Abstract
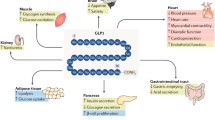
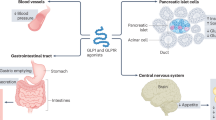
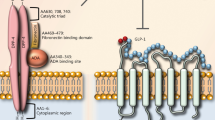
Cardiovascular disease is a leading cause of death in the United States and across the world, and better therapies are constantly being sought to improve patient outcomes. Recent studies have brought our attention to the mechanisms of glucagon-like peptide 1 (GLP-1). Not only does it demonstrate beneficial effects in regard to cardiovascular risk factors (ie, diabetes, lipid management, and weight control), but it also has been shown in animal studies to have positive cardiac effects irrespective of its effects on glucose control and weight loss. This review discusses the biology of GLP-1 and its effects on cardiovascular risk factors, and it also elaborates on the positive direct cardiovascular outcomes of GLP-1 in animal studies.
Similar content being viewed by others
References and Recommended Reading
Muller C, Neumann FJ, Ferenc M, et al.: Impact of diabetes mellitus on long-term outcome after unstable angina and non-ST-segment elevation myocardial infarction treated with a very early invasive strategy. Diabetologia 2004, 47:1188–1195.
Jung RT: Obesity as a disease. Br Med Bull 1997, 53:307–321.
Johnson JA, Majumdar SR, Simpson SH, Toth EL: Decreased mortality associated with the use of metformin compared with sulfonylurea monotherapy in type 2 diabetes. Diabetes Care 2002, 25:2244–2248.
Johnson JA, Simpson SH, Toth EL, Majumdar SR: Reduced cardiovascular morbidity and mortality associated with metformin use in subjects with Type 2 diabetes. Diabetes Med 2005, 22:497–502.
Evans JM, Ogston SA, Emslie-Smith A, Morris AD: Risk of mortality and adverse cardiovascular outcomes in type 2 diabetes: a comparison of patients treated with sulfonylureas and metformin. Diabetologia 2006, 49:930–936.
Simpson SH, Majumdar SR, Tsuyuki RT, et al.: Doseresponse relation between sulfonylurea drugs and mortality in type 2 diabetes mellitus: a population-based cohort study. Can Med Assoc J 2006, 174:169–174.
Drucker DJ, Nauck MA: The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006, 368:1696–1705.
Bullock BP, Heller RS, Habener JF: Tissue distribution of messenger ribonucleic acid encoding the rat glucagon-like peptide-1 receptor. Endocrinology 1996, 137:2968–2978.
Brubaker PL, Drucker DJ: Structure-function of the glucagon receptor family of G protein-coupled receptors: the glucagon, GIP, GLP-1, and GLP-2 receptors. Receptors Channels 2002, 8:179–188.
Ban K, Noyan-Ashraf MH, Hoefer J, et al.: Cardioprotective and vasodilatory actions of glucagon-like peptide 1 receptor are mediated through both glucagon-like peptide 1 receptor-dependent and -independent pathways. Circulation 2008, 117:2340–2350.
Perry T, Greig NH: Enhancing central nervous system endogenous GLP-1 receptor pathways for intervention in Alzheimer’s disease. Curr Alzheimer Res 2005, 2:377–385.
Nikolaidis LA, Doverspike A, Hentosz T, et al.: Glucagon-like peptide-1 limits myocardial stunning following brief coronary occlusion and reperfusion in conscious canines. J Pharmacol Exp Ther 2005, 312:303–308.
Nikolaidis LA, Elahi D, Hentosz T, et al.: Recombinant glucagon-like peptide-1 increases myocardial glucose uptake and improves left ventricular performance in conscious dogs with pacing-induced dilated cardiomyopathy. Circulation 2004, 110:955–961.
Nikolaidis LA, Mankad S, Sokos GG, et al.: Effects of glucagon-like peptide-1 in patients with acute myocardial infarction and left ventricular dysfunction after successful reperfusion. Circulation 2004, 109:962–965.
Bose AK, Mocanu MM, Carr RD, et al.: Glucagon-like peptide 1 can directly protect the heart against ischemia/reperfusion injury. Diabetes 2005, 54:146–151.
Bose AK, Mocanu MM, Carr RD, Yellon DM: Glucagon like peptide-1 is protective against myocardial ischemia/reperfusion injury when given either as a preconditioning mimetic or at reperfusion in an isolated rat heart model. Cardiovasc Drugs Ther 2005, 19:9–11.
Mayo KE, Miller LJ, Bataille D, et al.: International Union of Pharmacology. XXXV. The glucagon receptor family. Pharmacol Rev 2003, 55:167–194.
