Abstract
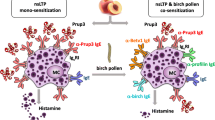
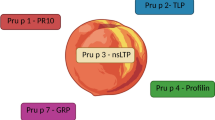
Nonspecific lipid transfer proteins (LTPs) are important allergens in fruits, vegetables, nuts, pollen, and latex. Despite their wide distribution throughout the plant kingdom, their clinical relevance is largely confined to the Mediterranean area. As they can sensitize via the gastrointestinal tract, LPTs are considered true food allergens, and IgE reactivity to LTPs is often associated with severe systemic symptoms. Although Pru p 3 represents the predominant LTP in terms of patients’ IgE recognition, the contribution of pollen LTPs in primary sensitization cannot be ruled out. Due to structural homology, LTPs from different allergen sources are generally IgE cross-reactive. However, sensitization profiles among allergic patients are extremely heterogeneous, and individual cross-reactivity patterns can be restricted to a single LTP or encompass many different LTPs. Molecule-based approaches in allergy research and diagnosis are important for better understanding of LTP allergy and could assist clinicians with providing adequate patient-tailored advice.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Finn RD, Tate J, Mistry J, et al.: The Pfam protein families database. Nucleic Acids Res 2008, 36:D281–D288.
Radauer C, Bublin M, Wagner S, et al.: Allergens are distributed into few protein families and possess a restricted number of biochemical functions. J Allergy Clin Immunol 2008, 121:847.e7–852.e7.
Hauser M, Roulias A, Ferreira F, et al.: Panallergens and their impact on the allergic patient. Allergy Asthma Clin Immunol 2010, 6:1.
Hoffmann-Sommergruber K: Pathogenesis-related (PR)-proteins identified as allergens. Biochem Soc Trans 2002, 30:930–935.
• Salcedo G, Sanchez-Monge R, Barber D, et al.: Plant non-specific lipid transfer proteins: an interface between plant defence and human allergy. Biochim Biophys Acta 2007, 1771:781–791. This review article provides detailed information on structural features and biological function of plant-nonspecific LTPs.
Yeats TH, Rose JK: The biochemistry and biology of extracellular plant lipid-transfer proteins (LTPs). Protein Sci 2008, 17:191–198.
Gonzalez-Rioja R, Asturias JA, Martinez A, et al.: Par j 1 and Par j 2, the two major allergens in Parietaria judaica, bind preferentially to monoacylated negative lipids. FEBS J 2009, 276:1762–1775.
Douliez J, Michon T, Elmorjani K, et al.: Structure, biological and technological functions of lipid transfer proteins and indolines, the major lipid binding proteins from cereal kernels. J Cereal Sci 2000, 32:1–20.
Regente MC, Giudici AM, Villalain J, et al.: The cytotoxic properties of a plant lipid transfer protein involve membrane permeabilization of target cells. Lett Appl Microbiol 2005, 40:183–189.
Barber D, de la Torre F, Feo F, et al.: Understanding patient sensitization profiles in complex pollen areas: a molecular epidemiological study. Allergy 2008, 63:1550–1558.
Scala E, Alessandri C, Bernardi ML, et al.: Cross-sectional survey on immunoglobulin E reactivity in 23 077 subjects using an allergenic molecule-based microarray detection system. Clin Exp Allergy 2010, 40:911–921.
Fernandez-Rivas M, Bolhaar S, Gonzalez-Mancebo E, et al.: Apple allergy across Europe: how allergen sensitization profiles determine the clinical expression of allergies to plant foods. J Allergy Clin Immunol 2006, 118:481–488.
Borges JP, Jauneau A, Brule C, et al.: The lipid transfer proteins (LTP) essentially concentrate in the skin of Rosaceae fruits as cell surface exposed allergens. Plant Physiol Biochem 2006, 44:535–542.
Sancho AI, van Ree R, van Leeuwen A, et al.: Measurement of lipid transfer protein in 88 apple cultivars. Int Arch Allergy Immunol 2008, 146:19–26.
Lauer I, Dueringer N, Pokoj S, et al.: The non-specific lipid transfer protein, Ara h 9, is an important allergen in peanut. Clin Exp Allergy 2009, 39:1427–1437.
Gaier S, Oberhuber C, Hemmer W, et al.: Pru p 3 as a marker for symptom severity for patients with peach allergy in a birch pollen environment. J Allergy Clin Immunol 2009, 124:166–167.
