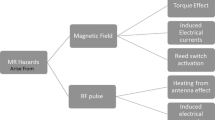
Abstract
Given the advances of MRI and cardiovascular technology, it is becoming increasingly likely that a patient with a cardiovascular device will be a candidate for an MRI procedure. However, many cardiac devices are currently considered to be contraindicated in the MR environment. This may prove to be a significant public health problem as many patients in need of MRI are denied the procedure because of the presence of a cardiovascular device. However, research studies have shown that with proper precautions and technique patients with cardiac devices can undergo successful MRI safely on the current platforms.
Similar content being viewed by others
References and Recommended Reading
Kalin R, Stanton MS: Current clinical issues for MRI scanning of pacemaker and defibrillator patients. Pacing Clin Electrophysiol 2005, 28:326–328.
United States Food and Drug Administration. A Primer on Medical Device Interactions with Magnetic Resonance Imaging Systems. Available at http://www.fda.gov/cdrh/ode/primerf6.html. Accessed October 26, 2006.
Society for Testing and Materials (ASTM) International, Designation: F2503-05. Standard Practice for Marking Medical Devices and Other Items for Safety in the Magnetic Resonance Environment. West Conshohocken, PA: ASTM International; 2005.
Pavlicek W, Geisinger M, Castle L, et al.: The effects of nuclear magnetic resonance on patients with cardiac pacemakers. Radiology 1983, 174:149–153.
Fetter J, Aram G, Holmes D, et al.: The effects of nuclear magnetic resonance imagers on external and implantable pulse generators. Pacing Clin Electrophysiol 1984, 7:720–727.
Lauck G, von Smekal A, Wolke S, et al.: Effects of nuclear magnetic resonance imaging on cardiac pacemakers. Pacing Clin Electrophysiol 1995, 18:1549–1555.
Luechinger R, Duru F, Zeijlemaker VA, et al.: Pacemaker reed switch behavior in 0.5, 1.5 and 3.0 Tesla magnetic resonance imaging units: are reed switches always closed in strong magnetic fields? Pacing Clin Electrophysiol 2002, 25:1419–1423.
Erlebacher JA, Cahill PT, Panizzo F, et al.: Effect of magnetic resonance imaging on DDD pacemakers. Am J Cardiol 1986, 57:437–440.
Achenbach S, Moshage W, Diem B, et al.: Effects of magnetic resonance imaging on cardiac pacemakers and electrodes. Am Heart J 1997, 134:467–473.
Sommer T, Valhaus C, Lauck G, et al.: MR imaging and cardiac pacemakers: in vitro evaluation and in vivo studies in 51 patients at 1.5 T. Radiology 2000, 215:869–879.
Shellock FG, O’Neil MO, Ivans V, et al.: Cardiac pacemakers and implantable cardioverter defibrillators are unaffected by operation of an extremity MR imaging system. Am J Roentgenol 1999, 172:165–170.
Holmes DR, Hayes DL, Gray JE, et al.: The effects of magnetic resonance imaging on implantable pulse generators. Pacing Clin Electrophysiol 1986, 9:360–370.
Hayes DL, Holmes DR, Gray JE, et al.: Effect of 1.5 Tesla nuclear magnetic resonance imaging scanner on implanted permanent pacemakers. J Am Coll Cardiol 1987, 10:782–786.
Peden CJ, Collins AG, Butson PC, et al.: Induction of microcurrents in critically ill patients in magnetic resonance systems. Crit Care Med 1993, 21:1923–1928.
Roguin A, Zviman MM, Meininger GR et al.: Modern pacemaker and implantable cardioverter/defibrillator systems can be magnetic resonance imaging safe: in vitro and in vivo assessment of safety and function at 1.5 T. Circulation 2004, 110:475–482.
Luechinger R, Zeijlemaker VA, Pedersen EM et al.: In vivo heating of pacemaker leads during magnetic resonance imaging. Eur Heart J 2005, 26:376–383.
Nahrendorf M, Hiller KH, Hu K, et al.: Pacing in high field cardiac magnetic resonance imaging. Pacing Clin Electrophysiol 2004, 27:671–674.
