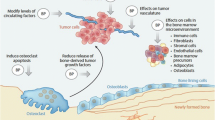
Opinion statement
Bisphosphonates have emerged as an important tool in the supportive care of women with early breast cancer. Whereas traditionally, these drugs have been part of the treatment of osseous metastasis, the key role of bisphosphonates in preserving bone health in patients with early breast cancer cannot be overemphasised. Currently the most established use of bisphosphonates in early breast cancer patients is in women receiving hormonal blockade, mostly aromatase inhibitors (AI), with concomitant osteopenia. To that end, it is recommended that every woman undergo a Dual Energy X-Ray absorptiometry (DEXA) scan before commencement of an AI and annually during the treatment duration. In addition, unless contraindicated, all women should receive calcium and Vitamin D supplementation. The use of bisphosphonates as part of the adjuvant therapy strategy, regardless of baseline bone density condition, has produced thought-provoking results, although this is not yet considered standard clinical practise.

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bloomfield DJ. Should bisphosphonates be part of the standard therapy of patients with multiple myeloma or bone metastases from other cancers? an evidence- based review. J Clin Oncol. 1998;16:1218–25.
Saad F, Gleason DM, Murray R, et al. A randomised placebo- controlled trial of zoledronic acid in patients with metastatic prostate carcinoma. J Natl Canc Inst. 2002;94:1458–68.
Rosen LS, Gordon D, Tchekmedyian S, et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: a phase III, double-blind, randomized trial. The Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol. 2003;21:3150–7.
Van Poznak C, Sauter NP, et al. Clinical management of osteoporosis in women with a history of breast carcinoma. Cancer. 2005;104(3):443.
Lacey DL, Timms E, Tan HL, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93:165–76.
Guise TA, Yin JJ, Taylor SD, et al. Evidence for a causal role of parathyroid hormone –related protein in the pathogenesis of human breast cancer-mediated osteolysis. J Clin Investig. 1996;98:1544–9.
Rodan GA, Fleisch HA, et al. Bisphosphonates: mechanisms of action. J Clin Investig. 1996;97(12):2692.
Stresing V, Daubine F, Benzaid I, et al. Bisphosphonates in cancer therapy. Cancer Lett. 2007;8;257(1):16–35
Baum M, Buzdar A, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early-stage breast cancer: results of the ATAC (arimidex, tamoxifen alone or in combination) trial efficacy and safety update analyses. Cancer. 2003;98:1802–10.
Goss PE, Ingle JN, Martino S, et al. A randomized trial of letrozole in postmenopausal women after 5 years of tamoxifen therapy for early-stage breast cancer. N Engl J Med. 2003;349:1793–802.
Coombes RC, Hall E, Gibson LJ, et al. A randomized trial of exemestane after 2 to 3 years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med. 2004;350:1081–92.
Jakesz R, Jonat W, Gnant M, et al. Switching of postmenopausal women with endocrine-responsive early breast cancer to anastrozole after 2 years’ adjuvant tamoxifen: combined results of ABCSG trial 8 and ARNO 95 trial. Lancet. 2005;366:455–62.
The Breast International Group (BIG) 1–98 Collaborative Group. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005;353:2747–57.
Vorobiof DA, Kleeberg VR, Perez Carrion R, et al. A randomized open parallel group trial to compare the endocrine effects of oral anastrozole (arimidex) with intramuscular formestane in post menopausal women with advanced breast cancer. Ann Oncol. 1999;10:1219–55.
Eastell R, Adams JE, Coleman RE, et al. Effect of anastrazole on bone mineral density: 5 year results from the anastrazole, tamoxifen, alone or in combination trial 18233230. J Clin Oncol. 2008;26:1051–7.
Coleman RE, Banks LM, Girgis SL, et al. Skeletal effects of exemestane on bone-mineral density, bone biomarkers and fracture incidence in postmenopausal women with early breast cancer participating in the Intergroup Exemestane Study (IES): a randomised controlled study. Lancet Oncol. 2007;8:119–27.
Brufsky A, Harker G, Beck JT, et al. The effect of zoledronic acid on aromatase inhibitor associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: the Z-FAST study 5 year final follow-up. Cancer Res. 2009;69:(24 Suppl); Abstract 4083. Upfront zoledronic acid delays the onset of osteoporosis in postmenopausal women treated with aromatase inhibitors, as well as a reduction in disease free survival events.
Eidtmann H, de Boer R, Bundred N, et al. Efficacy of zoledronic acid in postmenopausal women with breast cancer receiving adjuvant letrozole: 36- month results of the ZO-FAST study. Ann Oncol. 2010:21(11):2188–2194.
Eastell R, Adams J, Clack G, et al. Long term effects of anastrozole on bone mineral density: 7 year results from the ATAC trial. Ann Oncol. 2011;22:857–862.
Van Poznak C, Hannon RA, Clark G, et al. Prevention of aromatase inhibitor-induced bone loss using risedronate. The SABRE trial. J Clin Oncol. 2010;28:967–75.
Lester J, Dodwell D, Purohit OP, et al. Prevention of anastrazole induced bone loss with monthly ibandronate during adjuvant aromatase inhibitor therapy for breast cancer. Clin Canc Res. 2008;14:6336–42.
