Abstract
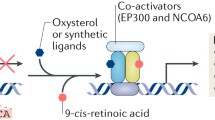
Liver X receptors (LXRs) belong to the nuclear receptor superfamily of ligand-dependent transcription factors. LXRs are activated by oxysterols, metabolites of cholesterol, and therefore act as intracellular sensors of this lipid. There are two LXR genes (α and β) that display distinct tissue/cell expression profiles. LXRs interact with regulatory sequences in target genes as heterodimers with retinoid X receptor. Such direct targets of LXR actions include important genes implicated in the control of lipid homeostasis, particularly reverse cholesterol transport. In addition, LXRs attenuate the transcription of genes associated with the inflammatory response indirectly by transrepression. In this review, we describe recent evidence that both highlights the key roles of LXRs in atherosclerosis and inflammation and provides novel insights into the mechanisms underlying their actions. In addition, we discuss the major limitations of LXRs as therapeutic targets for the treatment of atherosclerosis and how these are being addressed.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Chinetti-Gbaguidi G, Staels B. Lipid ligand-activated transcription factors regulating lipid storage and release in human macrophages. Biochim Biophys Acta. 2009;1791:486–93.
Bensinger SJ, Tontonoz P. Integration of metabolism and inflammation by lipid-activated nuclear receptors. Nature. 2008;454:470–7.
Oosterveer MH, Grefhorst A, Groen AK, Kuipers F. The liver X receptor: control of cellular lipid homeostasis and beyond: implications for drug design. Prog Lipid Res. 2010;49:343–52.
Pourcet B, Feig JE, Vengrenyuk Y, et al. LXRalpha regulates macrophage arginase 1 through PU.1 and interferon regulatory factor 8. Circ Res. 2011;109:492–501.
Schultz JR, Tu H, Luk A, et al. Role of LXRs in control of lipogenesis. Genes Dev. 2000;14:2831–8.
Cha JY, Repa JJ. The liver X receptor (LXR) and hepatic lipogenesis. The carbohydrate-response element-binding protein is a target gene of LXR. J Biol Chem. 2007;282:743–51.
•• Bischoff ED, Daige CL, Petrowski M, et al. Non-redundant roles for LXRalpha and LXRbeta in atherosclerosis susceptibility in low density lipoprotein receptor knockout mice. J Lipid Res. 2010;51:900–6. In contrast to numerous studies showing functional redundancy between LXR-α and -β, this work, utilizing individual knockouts of LXRα and LXRβ in the LDL receptor knockout background, shows that LXRα plays a more selective role in limiting atherosclerosis in response to hyperlipidemia.
Whitney KD, Watson MA, Goodwin B, et al. Liver X receptor (LXR) regulation of the LXRalpha gene in human macrophages. J Biol Chem. 2001;276:43509–15.
Hashimoto K, Matsumoto S, Yamada M, Satoh T, Mori M. Liver X receptor-alpha gene expression is positively regulated by thyroid hormone. Endocrinology. 2007;148:4667–75.
Kim MS, Sweeney TR, Shigenaga JK, et al. Tumor necrosis factor and interleukin 1 decrease RXRalpha, PPARalpha, PPARgamma, LXRalpha, and the coactivators SRC-1, PGC-1alpha, and PGC-1beta in liver cells. Metabolism. 2007;56:267–79.
Hao XR, Cao DL, Hu YW, et al. IFN-gamma down-regulates ABCA1 expression by inhibiting LXRalpha in a JAK/STAT signaling pathway-dependent manner. Atherosclerosis. 2009;203:417–28.
Chen M, Beaven S, Tontonoz P. Identification and characterization of two alternatively spliced transcript variants of human liver X receptor alpha. J Lipid Res. 2005;46:2570–9.
Hashimoto K, Ishida E, Matsumoto S, et al. A liver X receptor (LXR)-beta alternative splicing variant (LXRBSV) acts as an RNA co-activator of LXR-beta. Biochem Biophys Res Commun. 2009;390:1260–5.
Chen M, Bradley MN, Beaven SW, Tontonoz P. Phosphorylation of the liver X receptors. FEBS Lett. 2006;580:4835–41.
