/NCT00718939. Accessed 1 Apr 2012.
Lovett EG, Schafer J, Kaufman CL. Chronic baroreflex activation by the Rheos system: an overview of results from European and North American feasibility studies. Proceedings of the Engineering in Medicine and Biology Society, 2009 Annual International Conference of the IEEE, pp 4626–30.
Baroreflex activation therapy in heart failure. Available at: http://clinicaltrials.gov/ct2/show/NCT01484288. Accessed 1 Apr 2012.
Georgakopoulos D, Little WC, Abraham WT, et al. Chronic baroreflex activation: a potential therapeutic approach to treat heart failure with preserved ejection fraction. J Card Fail. 2011;17:167–78.
Rheos HOPE4HF trial. Available at: http://clinicaltrials.gov/ct2/show/NCT00957073. Accessed 1 Apr 2012.
Barostim neo system in the treatment of heart failure. Available at: http://clinicaltrials.gov/ct2/show/NCT01471860. Accessed 1 Apr 2012.
Lopshire JC, Zhou X, Dusa C, et al. Spinal cord stimulation improves ventricular function and reduces ventricular arrhythmias in a canine postinfarction heart failure model. Circulation. 2009;120:286–94.
Disclosures
Conflicts of interest: D.P. Zipes is the US Principal Investigator and J.C. Lopshire is a site Investigator for DEFEAT-HF, sponsored by Medtronic, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lopshire, J.C., Zipes, D.P. Device Therapy to Modulate the Autonomic Nervous System to Treat Heart Failure. Curr Cardiol Rep 14, 593–600 (2012). https://doi.org/10.1007/s11886-012-0292-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-012-0292-8