Valhaus C, Sommer T, Lewalter T, et al.: Interference with cardiac pacemakers by magnetic resonance imaging: are there irreversible changes at 0.5 Tesla? Pacing Clin Electrophysiol 2001, 24:489–495.
Garcia-Bolao I, Albaladejo V, Benito A, et al.: Magnetic resonance imaging in a patient with a dual-chamber pacemaker. Acta Cardiol 1998, 1:33–35.
Sommer T, Naehle CP, Yang A, et al.: Strategy for safe performance of extrathoracic MRI at 1.5 Tesla in the presence of cardiac pacemakers in non-pacemaker dependent patients. Circulation 2006, 114:1285–1292.
Goldshear D, Amikam S, Boulos M, et al.: Magnetic resonance imaging for patients with permanent pacemakers: initial clinical experience. IMAJ 2006, 8:91–94.
Del Ojo JL, Villalba J, Sanz O, et al.: Is magnetic resonance imaging safe in cardiac pacemaker recipients? Pacing Clin Electrophysiol 2005, 28:274–278.
Martin ET, Coman JC, Shellock FG, et al.: Magnetic resonance imaging and cardiac pacemaker safety at 1.5-T. J Am Coll Cardiol 2004, 43:1315–1324.
Nazarian S, Roguin A, Zviman MM, et al.: Clinical utility and safety of a protocol for noncardiac and cardiac magnetic resonance imaging of patients with permanent pacemakers and implantable-cardioverter defibrillators at 1.5 Tesla. Circulation 2006, 114:1277–1284.
Gimbel JR, Johnson D, Levine PA, et al.: Safe performance of magnetic resonance imaging on five patients with permanent cardiac pacemakers. Pacing Clin Electrophysiol 1996, 19:913–919.
Irnich W, Irnich B, Bartsch C, et al.: Do we need pacemakers resistant to magnetic resonance imaging? Europace 2005, 7:353–365.
Fontaine JM, Mohamed FB, Gottlieb C, et al.: Rapid ventricular pacing in a pacemaker patient undergoing magnetic resonance imaging. Pacing Clin Electrophysiol 1998, 21:1336–1339.
Luechinger R, Duru F, Scheidegger MB, et al.: Force and torque effects of a 1.5-Tesla MRI scanner on cardiac pacemakers and ICDs. Pacing Clin Electrophysiol 2001, 24:199–205.
Fiek M, Remp T, Reithmann C, et al.: Complete loss of ICD programmability after magnetic resonance imaging. Pacing Clin Electrophysiol 2004, 27:1002–1004.
Anfinsen O, Bernsten RF, Aass H, et al.: Implantable cardioverter defibrillator dysfunction during and after magnetic resonance imaging. Pacing Clin Electrophysiol 2002, 25:1400–1402.
Sandler DA, Coman JA, Auerbach EG, Martin ET: Magnetic resonance imaging of patients with pacemakers and implantable cardioverter defibrillators in a 1.5-Tesla magnet: a five year experience. Heart Rhythm Society, 27th Annual Scientific Sessions. Boston, MA: May 17–20, 2006.
Gimbel JR, Kanal E, Schwartz KM, et al.: Outcome of magnetic resonance imaging (MRI) in selected patients with implantable cardioverter defibrillators (ICDs). Pacing Clin Electrophysiol 2005, 28:270–273.
Murphy KJ, Cohan RH, Ellis JH: MR imaging in patients with epicardial pacemaker wires. AJR Am J Roentgenol 1999, 172:727–728.
Hartnell GG, Spence L, Hughes LA, et al.: Safety of MR imaging in patients who have retained metallic materials after cardiac surgery. AJR Am J Roentgenol 1997, 168:1157–1159.
Shellock FG, Riedinger MS: Reproducibility and accuracy of using room-temperature vs. ice-temperature injectate for thermodilution cardiac output determination. Heart Lung 1983, 12:175–176.
Shellock FG, Morisoli SM: Ex vivo evaluation of ferromagnetism, heating, and artifacts produced by heart valve prostheses exposed to a 1.5-T MR system. J Magn Reson Imaging 1994, 4:756–758.