Delvecchio CJ, Capone JP. Protein kinase C alpha modulates liver X receptor alpha transactivation. J Endocrinol. 2008;197:121–30.
Torra IP, Ismaili N, Feig JE, et al. Phosphorylation of liver X receptor alpha selectively regulates target gene expression in macrophages. Mol Cell Biol. 2008;28:2626–36.
Huang CJ, Feltkamp D, Nilsson S, Gustafsson JA. Synergistic activation of RLD-1 by agents triggering PKA and PKC dependent signalling. Biochem Biophys Res Commun. 1998;243:657–63.
Yamamoto T, Shimano H, Inoue N, et al. Protein kinase A suppresses sterol regulatory element-binding protein-1C expression via phosphorylation of liver X receptor in the liver. J Biol Chem. 2007;282:11687–95.
Huwait EA, Greenow KR, Singh NN, Ramji DP. A novel role for c-Jun N-terminal kinase and phosphoinositide 3-kinase in the liver X receptor-mediated induction of macrophage gene expression. Cell Signal. 2011;23:542–9.
Li X, Zhang S, Blander G, Tse JG, Krieger M, Guarente L. SIRT1 deacetylates and positively regulates the nuclear receptor LXR. Mol Cell. 2007;28:91–106.
Ghisletti S, Huang W, Ogawa S, et al. Parallel SUMOylation-dependent pathways mediate gene- and signal-specific transrepression by LXRs and PPARgamma. Mol Cell. 2007;25:57–70.
Anthonisen EH, Berven L, Holm S, Nygard M, Nebb HI, Gronning-Wang LM. Nuclear receptor liver X receptor is O-GlcNAc-modified in response to glucose. J Biol Chem. 2010;285:1607–15.
McLaren JE, Michael DR, Ashlin TG, Ramji DP. Cytokines, macrophage lipid metabolism and foam cells: implications for cardiovascular disease therapy. Prog Lipid Res. 2011;50:331–47.
Morello F, Saglio E, Noghero A, et al. LXR-activating oxysterols induce the expression of inflammatory markers in endothelial cells through LXR-independent mechanisms. Atherosclerosis. 2009;207:38–44.
Peter A, Weigert C, Staiger H, et al. Induction of stearoyl-CoA desaturase protects human arterial endothelial cells against lipotoxicity. Am J Physiol Endocrinol Metab. 2008;295:E339–49.
Zhu M, Fu Y, Hou Y, et al. Laminar shear stress regulates liver X receptor in vascular endothelial cells. Arterioscler Thromb Vasc Biol. 2008;28:527–33.
Blaschke F, Leppanen O, Takata Y, et al. Liver X receptor agonists suppress vascular smooth muscle cell proliferation and inhibit neointima formation in balloon-injured rat carotid arteries. Circ Res. 2004;95:e110–23.
Delvecchio CJ, Bilan P, Radford K, et al. Liver X receptor stimulates cholesterol efflux and inhibits expression of proinflammatory mediators in human airway smooth muscle cells. Mol Endocrinol. 2007;21:1324–34.
Delvecchio CJ, Bilan P, Nair P, Capone JP. LXR-induced reverse cholesterol transport in human airway smooth muscle is mediated exclusively by ABCA1. Am J Physiol Lung Cell Mol Physiol. 2008;295:L949–57.
Imayama I, Ichiki T, Patton D, et al. Liver X receptor activator downregulates angiotensin II type 1 receptor expression through dephosphorylation of Sp1. Hypertension. 2008;51:1631–6.
• Nakaya K, Tohyama J, Naik SU, et al. Peroxisome proliferator-activated receptor-α activation promotes macrophage reverse cholesterol transport through a liver X receptor-dependent pathway. Arterioscler Thromb Vasc Biol. 2011;31:1276–82. This study shows that a potent PPARα agonist stimulates reverse cholesterol transport in vivo through the PPARα-LXR pathway.
Chawla A, Boisvert WA, Lee CH, et al. A PPAR gamma-LXR-ABCA1 pathway in macrophages is involved in cholesterol efflux and atherogenesis. Mol Cell. 2001;7:161–71.
