Abstract
Background
JNK pathway-associated phosphatase (JKAP) is engaged in Alzheimer’s disease (AD) pathology via regulating immune response, cluster of differentiation 4 positive (CD4+) T cell differentiation, inflammation, and phosphorylated tau (p-tau). This study aimed to investigate its clinical value serving as a biomarker for AD.
Methods
Fifty AD patients, 50 Parkinson’s disease (PD) patients, and 50 controls (patients with non-degenerative neurological diseases with normal cognition) were enrolled. Their β-protein 42 (Aβ42), total tau (t-tau), p-tau, and Mini-Mental State Examination (MMSE) scale were assessed. Furthermore, JKAP in serum and T-help type 1 (Th1) and T-help type 17 (Th17) cells in CD4+ T cells were measured.
Results
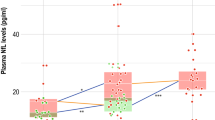
JKAP level was lower, while Th17 cell proportion (but not Th1 cell proportion) was higher in AD patients compared with PD patients and controls (all P < 0.01). Besides, JKAP level negatively correlated with both Th1 (r = − 0.306, P = 0.030) and Th17 (r = − 0.380, P = 0.006) cell proportions in AD patients but not PD patients and controls. Furthermore, in AD patients, JKAP positively correlated with Aβ42 (r = 0.307, P = 0.030) and MMSE score (r = 0.350, P = 0.013) while negatively correlated with p-tau (r = − 0.280, P = 0.048); Th17 cell proportion negatively associated with Aβ42 (r = − 0.281, P = 0.048) and MMSE score (r = − 0.366, P = 0.009). Notably, JKAP was negatively related to 1-year (r = − 0.297, P = 0.038) and 2-year MMSE decline (r = − 0.304, P = 0.048); Th17 cell proportion was positively linked with 1-year (r = 0.392; P = 0.008), 2-year (r = 0.482, P = 0.001), and 3-year (r = 0.365, P = 0.013) MMSE decline.
Conclusion
JKAP, Th1 cells, and Th17 cells are dysregulated and inter-correlated; among them, JKAP and Th17 cells relate to cognitive impairment progression in AD patients.



Similar content being viewed by others
References
Winblad B, Amouyel P, Andrieu S et al (2016) Defeating Alzheimer’s disease and other dementias: a priority for European science and society. Lancet Neurol 15(5):455–532
Tiwari S, Atluri V, Kaushik A et al (2019) Alzheimer’s disease: pathogenesis, diagnostics, and therapeutics. Int J Nanomedicine 14:5541–5554
Yu JT, Xu W, Tan CC et al (2020) Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomised controlled trials. J Neurol Neurosurg Psychiatry 91(11):1201–1209
Gaugler JE, Yu F, Davila HW, Shippee T (2014) Alzheimer’s disease and nursing homes. Health Aff (Millwood) 33(4):650–657
Weller J, Budson A (2018) Current understanding of Alzheimer's disease diagnosis and treatment. F1000Res 7
Monsonego A, Nemirovsky A, Harpaz I (2013) CD4 T cells in immunity and immunotherapy of Alzheimer’s disease. Immunology 139(4):438–446
Chuang HC, Tan TH (2019) MAP4K family kinases and DUSP Family phosphatases in T-cell signaling and systemic lupus erythematosus. Cells 8(11)
Yu D, Peng X, Li P (2020) The correlation between Jun N-terminal kinase pathway-associated phosphatase and Th1 cell or Th17 cell in sepsis and their potential roles in clinical sepsis management. Ir J Med Sci
Chuang HC, Chen YM, Hung WT et al (2016) Downregulation of the phosphatase JKAP/DUSP22 in T cells as a potential new biomarker of systemic lupus erythematosus nephritis. Oncotarget 7(36):57593–57605
Zhou R, Chang Y, Liu J et al (2017) JNK pathway-associated phosphatase/DUSP22 suppresses CD4(+) T-cell activation and Th1/Th17-cell differentiation and negatively correlates with clinical activity in inflammatory bowel disease. Front Immunol 8:781
Kubick N, Flournoy PCH, Enciu AM et al (2020) Drugs modulating CD4+ T cells blood-brain barrier interaction in Alzheimer's disease. Pharmaceutics 12(9)