Flinterman AE, Akkerdaas JH, den Hartog Jager CF, et al.: Lipid transfer protein-linked hazelnut allergy in children from a non-Mediterranean birch-endemic area. J Allergy Clin Immunol 2008, 121:423.e2–428.e2.
Asturias JA, Gomez-Bayon N, Eseverri JL, et al.: Par j 1 and Par j 2, the major allergens from Parietaria judaica pollen, have similar immunoglobulin E epitopes. Clin Exp Allergy 2003, 33:518–524.
Lauer I, Miguel-Moncin MS, Abel T, et al.: Identification of a plane pollen lipid transfer protein (Pla a 3) and its immunological relation to the peach lipid-transfer protein, Pru p 3. Clin Exp Allergy 2007, 37:261–269.
Lombardero M, Garcia-Selles FJ, Polo F, et al.: Prevalence of sensitization to Artemisia allergens Art v 1, Art v 3 and Art v 60 kDa. Cross-reactivity among Art v 3 and other relevant lipid-transfer protein allergens. Clin Exp Allergy 2004, 34:1415–1421.
Ciardiello MA, Palazzo P, Bernardi ML, et al.: Biochemical, immunological and clinical characterization of a cross-reactive nonspecific lipid transfer protein 1 from mulberry. Allergy 2010, 65:597–605.
•• Zuidmeer L, van Ree R: Lipid transfer protein allergy: primary food allergy or pollen/food syndrome in some cases. Curr Opin Allergy Clin Immunol 2007, 7:269–273. This review gives an excellent overview on the impact of protein stability and localization of LTPs related to sensitization and allergic reactions.
Pastorello EA, Pravettoni V, Farioli L, et al.: Hypersensitivity to mugwort (Artemisia vulgaris) in patients with peach allergy is due to a common lipid transfer protein allergen and is often without clinical expression. J Allergy Clin Immunol 2002, 110:310–317.
Reuter A, Lidholm J, Andersson K, et al.: A critical assessment of allergen component-based in vitro diagnosis in cherry allergy across Europe. Clin Exp Allergy 2006, 36:815–823.
Asero R, Mistrello G, Amato S, et al.: Peach fuzz contains large amounts of lipid transfer protein: is this the cause of the high prevalence of sensitization to LTP in Mediterranean countries? Eur Ann Allergy Clin Immunol 2006, 38:118–121.
Rihs HP, Rueff F, Lundberg M, et al.: Relevance of the recombinant lipid transfer protein of Hevea brasiliensis: IgE-binding reactivity in fruit-allergic adults. Ann Allergy Asthma Immunol 2006, 97:643–649.
• Schulten V, Radakovics A, Hartz C, et al.: Characterization of the allergic T-cell response to Pru p 3, the nonspecific lipid transfer protein in peach. J Allergy Clin Immunol 2009, 124:100–107. Within this study, T-cell epitopes of Pru p 3 were mapped, and it was further shown that the T-cell response is dominated by T-helper type 2 cells, which are presumably primed in the gut.
Tordesillas L, Cuesta-Herranz J, Gonzalez-Munoz M, et al.: T-cell epitopes of the major peach allergen, Pru p 3: identification and differential T-cell response of peach-allergic and non-allergic subjects. Mol Immunol 2009, 46:722–728.
Bohle B, Zwolfer B, Heratizadeh A, et al.: Cooking birch pollen-related food: divergent consequences for IgE- and T cell-mediated reactivity in vitro and in vivo. J Allergy Clin Immunol 2006, 118:242–249.
Garcia-Casado G, Pacios LF, Diaz-Perales A, et al.: Identification of IgE-binding epitopes of the major peach allergen Pru p 3. J Allergy Clin Immunol 2003, 112:599–605.
Borges JP, Barre A, Culerrier R, et al.: Lipid transfer proteins from Rosaceae fruits share consensus epitopes responsible for their IgE-binding cross-reactivity. Biochem Biophys Res Commun 2008, 365:685–690.
Borges JP, Barre A, Culerrier R, et al.: How reliable is the structural prediction of IgE-binding epitopes of allergens? The case study of plant lipid transfer proteins. Biochimie 2007, 89:83–91.
Pacios LF, Tordesillas L, Cuesta-Herranz J, et al.: Mimotope mapping as a complementary strategy to define allergen IgE-epitopes: peach Pru p 3 allergen as a model. Mol Immunol 2008, 45:2269–2276.