Deacon CF, Nauck MA, Toft-Nielsen M, et al.: Both subcutaneously and intravenously administered glucagon-like peptide I are rapidly degraded from the NH2-terminus in type II diabetic patients and in healthy subjects. Diabetes 1995, 44:1126–1131.
Avenell A, Broom J, Brown TJ, et al.: Systematic review of the long-term effects and economic consequences of treatments for obesity and implications for health improvement. Health Technol Assess 2004, 8:iii–iv, 1–182.
Okerson T, Yan P, Stonehouse A, et al.: Exenatide improved systolic blood pressure compared to insulin or placebo in patients with type 2 diabetes [abstract 877]. Presented at the 44th Annual meeting of the EASD. Rome, Italy; September 7–11, 2008.
Colagiuri S, Frid A, Zdravkovic M, et al.: Liraglutide, a human GLP-1 analogue, reduces systolic blood pressure in subjects with type 2 diabetes in subjects with type 2 diabetes [abstract 899]. Presented at the 44th Annual meeting of the EASD. Rome, Italy; September 7–11, 2008.
Drucker DJ, Buse JB, Taylor K, et al.: Exenatide once weekly versus twice daily for the treatment of type 2 diabetes: a randomised, open-label, non-inferiority study. Lancet 2008 (in press).
Klonoff DC, Buse JB, Nielsen LL, et al.: Exenatide effects on diabetes, obesity, cardiovascular risk factors and hepatic biomarkers in patients with type 2 diabetes treated for at least 3 years. Curr Med Res Opin 2008, 24:275–286.
Boquist S, Ruotolo G, Tang R, et al.: Alimentary lipemia, postprandial triglyceride-rich lipoproteins, and common carotid intima-media thickness in healthy, middle-aged men. Circulation 1999, 100:723–728.
Karpe F, Steiner G, Uffelman K, et al.: Postprandial lipoproteins and progression of coronary atherosclerosis. Atherosclerosis 1994, 106:83–97.
Weintraub MS, Grosskopf I, Rassin T, et al.: Clearance of chylomicron remnants in normolipidaemic patients with coronary artery disease: case control study over three years. BMJ 1996, 312:935–939.
Bunck MC, Corner A, Diamant M, et al.: Exenatide improves postprandial hyperglycaemia and dyslipidaemia in metformin treated patients with type 2 diabetes [abstract 879]. Presented at the 44th Annual meeting of the EASD. Rome, Italy; September 7–11, 2008.
Dear AE, Bin Liu H, Knudsen LB, Simpson RW: Liraglutide inhibits hyperglycemia-induced expression of PAI-1 and VCAM-1 in vascular endothelial cells. [Abstract 503]. Presented at 68th Scientific session of the American Diabetes Association. San Francisco, CA; June 6–10, 2008.
Kendall DM, Bhole D, Guan X, et al.: Exenatide treatment for 82 weeks reduced C-reactive protein, HbA1c, and body weight in patients with type 2 diabetes mellitus. Presented at 42nd Congress of EASD. Copenhagen, Denmark; September 14–17, 2006.
Nystrom T, Gutniak MK, Zhang Q, et al.: Effects of glucagon-like peptide-1 on endothelial function in type 2 diabetes patients with stable coronary artery disease. Am J Physiol Endocrinol Metab 2004, 287:E1209–E1215.
Basu A, Charkoudian N, Schrage W, et al.: Beneficial effects of GLP-1 on endothelial function in humans: dampening by glyburide but not by glimepiride. Am J Physiol Endocrinol Metab 2007, 293:E1289–E1295.
Mootha VK, Arai AE, Balaban RS: Maximum oxidative phosphorylation capacity of the mammalian heart. Am J Physiol 1997, 272(2 Pt 2):H769–H775.
Taegtmeyer H: Switching metabolic genes to build a better heart. Circulation 2002, 106:2043–2045.
Stanley WC, Recchia FA, Lopaschuk GD: Myocardial substrate metabolism in the normal and failing heart. Physiol Rev 2005, 85:1093–1129.
Doenst T, Bugger H, Schwarzer M, et al.: Three good reasons for heart surgeons to understand cardiac metabolism. Eur J Cardiothorac Surg 2008, 33:862–871.
Kavianipour M, Ehlers MR, Malmberg K, et al.: Glucagon- like peptide-1 (7–36) amide prevents the accumulation of pyruvate and lactate in the ischemic and non-ischemic porcine myocardium. Peptides 2003, 24:569–578.
Zhao T, Parikh P, Bhashyam S, et al.: Direct effects of glucagon-like peptide-1 on myocardial contractility and glucose uptake in normal and postischemic isolated rat hearts. J Pharmacol Exp Ther 2006, 317:1106–1113.