Terzioglu G, Ormeci B, Turksoy O et al (2017) Mitochondrial depletion in CD4(+) and CD19(+) peripheral lymphocytes in early stage Alzheimer’s disease. Mech Ageing Dev 167:24–29
Mittal K, Eremenko E, Berner O et al (2019) CD4 T cells induce a subset of MHCII-Expressing microglia that attenuates alzheimer pathology. iScience 16:298–311
Saresella M, Calabrese E, Marventano I et al (2011) Increased activity of Th-17 and Th-9 lymphocytes and a skewing of the post-thymic differentiation pathway are seen in Alzheimer’s disease. Brain Behav Immun 25(3):539–547
Jack CR Jr, Albert MS, Knopman DS et al (2011) Introduction to the recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7(3):257–262
Berardelli A, Wenning GK, Antonini A et al (2013) EFNS/MDS-ES/ENS [corrected] recommendations for the diagnosis of Parkinson’s disease. Eur J Neurol 20(1):16–34
Cumming TB, Churilov L, Linden T, Bernhardt J (2013) Montreal cognitive assessment and mini-mental state examination are both valid cognitive tools in stroke. Acta Neurol Scand 128(2):122–129
Sanchez-Mut JV, Aso E, Heyn H et al (2014) Promoter hypermethylation of the phosphatase DUSP22 mediates PKA-dependent TAU phosphorylation and CREB activation in Alzheimer’s disease. Hippocampus 24(4):363–368
Ciccocioppo F, Lanuti P, Pierdomenico L et al (2019) The characterization of regulatory t-cell profiles in Alzheimer’s disease and multiple sclerosis. Sci Rep 9(1):8788
Monteiro FA, Sousa MM, Cardoso I et al (2006) Activation of ERK1/2 MAP kinases in familial amyloidotic polyneuropathy. J Neurochem 97(1):151–161
Banzhaf-Strathmann J, Benito E, May S et al (2014) MicroRNA-125b induces tau hyperphosphorylation and cognitive deficits in Alzheimer’s disease. EMBO J 33(15):1667–1680
Marsh SE, Abud EM, Lakatos A et al (2016) The adaptive immune system restrains Alzheimer’s disease pathogenesis by modulating microglial function. Proc Natl Acad Sci U S A 113(9):E1316-1325
Kisler K, Nelson AR, Montagne A, Zlokovic BV (2017) Cerebral blood flow regulation and neurovascular dysfunction in Alzheimer disease. Nat Rev Neurosci 18(7):419–434
Chen X, Hu Y, Cao Z, Liu Q et al (2018) Cerebrospinal fluid inflammatory cytokine aberrations in Alzheimer’s disease, Parkinson’s disease and amyotrophic lateral sclerosis: a systematic review and meta-analysis. Front Immunol 9:2122
Chen J, Liu X, Zhong Y (2020) Interleukin-17A: the key cytokine in neurodegenerative diseases. Front Aging Neurosci 12:566922
Faraco G, Brea D, Garcia-Bonilla L et al (2018) Dietary salt promotes neurovascular and cognitive dysfunction through a gut-initiated TH17 response. Nat Neurosci 21(2):240–249
Perez-Sen R, Queipo MJ, Gil-Redondo JC et al (2019) Dual-specificity phosphatase regulation in neurons and glial cells. Int J Mol Sci 20(8)
Queipo MJ, Gil-Redondo JC, Morente V et al (2017) P2X7 Nucleotide and EGF receptors exert dual modulation of the dual-specificity phosphatase 6 (MKP-3) in granule neurons and astrocytes, contributing to negative feedback on erk signaling. Front Mol Neurosci 10:448
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
The ethic approval from the Institutional Review Board was acquired before initiation of study, and the written informed consent was obtained from each enrolled subject.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zeng, J., Liu, J., Qu, Q. et al. JKAP, Th1 cells, and Th17 cells are dysregulated and inter-correlated, among them JKAP and Th17 cells relate to cognitive impairment progression in Alzheimer’s disease patients. Ir J Med Sci 191, 1855–1861 (2022). https://doi.org/10.1007/s11845-021-02749-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-021-02749-2