Lu KY, Ching LC, Su KH, et al. Erythropoietin suppresses the formation of macrophage foam cells: role of liver X receptor alpha. Circulation. 2010;121:1828–37.
Qiu G, Hill JS. Atorvastatin inhibits ABCA1 expression and cholesterol efflux in THP-1 macrophages by an LXR-dependent pathway. J Cardiovasc Pharmacol. 2008;51:388–95.
•• Chinetti-Gbaguidi G, Baron M, Bouhlel MA, et al. Human atherosclerotic plaque alternative macrophages display low cholesterol handling but high phagocytosis because of distinct activities of the PPARγ and LXRα pathways. Circ Res. 2011;108:985–95. This study provides a link between LXRs and CD68(+) mannose receptor M2 macrophages seen in human atherosclerotic lesions. These macrophages have reduced capacity to handle and efflux choesterol because of low levels of LXRα and its target genes.
Buono C, Li Y, Waldo SW, Kruth HS. Liver X receptors inhibit human monocyte-derived macrophage foam cell formation by inhibiting fluid-phase pinocytosis of LDL. J Lipid Res. 2007;48:2411–8.
•• Zelcer N, Hong C, Boyadjian R, Tontonoz P. LXR regulates cholesterol uptake through Idol-dependent ubiquitination of the LDL receptor. Science. 2009;325:100–4. This study shows that LXRs also suppress LDL uptake via a novel pathway that involves transcriptional induction of the E3 ubiquitin ligase Idol (inducible degrader of the LDLR), which ubiquinates LDLR on its cytoplasmic domain, thereby targeting it for degradation. Adenoviral-mediated expression of Idol in mouse liver results in elevation of plasma LDL levels.
Bultel S, Helin L, Clavey V, et al. Liver X receptor activation induces the uptake of cholesteryl esters from high density lipoproteins in primary human macrophages. Arterioscler Thromb Vasc Biol. 2008;28:2288–95.
Schuster GU, Parini P, Wang L, et al. Accumulation of foam cells in liver X receptor-deficient mice. Circulation. 2002;106:1147–53.
Tangirala RK, Bischoff ED, Joseph SB, et al. Identification of macrophage liver X receptors as inhibitors of atherosclerosis. Proc Natl Acad Sci U S A. 2002;99:11896–901.
Naik SU, Wang X, Da Silva JS, et al. Pharmacological activation of liver X receptors promotes reverse cholesterol transport in vivo. Circulation. 2006;113:90–7.
Scotti E, Hong C, Yoshinaga Y, et al. Targeted disruption of the idol gene alters cellular regulation of the low-density lipoprotein receptor by sterols and liver x receptor agonists. Mol Cell Biol. 2011;31:1885–93.
Joseph SB, McKilligin E, Pei L, et al. Synthetic LXR ligand inhibits the development of atherosclerosis in mice. Proc Natl Acad Sci U S A. 2002;99:7604–9.
•• Spyridon M, Moraes LA, Jones CI, et al. LXR as a novel antithrombotic target. Blood. 2011;117:5751–61. This report demonstrates LXRs as novel antithrombotic targets. LXR agonists were found to inhibit platelet aggregation by a range of agonists and reduced the size and stability of thrombi in an in vivo model of thrombosis in mice.
Terasaka N, Hiroshima A, Koieyama T, et al. T-0901317, a synthetic liver X receptor ligand, inhibits development of atherosclerosis in LDL receptor-deficient mice. FEBS Lett. 2003;536:6–11.
Levin N, Bischoff ED, Daige CL, et al. Macrophage liver X receptor is required for antiatherogenic activity of LXR agonists. Arterioscler Thromb Vasc Biol. 2005;25:135–42.
• Verschuren L, de Vries-van der Weij J, Zadelaar S, Kleemann R, Kooistra T. LXR agonist suppresses atherosclerotic lesion growth and promotes lesion regression in apoE*3Leiden mice: time course and mechanisms. J Lipid Res. 2009;50:301–11. This study shows that LXR agonists not only suppress atherosclerotic lesion growth but also promote lesion regression.
Feig JE, Pineda-Torra I, Sanson M, et al. LXR promotes the maximal egress of monocyte-derived cells from mouse aortic plaques during atherosclerosis regression. J Clin Invest. 2010;120:4415–24.
