Abstract
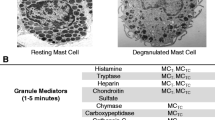
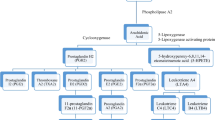
Mast cell activation syndrome (MCAS) is a condition with signs and symptoms involving the skin, gastrointestinal, cardiovascular, respiratory, and neurologic systems. It can be classified into primary, secondary, and idiopathic. Earlier proposed criteria for the diagnosis of MCAS included episodic symptoms consistent with mast cell mediator release affecting two or more organ systems with urticaria, angioedema, flushing, nausea, vomiting, diarrhea, abdominal cramping, hypotensive syncope or near syncope, tachycardia, wheezing, conjunctival injection, pruritus, and nasal stuffiness. Other criteria included a decrease in the frequency, severity, or resolution of symptoms with anti-mediator therapy including H1 and H2histamine receptor antagonists, anti-leukotrienes, or mast cell stabilizers. Laboratory data that support the diagnosis include an increase of a validated urinary or serum marker of mast cell activation (MCA), namely the documentation of an increase of the marker above the patient’s baseline value during symptomatic periods on more than two occasions, or baseline serum tryptase levels that are persistently above 15 ng/ml, or documentation of an increase of the tryptase level above baseline value on one occasion. Less specific assays are 24-h urine histamine metabolites, PGD2 (Prostaglandin D2) or its metabolite, 11-β-prostaglandin F2 alpha. A recent global definition, criteria, and classification include typical clinical symptoms, a substantial transient increase in serum total tryptase level or an increase in other mast cell derived mediators, such as histamine or PGD2 or their urinary metabolites, and a response of clinical symptoms to agents that attenuate the production or activities of mast cell mediators.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Kinet J-P. The high affinity receptor for IgE. Curr Opin Immunol. 1990;2:499–505.
Cambier JC. Antigen and Fc receptor signaling: the awesome power of the immunoreceptor tyrosine based activation motif (ITAM). J Immunol. 1995;155:3281–5.
Fukamachi H, Kawakami Y, Takei M, et al. Association of protein tyrosine kinase with phospholipase C-γ1 in bone marrow-derived mouse mast cells. Proc Natl Acad Sci USA. 1992;89:9524–8.
Bischoff SC, Dahinden CA. c-Kit ligand: a unique potentiator of mediator release by human lung mast cells. J Exp Med. 1992;175:237–44.
Galli SJ, Tsai M, Wershil BK. The c-kit receptor, stem cell factor, and mast cells. What each is teaching us about the others. Am J Pathol. 1993;142(4):965–74.
Galli SJ, Zsebo KM, Geissler EN. The kit ligand, stem cell factor. Adv Immunol. 1994;55:1–96.
Lukacs NW, Strieter RM, Lincoln PM, et al. Stem cell factor (c-kit ligand) influences eosinophil recruitment and histamine levels in allergic airway inflammation. J Immunol. 1996;156(10):3945–51.
Bingham III CO, Austen KF. Mast-cell responses in the development of asthma. J Allergy Clin Immunol. 2000;105(2 Pt 2):S527–34.
Iemura A, Tsai M, Ando A, et al. The c-kit ligand, stem cell factor, promotes mast cell survival by suppressing apoptosis. Am J Pathol. 1994;144(2):321–8.
Meninger CJ, Yano H, Rottapel R, et al. The c-kit receptor ligand functions as a mast cell chemoattractant. Blood. 1992;4:958–63.
Ito T, Smrž D, Jung MY, et al. Stem cell factor programs the mast cell activation phenotype. J Immunol. 2012;188(11):5428–37.
Gilfillan AM, Rivera J. The tyrosine kinase network regulating mast cell activation. J Immunol Rev Immunol Rev. 2009;228(1):149–69.
Bai Y, Bandara G, Ching Chan E. Targeting the KIT activating switch control pocket: a novel mechanism to inhibit neoplastic mast cell proliferation and mast cell activation. Leukemia. 2012. doi:10.1038.
Bax HJ, Keeble AH, Gould HJ. Cytokinergic IgE action in mast cell activation. Front Immunol Inflamm Vol; 2012 (3) 8
•• Akin C, Valent P, Metcalfe DD. Mast cell activation syndrome: proposed diagnostic criteria. J Allergy Clin Immunol. 2010;126:1099–104. This excellent paper explored and proposed criteria for the diagnosis of the mast cell activation syndrome. The proposed criteria was discussed in the context of other disorders involving mast cells or with similar presentations and as a basis for further scientific study and validation.