Tordesillas L, Pacios LF, Palacin A, et al.: Molecular basis of allergen cross-reactivity: non-specific lipid transfer proteins from wheat flour and peach fruit as models. Mol Immunol 2009, 47:534–540.
Sancho AI, Rigby NM, Zuidmeer L, et al.: The effect of thermal processing on the IgE reactivity of the non-specific lipid transfer protein from apple, Mal d 3. Allergy 2005, 60:1262–1268.
Gaier S, Marsh J, Oberhuber C, et al.: Purification and structural stability of the peach allergens Pru p 1 and Pru p 3. Mol Nutr Food Res 2008, 52(Suppl 2):S220–S229.
Bonura A, Amoroso S, Locorotondo G, et al.: Hypoallergenic variants of the Parietaria judaica major allergen Par j 1: a member of the non-specific lipid transfer protein plant family. Int Arch Allergy Immunol 2001, 126:32–40.
Garcia-Selles FJ, Diaz-Perales A, Sanchez-Monge R, et al.: Patterns of reactivity to lipid transfer proteins of plant foods and Artemisia pollen: an in vivo study. Int Arch Allergy Immunol 2002, 128:115–122.
Sirvent S, Palomares O, Vereda A, et al.: nsLTP and profilin are allergens in mustard seeds: cloning, sequencing and recombinant production of Sin a 3 and Sin a 4. Clin Exp Allergy 2009, 39:1929–1936.
Hartz C, San Miguel-Moncin Mdel M, Cistero-Bahima A, et al.: Molecular characterisation of Lac s 1, the major allergen from lettuce (Lactuca sativa). Mol Immunol 2007, 44:2820–2830.
Zoccatelli G, Pokoj S, Foetisch K, et al.: Identification and characterization of the major allergen of green bean (Phaseolus vulgaris) as a non-specific lipid transfer protein (Pha v 3). Mol Immunol 2010, 47:1561–1568.
Palacin A, Cumplido J, Figueroa J, et al.: Cabbage lipid transfer protein Bra o 3 is a major allergen responsible for cross-reactivity between plant foods and pollens. J Allergy Clin Immunol 2006, 117:1423–1429.
Zuidmeer L, Salentijn E, Rivas MF, et al.: The role of profilin and lipid transfer protein in strawberry allergy in the Mediterranean area. Clin Exp Allergy 2006, 36:666–675.
Gadermaier G, Harrer A, Girbl T, et al.: Isoform identification and characterization of Art v 3, the lipid-transfer protein of mugwort pollen. Mol Immunol 2009, 46:1919–1924.
Pravettoni V, Primavesi L, Farioli L, et al.: Tomato allergy: detection of IgE-binding lipid transfer proteins in tomato derivatives and in fresh tomato peel, pulp, and seeds. J Agric Food Chem 2009, 57:10749–10754.
Vassilopoulou E, Rigby N, Moreno FJ, et al.: Effect of in vitro gastric and duodenal digestion on the allergenicity of grape lipid transfer protein. J Allergy Clin Immunol 2006, 118:473–480.
D’Amato G, Ruffilli A, Sacerdoti G, et al.: Parietaria pollinosis: a review. Allergy 1992, 47:443–449.
Enrique E, Pineda F, Malek T, et al.: Sublingual immunotherapy for hazelnut food allergy: a randomized, double-blind, placebo-controlled study with a standardized hazelnut extract. J Allergy Clin Immunol 2005, 116:1073–1079.
•• Fernandez-Rivas M, Garrido Fernandez S, Nadal JA, et al.: Randomized double-blind, placebo-controlled trial of sublingual immunotherapy with a Pru p 3 quantified peach extract. Allergy 2009, 64:876–883. This article describes the first exploratory clinical trial for sublingual immunotherapy using a peach extract standardized on Pru p 3, providing promising therapeutic options for the treatment of food allergy.
Asero R, Mistrello G, Roncarolo D, et al.: Detection of some safe plant-derived foods for LTP-allergic patients. Int Arch Allergy Immunol 2007, 144:57–63.
Acknowledgment
The authors contributing to this work were supported by the Christian Doppler Research Association and Biomay AG, Vienna, Austria.
Disclosure
Dr. Ferreira has served as a consultant for Biomay AG. No other potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Egger, M., Hauser, M., Mari, A. et al. The Role of Lipid Transfer Proteins in Allergic Diseases. Curr Allergy Asthma Rep 10, 326–335 (2010). https://doi.org/10.1007/s11882-010-0128-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11882-010-0128-9