Rigamonti E, Chinetti-Gbaguidi G, Staels B. Regulation of macrophage functions by PPAR-alpha, PPAR-gamma, and LXRs in mice and men. Arterioscler Thromb Vasc Biol. 2008;28:1050–9.
Menke JG, Macnaul KL, Hayes NS, et al. A novel liver X receptor agonist establishes species differences in the regulation of cholesterol 7alpha-hydroxylase (CYP7a). Endocrinology. 2002;143:2548–58.
Larrede S, Quinn CM, Jessup W, et al. Stimulation of cholesterol efflux by LXR agonists in cholesterol-loaded human macrophages is ABCA1-dependent but ABCG1-independent. Arterioscler Thromb Vasc Biol. 2009;29:1930–6.
• Peng D, Hiipakka RA, Xie JT, et al. A novel potent synthetic steroidal liver X receptor agonist lowers plasma cholesterol and triglycerides and reduces atherosclerosis in LDLR(-/-) mice. Br J Pharmacol. 2011;162:1792–804. A recent study demonstrating the efficacy of novel LXR agonists (ATI-111 in this case) that significantly attenuate atherosclerosis in mouse model systems without inducing fatty liver and hypertriglyceridemia.
Kratzer A, Buchebner M, Pfeifer T, et al. Synthetic LXR agonist attenuates plaque formation in apoE-/- mice without inducing liver steatosis and hypertriglyceridemia. J Lipid Res. 2009;50:312–26.
Peng D, Hiipakka RA, Dai Q, et al. Antiatherosclerotic effects of a novel synthetic tissue-selective steroidal liver X receptor agonist in low-density lipoprotein receptor-deficient mice. J Pharmacol Exp Ther. 2008;327:332–42.
van der Hoorn J, Lindén D, Lindahl U, et al. Low dose of the liver X receptor agonist, AZ876, reduces atherosclerosis in APOE*3Leiden mice without affecting liver or plasma triglyceride levels. Br J Pharmacol. 2011;162:1553–63.
Yan W, Zhang T, Cheng J, Zhou X, Qu X, Hu H. Liver X receptor agonist methyl-3β-hydroxy-5α,6α-epoxycholanate attenuates atherosclerosis in apolipoprotein E knockout mice without increasing plasma triglyceride. Pharmacology. 2010;86:306–12.
• Giannarelli C, Cimmino G, Connolly TM, et al. Synergistic effect of liver X receptor activation and simvastatin on plaque regression and stabilization: an magnetic resonance imaging study in a model of advanced atherosclerosis. Eur Heart J. 2011;33:264–73. doi:10.1093/eurheartj/ehr136. This study demonstrates the potential of combination therapy (LXR agonist and statins in this case) in the treatment of atherosclerosis.
Quinet EM, Basso MD, Halpern AR, et al. LXR ligand lowers LDL cholesterol in primates, is lipid neutral in hamster, and reduces atherosclerosis in mouse. J Lipid Res. 2009;50:2358–70.
DiBlasio-Smith EA, Arai M, Quinet EM, et al. Discovery and implementation of transcriptional biomarkers of synthetic LXR agonists in peripheral blood cells. J Transl Med. 2008;6:59.
Bradley MN, Hong C, Chen M, et al. Ligand activation of LXR beta reverses atherosclerosis and cellular cholesterol overload in mice lacking LXR alpha and apoE. J Clin Invest. 2007;117:2337–46.
Quinet EM, Savio DA, Halpern AR, et al. Liver X receptor (LXR)-beta regulation in LXRalpha-deficient mice: implications for therapeutic targeting. Mol Pharmacol. 2006;70:1340–9.
Mukherjee R, Locke KT, Miao B, et al. Novel peroxisome proliferator-activated receptor alpha agonists lower low-density lipoprotein and triglycerides, raise high-density lipoprotein, and synergistically increase cholesterol excretion with a liver X receptor agonist. J Pharmacol Exp Ther. 2008;327:716–26.
Teupser D, Kretzschmar D, Tennert C, et al. Effect of macrophage overexpression of murine liver X receptor-alpha (LXR-alpha) on atherosclerosis in LDL-receptor deficient mice. Arterioscler Thromb Vasc Biol. 2008;28:2009–15.