Shibao C, Arzubiaga C, Roberts 2nd LJ, et al. Hyperadrenergic postural tachycardia syndrome in mast cell activation disorders. Hypertension. 2005;45(3):385–90.
Frieri M. Mastocytosis. In: Massoud Mahmoud editor. Challenging Cases in Allergy and Immunology, Chapter 14. Humana Press; 2009. pp 201–212.
Akin C, Scott LM, Kocabas CN, et al. Demonstration of an aberrant mast-cell population with clonal markers in a subset of patients with “idiopathic” anaphylaxis. Blood. 2007;110:2331–3.
Koterba AP, Akin C. Differences in the clinical presentation of anaphylaxis in patients with indolent systemic mastocytosis (ISM) versus idiopathic anaphylaxis (IA). J Allergy Clin Immunol. 2008;121:S68. abstract.
An Updated Practice Parameter. In: Lieberman P et al., editor. Idiopathic Anaphylaxis. The Diagnosis and Management of Anaphylaxis. 2005;115; 3:S483–S523.
• Frieri M. Anaphylaxis. Chapter 68. In: Manual of critical care. Philadelphia: American College of Physicians; 2009. p. 721–30. This chapter reviewed the etiology, incidence, unusual manifestations, differential diagnosis, pathogenesis, prevention and treatment of anaphylaxis with a comprehensive table of mast cell and basophil mediators and clinical findings in anaphylaxis and anaphylactoid reactions.
Frieri M, Alling DW, Metcalfe DD. Comparison of the therapeutic efficacy of cromolyn sodium with that of combined chlorpheniramine and cimetidine in systemic mastocytosis. Results of a double-blind clinical trial. Am J Med. 1985;78:9–14.
Metcalfe DD. Differential diagnosis of the patient with unexplained flushing/anaphylaxis. Allergy Asthma Proc. 2000;21(1):21–4.
Alvarez-Twose I, González de Olano D, Sánchez-Muñoz L, et al. Clinical, biological, and molecular characteristics of clonal mast cell disorders presenting with systemic mast cell activation symptoms. J Allergy Clin Immunol. 2010;125(6):1269–78.
Molderings G, Brettner S, Homann J, Afrin L. Mast cell activation disease: a concise practical guide for diagnostic work up and therapeutic options. J Hematol Oncol. 2011;4:10.
Hamilton MJ, Hornick JL, Akin C, et al. A newly recognized disorder with systemic clinical manifestations. J Allergy Clin Immunol. 2011;128(1):147–52.
•• Valent P, Akin C, Arock M, et al. Definitions, criteria & global classification of MC disorders with special reference to MCAS: a consensus proposal. Int Arch Allergy Immunol. 2012;157(3):215–25. This excellent paper proposed a global unifying classification of all MC disorders and pathologic MC reactions which included three types of 'MCA syndromes' (MCASs), namely primary MCAS, secondary MCAS, and idiopathic MCAS. It assisted in the identification and diagnosis of patients with MCAS, and in avoiding misdiagnoses or over interpretation of clinical symptoms in daily practice.
Pardanani A. Systemic mastocytosis in adults: 2012 update on diagnosis, risk stratification, and management. Am J Hematol. 2012;87(4):401–11.
Frieri M, Linn N, Schweitzer M, Angadi C, et al. Lymphadenopathic mastocytosis with eosinophilia and biclonal gammopathy. J Allergy Clin Immunol. 1990;86:l26–l132.
Gotlib J, Akin C. Mast cells and eosinophils in mastocytosis, chronic eosinophilic leukemia, and non-clonal disorders. Semin Hematol. 2012;49(2):128–37.
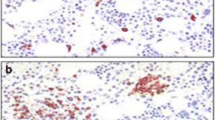
Patel R, Celestin J, Frieri M. An unusual case of mast cell activation syndrome. J Allergy Clin Immunol, abs #475, 2012.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Frieri, M., Patel, R. & Celestin, J. Mast Cell Activation Syndrome: A Review. Curr Allergy Asthma Rep 13, 27–32 (2013). https://doi.org/10.1007/s11882-012-0322-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11882-012-0322-z