Gnant M, Milneritsch B, Schippinger W, et al. Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N Engl J Med. 2009;360:679–691.
Gnant M, Milneritsch B, Luschin-Ebengreuth G, et al. Austrian Breast and Colorectal Cancer Study Group adjuvant endocrine therapy plus zoledronic acid in premenopausal woman with early stage breast cancer. 5 year follow up of the ABCSG-12 bone mineral substudy. Lancet Oncol. 2008;9:840–9.
Paget S et al. The distribution of secondary growths in cancer of the breast. Lancet. 1889;1:571–3.
Bussard KM, Gay CV, Mastro AM, et al. The bone microenvironment in metastasis; what is special about bone? Canc Metastasis Rev. 2008;27:41–55.
Psaila B, Lyden D, et al. The metastatic niche: adapting the foreign soil. Nat Rev Canc. 2009;9:285–93.
Diel IJ, Solomayer EF, Costa SD, et al. Reduction in new metastases in breast cancer with adjuvant clodronate treatment. N Engl J Med. 1998;339:357–63.
Powles T, Paterson A, McCloskey E, et al. Reduction in bone relapse and improved survival with oral clodronate for adjuvant treatment of operable breast cancer. J Clin Oncol. 2002;20;32:19–3224
Saarto T, Vehmanen L, Virkkunen P, et al. Ten-year follow up of a randomised controlled trial of adjuvant clodronate treatment in node-positive breast cancer patients. Acta Oncol. 2004;43:650–6.
Rack B, Juckstock J, Genss EM, et al. Effect of zoledronate on persisting isolated tumour cells in patients with early breast cancer. Anticancer Res. 2010;30:1807–13.
Greenberg S, Park J, Melisko M, et al. Effect of adjuvant zoledronic acid on disseminated tumor cells in the bone marrow of women with early-stage breast cancer: updated results. J Clin Oncol. 2010;28 suppl 114s:Abstract 1002
Aft R, Naughton M, Trinkaus K, et al. Effect of zoledronic acid on disseminated tumor cells in women with locally advanced breast cancer, an open label, randomised, phase 2 trial. Lancet Oncol. 2010;11:421–8.
Coleman RE, Thorpe HC, Cameron D, et al. Adjuvant treatment with zoledronic acid in stage II/III breast cancer. The AZURE trial (BIG01/04) SABCS. 2010 abstract S4–5. This study tested zoledronic acid in the postmenopausal women with stage II–III breast cancer, in addition to hormonal blockade following adjuvant chemotherapy, and did not show benefit to the addition of zoledronic acid. However, a subset analysis suggests that women who had been menopausal for more than 5 years seemed to derive more benefit from the addition of zoledronic acid. This is an interesting observation, and needs to be further evaluated in clinical trials.
Coleman RE, Winter MC, Cameron D, et al. The effects of adding zoledronic acid to neoadjuvant chemotherapy on tumour response: exploratory evidence for direct antitumour activity in breast cancer. Br J Cancer. 2010;102(7):1099–1105.
Chlebowski R, Chen Z, Cauley J, et al. Oral bisphosphonate use and breast cancer incidence in postmenopausal women. J Clin Oncol. 2010;28:3582–90.
Rennert G, Pinchev M, Rennert HS, et al. Use of bisphosphonates and risk of postmenopausal breast cancer. J Clin Oncol. 2010;28:3577–3581.
Coleman R, Bundred N, De Boer R, et al. Impact of zoledronic acid in postmenopausal women with early breast cancer receiving adjuvant letrozole. Canc Res. 2009;69(24):suppl 3–suppl 4082.
Hadji P, Aapro MS, Body JJ, et al. Management of aromatase inhibitor associated bone loss in postmenopausal women: practical guidance for prevention and treatment. Ann Oncol. 2011 April 2(epub ahead of print). This manuscript provides practical advice for clinicians in the management of bone health in breast cancer patients receiving adjuvant hormonal blockade.
Aebi S, Davidson S, Gruber G, on behalf of the ESMO guidelines working group, et al. Ann Oncol. 2010;21(5):v9–v14.
Personal communication, St Gallen breast cancer conference, 2011
Hillner B, Ingle J, Chlebowski R, et al. American society of clinical oncology 2003 update on the role of bisphosphonates and bone health issues in women with breast cancer. J Clin Oncol. 2003;21:4042–57.
Stopeck AT, Lipton A, Body J, et al. Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer. A randomised double blind study. J Clin Oncol. 2010;28:5132–9.
Ellis GK, Bone H, Chlebowski R, et al. Randomized trial of denosumab in patients receiving adjuvant aromatase inhibitors for non metastatic breast cancer. J Clin Oncol. 2008;4875–4882
Disclosure
D. Vorobiof: Consultancy for Amgen, MSD, and Roche, received honoraria, payments for development of educational presentations and had travel/accommodations expenses covered/reimbursed from Amgen, MSD, and Roche; K. Tabane: Received honoraria from Roche and had travel/accommodations expenses covered/reimbursed from Roche and AstraZeneca.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tabane, K., Vorobiof, D.A. Bone Targeted Therapies in Early Breast Cancer. Curr. Treat. Options in Oncol. 12, 412–423 (2011). https://doi.org/10.1007/s11864-011-0163-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11864-011-0163-3