Li G, Biju KC, Xu X, et al. Macrophage LXRα gene therapy ameliorates atherosclerosis as well as hypertriglyceridemia in LDLR(-/-) mice. Gene Ther. 2011;18:835–41.
Rippmann JF, Schoelch C, Nolte T, et al. Improved lipid profile through liver-specific knockdown of liver X receptor alpha in KKAy diabetic mice. J Lipid Res. 2009;50:22–31.
•• Lo Sasso G, Murzilli S, Salvatore L, et al. Intestinal specific LXR activation stimulates reverse cholesterol transport and protects from atherosclerosis. Cell Metabol. 2010;12:187–93. This study demonstrates that intestinal-specific LXR activation reduces intestinal cholesterol absorption, improves lipoprotein profile, increases reverse cholesterol transport in vivo, and protects against atherosclerosis in a mouse model system.
Joseph SB, Castrillo A, Laffitte BA, Mangelsdorf DJ, Tontonoz P. Reciprocal regulation of inflammation and lipid metabolism by liver X receptors. Nat Med. 2003;9:213–9.
Hindinger C, Hinton DR, Kirwin SJ, et al. Liver X receptor activation decreases the severity of experimental autoimmune encephalomyelitis. J Neurosci Res. 2006;84:1225–34.
Zelcer N, Khanlou N, Clare R, et al. Attenuation of neuroinflammation and Alzheimer’s disease pathology by liver x receptors. Proc Natl Acad Sci U S A. 2007;104:10601–6.
A-Gonzalez N, Bensinger SJ, Hong C, et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity. 2009;31:245–58.
Zhang-Gandhi CX, Drew PD. Liver X receptor and retinoid X receptor agonists inhibit inflammatory responses of microglia and astrocytes. J Neuroimmunol. 2007;183:50–9.
Morales JR, Ballesteros I, Deniz JM, et al. Activation of liver X receptors promotes neuroprotection and reduces brain inflammation in experimental stroke. Circulation. 2008;118:1450–9.
Korf H, Vander Beken S, Romano M, et al. Liver X receptors contribute to the protective immune response against Mycobacterium tuberculosis in mice. J Clin Invest. 2009;119:1626–37.
Hanley TM, Blay Puryear W, Gummuluru S, Viglianti GA. PPARgamma and LXR signaling inhibit dendritic cell-mediated HIV-1 capture and trans-infection. PLoS Pathog. 2010;6:e1000981.
Marathe C, Bradley MN, Hong C, et al. The arginase II gene is an anti-inflammatory target of liver X receptor in macrophages. J Biol Chem. 2006;281:32197–206.
Castrillo A, Joseph SB, Marathe C, Mangelsdorf DJ, Tontonoz P. Liver X receptor-dependent repression of matrix metalloproteinase-9 expression in macrophages. J Biol Chem. 2003;278:10443–9.
Ogawa D, Stone JF, Takata Y, et al. Liver x receptor agonists inhibit cytokine-induced osteopontin expression in macrophages through interference with activator protein-1 signaling pathways. Circ Res. 2005;96:e59–67.
Blaschke F, Takata Y, Caglayan E, et al. A nuclear receptor corepressor-dependent pathway mediates suppression of cytokine-induced C-reactive protein gene expression by liver X receptor. Circ Res. 2006;99:e88–99.
•• Venteclef N, Jakobsson T, Ehrlund A, et al. GPS2-dependent corepressor/SUMO pathways govern anti-inflammatory actions of LRH-1 and LXRbeta in the hepatic acute phase response. Genes Dev. 2010;24:381–95. This study provides mechanistic insight into the anti-inflammatory actions of LXRs in the hepatic acute phase response (APR). The work demonstrates that the anti-inflammatory actions of LXR agonists are mediated selectively by LXRβ and involve SUMOylation-dependent recruitment of the nuclear receptor to hepatic APR promoters leading to the prevention of clearance of NCoR complex.