Edwards MB, Taylor KM, Shellock FG: Prosthetic heart valves: evaluation of magnetic field interactions, heating, and artifacts at 1.5 T. J Magn Reson Imaging 2000, 12:363–369.
Shellock FG: Prosthetic heart valves and annuloplasty rings: assessment of magnetic field interactions, heating, and artifacts at 1.5 Tesla. J Cardiovasc Magn Reson 2001, 3:317–324.
Soulen RL, Budinger TF, Higgins CB: Magnetic resonance imaging of prosthetic heart valves. Radiology 1985, 154:705–707.
Edwards MB, Draper ER, Hand JW, et al.: Mechanical testing of human cardiac tissue: some implications for MRI safety. J Cardiovasc Magn Reson 2005, 7:835–840.
Pruefer D, Kalden P, Schreiber W, et al.: In vitro investigation of prosthetic heart valves in magnetic resonance imaging: evaluation of potential hazards. J Heart Valve Dis 2001, 10:410–414.
Randall PA, Kohman LJ, Scalzetti EM, et al.: Magnetic resonance imaging of prosthetic cardiac valves in vitro and in vivo. Am J Cardiol 1988, 62:973–976.
Condon B, Hadley DM: Potential MR hazard to patients with metallic heart valves: the Lenz effect. J Magn Reson Imaging 2000, 12:171–176.
Shellock FG: Biomedical implants and devices: assessment of magnetic field interactions with a 3.0-Tesla MR system. J Magn Reson Imaging 2002, 16:721–732.
Shellock FG: Reference Manual for Magnetic Resonance Safety, Implants, and Devices: 2006 Edition. Los Angeles: Biomedical Research Publishing Group; 2006.
Rajappan K, Melina G, Bellenger NG, et al.: Evaluation of left ventricular function and mass after Medtronic Freestyle versus homograft aortic root replacement using cardio-vascular magnetic resonance. J Heart Valve Dis 2002, 11:60–65.
Vliegen HW, van Straten A, de Roos A, et al.: Magnetic resonance imaging to assess the hemodynamic effects of pulmonary valve replacement in adults late after repair of Tetralogy of Fallot. Circulation 2002, 106:1703–1707.
Khambadkone S, Coats L, Taylor A, et al.: Percutaneous pulmonary valve implantation in humans: results in 59 consecutive patients. Circulation 2005, 112:1189–1197.
Scott NA, Pettigrew RI: Absence of movement of coronary stents after placement in a magnetic resonance imaging field. Am J Cardiol 1994, 73:900–901.
Friedrich MG, Strohm O, Kivelitz D et al.: Behavior of implantable coronary stents during magnetic resonance imaging. Int J Cardiovasc Intervent 1999, 2:217–222.
Shellock FG, Shellock VJ: Metallic stents: evaluation of MR imaging safety. AJR Am J Roentgenol 1999, 173:543–547.
Hug J, Nagel E, Bornstedt A, et al.: Coronary arterial stents: safety and artifacts during MR imaging. Radiology 2000, 216:781–787.
Shellock FG: Biomedical implants and devices: assessment of magnetic field interactions with a 3.0-Tesla MR system. J Magn Reson Imaging 2002, 16:721–732.
Shellock FG, Forder JR: Drug eluting coronary stent: in vitro evaluation of magnet resonance safety at 3 Tesla. J Cardiovasc Magn Reson 2005, 7:415–419.
Shellock FG: MR safety at 3-Tesla: bare metal and drug eluting coronary artery stents. Signals 2005, 53:26–27.
Syed MA, Carlson K, Murphy M, et al.: Long-term safety of cariac magnetic resonance imaging in the first few days after bare-metal stent implantation. J Magn Reson Imaging 2006, 24:1056–1061.
Patel M, Albert TS, Kandzari DE, et al.: Acute myocardial infarction: Safety of cardiac MR imaging after percutaneous revascularization with stents. Radiology 2006, 240:674–680.
Shellock FG, Morisoli SM: Ex vivo evaluation of ferromagnetism and artifacts of cardiac occluders exposed to a 1.5-T MR system. J Magn Reson Imaging 1994, 4:213–215.
Bock M, Mohrs OK, Voigtlaender T, et al.: MRI safety aspects and artifacts of atrial septal defect and patent foramen ovale occluders at 1.5 Tesla: a phantom study. Rofo 2006, 178:272–277.