Poornima I, Brown S, Bhashyam S, et al.: Chronic glucagon-like peptide-1 (GLP-1) infusion sustains LV systolic function and prolongs survival in the spontaneously hypertensive-heart failure prone rat. Circ Heart Fail 2008, 1:153–160.
Sokos GG, Bolukoglu H, German J, et al.: Effect of glucagon- like peptide-1 (GLP-1) on glycemic control and left ventricular function in patients undergoing coronary artery bypass grafting. Am J Cardiol 2007, 100:824–829.
Garg S, Hofstra L, Reutelingsperger C, Narula J: Apoptosis as a therapeutic target in acutely ischemic myocardium. Curr Opin Cardiol 2003, 18:372–377.
Rezkalla SH, Kloner RA: Ischemic preconditioning and preinfarction angina in the clinical arena. Nat Clin Pract Cardiovasc Med 2004, 1:96–102.
Yellon DM, Hausenloy DJ: Realizing the clinical potential of ischemic preconditioning and postconditioning. Nat Clin Pract Cardiovasc Med 2005, 2:568–575.
Terzic A, Jahangir A, Kurachi Y: Cardiac ATP-sensitive K+ channels: regulation by intracellular nucleotides and K+ channel-opening drugs. Am J Physiol 1995, 269(3 Pt 1):C525–C545.
Garratt KN, Brady PA, Hassinger NL, et al.: Sulfonylurea drugs increase early mortality in patients with diabetes mellitus after direct angioplasty for acute myocardial infarction. J Am Coll Cardiol 1999, 33:119–124.
Riddle MC: Sulfonylureas differ in effects on ischemic preconditioning—is it time to retire glyburide? [editorial]. J Clin Endocrinol Metab 2003, 88:528–530.
Feuerstein G, Yue TL, Ma X, Ruffolo RR: Novel mechanisms in the treatment of heart failure: inhibition of oxygen radicals and apoptosis by carvedilol. Prog Cardiovasc Dis 1998, 41(1 Suppl 1):17–24.
Goussev A, Sharov VG, Shimoyama H, et al.: Effects of ACE inhibition on cardiomyocyte apoptosis in dogs with heart failure. Am J Physiol 1998, 275(2 Pt 2):H626–H631.
Li Y, Hansotia T, Yusta B, et al.: Glucagon-like peptide-1 receptor signaling modulates beta cell apoptosis. J Biol Chem 2003, 278:471–478.
Buteau J, Foisy S, Rhodes CJ, et al.: Protein kinase Czeta activation mediates glucagon-like peptide-1-induced pancreatic beta-cell proliferation. Diabetes 2001, 50:2237–2243.
Urusova IA, Farilla L, Hui H, et al.: GLP-1 inhibition of pancreatic islet cell apoptosis. Trends Endocrinol Metab 2004, 15:27–33.
Hui H, Nourparvar A, Zhao X, Perfetti R: Glucagon-like peptide-1 inhibits apoptosis of insulin-secreting cells via a cyclic 5′-adenosine monophosphate-dependent protein kinase A- and a phosphatidylinositol 3-kinase-dependent pathway. Endocrinology 2003, 144:1444–1455.
Dokken BB, Labonte LR, Davis-Gorman G, Mcdonagh PF: Postconditioning with GLP-1 in vivo decreases myocardial infarct size in rats [abstract 58-OR]. Presented at the 67th Scientific sessions of the American Diabetes Association. Chicago, IL; June 22–26, 2008.
Sonne DP, Engstrom T, Treiman M: Protective effects of GLP-1 analogues exendin-4 and GLP-1(9–36) amide against ischemia-reperfusion injury in rat heart. Regul Pept 2008, 146:243–249.
Noyan-Ashraf MH, Ban K, Sadi A, et al.: The GLP-1R agonist liraglutide protects cardiomyocytes and improves survival and cardiac function after experimental murine infarction [abstract 190-OR]. Presented at the 68th Scientific sessions of the American Diabetes Associaton. San Francisco, CA; June 6–10, 2008.
Kelley DE, Goodpaster B, Wing RR, Simoneau JA: Skeletal muscle fatty acid metabolism in association with insulin resistance, obesity, and weight loss. Am J Physiol 1999, 277:E1130–E1141.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sulistio, M., Carothers, C., Mangat, M. et al. GLP-1 agonist-based therapies: An emerging new class of antidiabetic drug with potential cardioprotective effects. Curr Atheroscler Rep 11, 93–99 (2009). https://doi.org/10.1007/s11883-009-0015-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11883-009-0015-9