Abstract
Introduction
Prostate cancer cells can switch from an androgen-dependent state to an androgen-independent state after a continuous androgen ablation therapy. However, the molecular mechanisms underlying this switch are still unclear. Therefore, we explored the change in androgen receptor (AR)-related gene expression during this transition in a novel cell model.
Material and methods
Prostate cancer cells were continuously treated with competitive androgen receptor inhibitor hydroxyflutamide for 1.5 years, which yielded an flutamide-insensitive LNCaP subline, LNCaP-flu, as confirmed by MTT assays, flow cytometry, and electron microscopy. We analyzed the differences in gene expression in LNCaP-flu cells and LNCaP cells using gene chips and follow-up RT-PCR.
Results
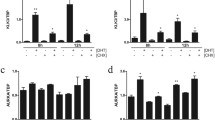
Over 2,428 genes were differentially expressed between these cell lines: 1,194 were down-regulated and 1,234 were up-regulated. Three genes in particular were considered related to the androgen-dependent transition: NCOR1, TIF2 (NCOA2), and ARA70 (NCOA4). There were no apparent changes in expression of the androgen receptor or prostate-specific antigen.
Conclusion
ARs and associated coregulators play a central role in the flutamide-insensitive transition of prostate cancer cells. Although AR expression does not change during this transition, the change in AR coregulators may be a critical factor in the development of antiandrogen insensitivity




Similar content being viewed by others
References
Herranz Amo F, Arias Funez F, Arrizabalaga Moreno M et al (2003) The prostate cancer in the community of Madrid in 2000 I.- Incidence. Actas Urol Esp 27:323–334
Drummond FJ, Carsin AE, Sharp L et al (2010) Trends in prostate specific antigen testing in Ireland: lessons from a country without guidelines. Ir J Med Sci 179:43–49
Leitzmann MF, Platz EA, Stampfer MJ et al (2004) Ejaculation frequency and subsequent risk of prostate cancer. JAMA 291:1578–1586
Neumann F (1982) Pharmacology and clinical use of antiandrogens: a short review. Ir J Med Sci 151:61–70
Huggins C, Hodges CV. Studies on prostatic cancer: I The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res 293–297
Koivisto P, Visakorpi T, Kallioniemi OP (1996) Androgen receptor gene amplification: a novel molecular mechanism for endocrine therapy resistance in human prostate cancer. Scand J Clin Lab Investig Suppl 226:57–63
Miyoshi Y, Uemura H, Fujinami K et al (2000) Fluorescence in situ hybridization evaluation of c-myc and androgen receptor gene amplification and chromosomal anomalies in prostate cancer in Japanese patients. Prostate 43:225–232
Gregory CW, He B, Johnson RT et al (2001) A mechanism for androgen receptor-mediated prostate cancer recurrence after androgen deprivation therapy. Cancer Res 61:4315–4319
Wang Y, Shao C, Shi CH et al (2005) Change of the cell cycle after flutamide treatment in prostate cancer cells and its molecular mechanism. Asian J Androl 7:375–380
Shao C, Wang Y, Yue HH et al (2007) Biphasic effect of androgens on prostate cancer cells and its correlation with androgen receptor coactivator dopa decarboxylase. J Androl 28:804–812
Iliescu R, Campos LA, Schlegel WP et al (2003) Androgen receptor independent cardiovascular action of the antiandrogen flutamide. J Mol Med 7:420–427
Klocker H, Culig Z, Kaspar F et al (1994) Androgen signal transduction and prostatic carcinoma. World J Urol 2:99–103
Olea N, Sakabe K, Soto AM et al (1990) The proliferative effect of “anti-androgens” on the androgen-sensitive human prostate tumor cell line LNCaP. Endocrinology 126:1457–1463
Sonnenschein C, Olea N, Pasanen ME et al (1989) Negative controls of cell proliferation: human prostate cancer cells and androgens. Cancer Res 49:3474–3481
Attar RM, Takimoto CH, Gottardis MM (2009) Castration-resistant prostate cancer: locking up the molecular escape routes. Clin Cancer Res 15:3251–3255
Acknowledgments
This project was funded by a Grant from the National Natural Science Fund of China (No. 30901498) and the Natural Science Fund of Shaanxi Province (No. 2009JQ4003). The authors thank Professor Jian-Guang Zhou of the Academy of Military Medical Sciences for her generous donation of the LNCaP cells used for this research.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Y. Wang, J.-Q. Li and C. Shao contributed equally to this research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Y., Li, JQ., Shao, C. et al. Androgen receptor coregulators NOCR1, TIF2, and ARA70 may account for the hydroxyflutamide insensitivity of prostate cancer cells. Ir J Med Sci 180, 865–872 (2011). https://doi.org/10.1007/s11845-011-0714-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-011-0714-4