Shellock FG, Valencerina S: Septal repair implants: evaluation of magnetic resonance imaging safety at 3 T. Magn Reson Imaging 2005, 23:1021–1025.
Shellock FG, Tkach JA, Ruggieri PM, Masaryk TJ: Cardiac pacemakers, ICDs, and loop recorder: evaluation of translational attraction using conventional (“long-bore”) and “short-bore” 1.5-and 3.0-Tesla MR systems. J Cardiovasc Magn Reson 2003, 5:387–397.
Gimbel JR, Zarghami J, Machado C, Wilkoff BL: Safe scanning, but frequent artifacts mimicking bradycardia and tachycardia during magnetic resonance imaging (MRI) in patients with an implantable loop recorder (ILR). Ann Noninvasive Electrocardiol 2005, 10:404–408.
Anfinsen O, Berntsen RF, Aass H, et al.: Implantable cardioverter defibrillator dysfunction during and after magnetic resonance imaging. Pacing Clin Electrophysiol 2002, 25:1400–1402.
Wollmann C, Grude M, Tombach B, et al.: Safe performance of magnetic resonance imaging on a patient with an ICD. PACE 2005, 28:339–342.
Roguin A, Donahue JK, Bomma CS, et al.: Cardiac magnetic resonance imaging in a patient with implantable cardioverter defibrillator. Pacing Clin Electro Physiol 2005, 28:336–338.
Gimbel JR, Kanal E, Schwartz KM, et al.: Outcome of magnetic resonance imaging (MRI) in selected patients with implantable cardioverter defibrillators (ICDs). PACE 2005, 28:270–273.
Nemec J: Suppression of implantable cardioverter defibrillator therapy during magnetic resonance imaging. J Cardiovasc Electrophys 2006, 17:444–445.
Iberer F, Justich E, Stenzl W, et al.: Nuclear magnetic resonance imaging of a patient with implanted transvenous pacemaker. Herz 1987, 7:196–199.
Achenbach S, Moshage W, Kuhn I, et al.: Kernspintomagraphie bei einem patienten mit zweikammerschrittmachersystem. Z Kardiol 1995, 87(Suppl):119.
Alagona P, Toole JC, Maniscalco BS, et al.: Nuclear magnetic resonance imaging in a patient with a DDD pacemaker [letter]. PACE 1989, 12:619.
Inbar S, Larson J, Burt T, et al.: Case report: nuclear magnetic resonance imaging in a patient with a pacemaker. Am J Med Sci 1993, 3:174–175.
Johnson D: Magnetic resonance imaging effects and considerations with permanent cardiac pacemakers [abstract]. PACE 1994, 17(Suppl):772.
Sommer T, Lauck G, Schimpf R, et al.: MRI in patients with cardiac pacemakers: in vitro and in vivo evaluation at 0.5 Tesla. Rofo 1998, 168:36–43.
Pennell DJ: Cardiac magnetic resonance with a pacemaker in situ: can it be done [abstract]? J Cardiovasc Magn Reson 1999, 1:72.
Juratli N, Sparker J, Gimbel JR, et al.: Strategies for the safe performance of magnetic resonance imaging in selected pacemaker patients [abstract]. Circulation 2001, 104:II638–II639.
Schmiedel A, Hackenbroch M, Reinke M, et al.: Magnetic resonance imaging of the brain at 1.5 tesla in patients with cardiac pacemakers: can it be done [abstract]? J Cardiovasc Magn Reson 2002, 4:39–40.
Maldonado JG, Pereira ME, Albuquerque KR et al.: Magnetic resonance imaging in a patient with a pacemaker. Arquivos Brasileiros de Cardiologia 2005, 84:428–430.
Goldsher D, Amikam S, Boulos M, et al.: Magnetic resonance imaging for patients with permanent pacemakers: initial clinical experience. IMAJ 2006, 8:91–94.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martin, E.T., Sandler, D.A. MRI in patients with cardiac devices. Curr Cardiol Rep 9, 63–71 (2007). https://doi.org/10.1007/s11886-007-0012-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-007-0012-y