•• Ghisletti S, Huang W, Jepsen K, et al. Cooperative NCoR/SMRT interactions establish a corepressor-based strategy for integration of inflammatory and anti-inflammatory signaling pathways. Genes Dev. 2009;23:681–93. This study provides molecular insight into the mechanisms underlying transrepression of a large cohort of inflammatory genes and demonstrates that NCoR and SMRT corepressors are required for nearly all the transrepression activities of LXRs.
• Huang W, Ghisletti S, Perissi V, Rosenfeld MG, Glass CK. Transcriptional integration of TLR2 and TLR4 signaling at the NCoR derepression checkpoint. Mol Cell. 2009;3548–57. This report shows that a pathway in which TLR4 uses NFκB to deliver IKKε to target gene promoters containing κB and AP1 sites and leads to the phosphorylation of c-Jun, and the clearance of the NCoR complex is sensitive to transrepression by LXRs.
•• Huang W, Ghisletti S, Saijo K, et al. Coronin 2A mediates actin-dependent de-repression of inflammatory response genes. Nature. 2011, 470:414–8. This work identifies a coronin 2A-actin dependent mechanism for the de-repression of inflammatory genes and provides insight on how post-translational modification of LXR in response to inflammatory signals can impact its transrepression activity.
McLaren J, Ramji DP. Interferon gamma: a master regulator of atherosclerosis. Cytokine Growth Factor Rev. 2009;20:125–35.
Li N, Salter RC, Ramji DP. Molecular mechanisms underlying the inhibition of IFN-gamma-induced, STAT1-mediated gene transcription in human macrophages by simvastatin and agonists of PPARs and LXRs. J Cell Biochem. 2010;112:675–83.
• Lee JH, Park SM, Kim OS, et al. Differential SUMOylation of LXRalpha and LXRbeta mediates transrepression of STAT1 inflammatory signaling in IFN-gamma-stimulated brain astrocytes. Mol Cell. 2009;35:806–17. This work demonstrates that LXR ligands inhibit STAT1 binding to target gene promoters and provides insight into the mechanisms underlying transrepression by LXRs through STAT1.
Fontaine C, Rigamonti E, Nohara A, et al. Liver X receptor activation potentiates the lipopolysaccharide response in human macrophages. Circ Res. 2007;101:40–9.
Asquith DL, Miller AM, Hueber AJ, et al. Liver X receptor agonism promotes articular inflammation in murine collagen-induced arthritis. Arthritis Rheum. 2009;60:2655–65.
Torocsik D, Barath M, Benko S, et al. Activation of liver X receptor sensitizes human dendritic cells to inflammatory stimuli. J Immunol. 2010;184:5456–65.
Choi JH, Banks AS, Kamenecka TM, et al. Antidiabetic actions of a non-agonist PPARgamma ligand blocking Cdk5-mediated phosphorylation. Nature. 2011;477:477–81.
Acknowledgments
We thank the British Heart Foundation for financial support (Grants PG/05/096, PG/08/073/25520 and PG/10/55/28467).
Disclosure
D.R. Michael has his salary paid for by British Heart Foundation grant PG/10/55/28467. T.G. Ashlin: none. M.L. Buckley: none. D.P. Ramji received British Heart Foundation project grants that included salaries for staff and expenses for consumables for research carried out on various aspects of atherosclerosis in the laboratory, including the LXRs. These are acknowledged in the Acknowledgment section. There was no direct payment for the submitted work and no one gains from this.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Michael, D.R., Ashlin, T.G., Buckley, M.L. et al. Liver X Receptors, Atherosclerosis and Inflammation. Curr Atheroscler Rep 14, 284–293 (2012). https://doi.org/10.1007/s11883-012-0239-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11883-012-0239-y
Keywords
- Atherosclerosis
- Apolipoprotein E
- ATP-binding cassette transporters
- Cholesterol efflux
- Cholesterol homeostasis
- Cytokines
- Derepression
- Endothelial cells
- Foam cells
- Hepatic steatosis
- Hypertriglyceridemia
- Inflammation
- Ligand
- Lipopolysaccharide
- Liver X receptors
- Low density lipoproteins
- Macrophages
- Nuclear factor kappa B
- Oxysterols
- Retinoid X receptor
- Reverse cholesterol transport
- Smooth muscle cells
- SUMOylation
- Transcription factors
- Transrepression