Abstract
Introduction
Intervertebral spacers are made of different materials, which can affect the postfusion magnetic resonance imaging (MRI) scans. Susceptibility artifacts, especially for metallic implants, can decrease the image quality. This study aimed to determine whether magnesium as a lightweight and biocompatible metal is suitable as a biomaterial for spinal implants based on its MRI artifacting behavior.
Materials and methods
To compare artifacting behaviors, we implanted into one porcine cadaveric spine different test spacers made of magnesium, titanium, and CFRP. All test spacers were scanned using two T1-TSE MRI sequences. The artifact dimensions were traced on all scans and statistically analyzed.
Results
The total artifact volume and median artifact area of the titanium spacers were statistically significantly larger than magnesium spacers (P < 0.001), while magnesium and CFRP spacers produced almost identical artifacting behaviors (P > 0.05).
Conclusion
Our results suggest that spinal implants made with magnesium alloys will behave more like CFRP devices in MRI scans.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal fusion devices such as implantable interbody spacers are well-established and routinely used by spine surgeons to keep adjacent vertebrae spaced apart while bone ingrowth and fusion take place. Such spacers also provide weight-bearing support between adjacent vertebrae. The principal state-of-the-art spinal implants are made from titanium alloys and carbon fiber-reinforced polymers (CFRP). These biomaterials have enjoyed clinical success and rapid widespread use by improving patient outcomes.
However, these materials have clinical and radiological limitations. Titanium is an excellently bioinert material that exhibits high biocompatibility. Titanium spacers produce good bone ingrowth without bone grafting. However, in magnetic resonance imaging (MRI) studies, titanium-based implants tend to cause distortion of the magnetic field which may obscure normal regional anatomy [1]. These properties pose difficulties in the postoperative MRI follow-up and evaluation of the fusion process due to the artifacting of its causes [5, 6].
The other principal material used for spacers consists of CFRP. Spacers made of this non-metallic biomaterial are not associated with the postoperative diagnostic problems of titanium because carbon produces a very low rate of artifact reactions and its radiolucency properties allow easier evaluation of the fusion process by MRI [3]. Carbon’s modulus of elasticity affords good load-bearing with sufficient hardness. But unlike titanium, carbon spacers undergo poor osteointegration because a soft tissue interface develops around the material surface that prevents direct ingrowth of bone. As a result, carbon spacers have to be filled with bone allografts to achieve long-term stability [1, 3]. CFRP implants have, therefore, been reviewed very critically in the literature [13].
Surgeons, over a century ago, recognized the potential of the lightweight metal magnesium as a biocompatible, osteoconductive, degradable implant material [7]. In 1907, Lambotte [7] was the first to introduce magnesium-based orthopedic devices; using a pure magnesium plate; he secured a bone fracture of the lower leg with gold-plated nails. A half a century later, magnesium-based metals were reported to have osteoconductive bioactivity and produce a more rapid formation of hard callus when used to support fractures in humans [16, 21]. The large amounts of evidence supporting the clinical advantages of magnesium have been summarized in a recent review paper [14]. None of the studies to date have yet investigated the diagnostic behavior of magnesium in MRI. This situation motivated us to determine whether magnesium is a suitable biomaterial for spinal implants by studying its MRI artifacting behavior.
Materials and methods
To evaluate the behavior of spacers made with a magnesium alloy, we compared their artifacting in diagnostic MRI scans with that of spacers made of a conventional titanium alloy and of CFRP. We consecutively implanted three spacers made of each of the three biomaterials dimensioned in small, medium, and large sizes in one cadaveric spine of a Gottingen mini pig (Figs. 1a–c, 2). The three spacers in group I were made of a magnesium–aluminium–manganese alloy (MgAlMn50), the three in group II of a titanium–aluminum–vanadium alloy (TiAl6V4), and those in group III of a carbon fiber-reinforced polymer (CFRP).
Table 1 presents the implant characteristics. A cylinder was chosen as for spacer shape because cylinders have demonstrated lowest rate of MRI artifacting behavior [4]. The spacer sizes—small, medium, and large—were dimensioned the same for each group (height in cm × base area in cm2); and their implant volume (IV) in cm3 and cross sectional area (CSA) in cm² was calculated for each size (Table 1). The spacer sizes were dimensioned as listed after a Newman–Keuls multiple comparison analysis showed that the selected sizes would produce significantly different artifacting behaviors (P < 0.001). Thus, a total of nine individual spacers were implanted, scanned by MRI, and evaluated for their artifacting behavior on the scans.
Spacer implantation
For each serial MRI study, the cylindrical implant was placed exactly between two adjacent vertebrae of the cadaveric porcine spine. The spine with implant was then completely packed in a soft-tissue mass and placed in a plastic container [4]. To create comparable trial conditions, markings were drawn on the container wall to demarcate the vertebrae and implant positions. These demarcations were used to define the median artifact area (MAA). The container with the spine implanted with each spacer was examined by serial MRI (Fig. 3).
Magnetic resonance imaging
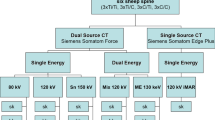
Magnetic resonance imaging was performed with a 1.5 T MRI (Magnetom Symphony, Siemens AG Medical Solutions, Erlangen, Germany). The T1w-TSE sequences were used to acquire a slice thickness of 3 mm (Fig. 3a–c) which included a first sequence (TR 600; TE 14; flip angle 15; band width 150), and a second sequence (TR 2,260, TE 14, flip angle 15, band width 150). We selected a matrix of 512 × 512 pixels combined with a field of view (FOV) of 500 mm. The T1w-TSE sequence has been established to produce best imaging results for implants and the least amount of intrinsic artifacting [3, 5, 6, 8–10, 12, 20].
Using a current version of DICOM reader software, one author (TE) measured the artifact area on the scan of each of the nine implants six times, i.e., a total of 54 individual tracings were recorded and analyzed. The measurements started with the slice with the first artifacting reaction and ended with the last slice exhibiting an artifact reaction. Corresponding to the respective implant’s CSA, the middle slice of all slices exhibiting artifact reactions was defined as the MAA for each implant. To calculate the total artifact volume (TAV) for each spacer, all artifact areas measured for that spacer were added and multiplied by the slice thickness of 3 mm according to the multisection slice technique described by Debatin et al. [2]. The ratio of CSA to MAA and the ratio of IV to TAV were calculated and presented in tables (Table 1).
Statistical analysis
Newman–Keuls multiple comparisons were used to calculate intragroup differences in TAV and MAA (Table 2). T test correlations were performed to determine any intergroup differences regarding the implant materials (Table 2). A P value of <0.05 indicated a significant difference between the means of any two groups.
Results
Table 1 presents the spacer dimensions. Table 2 shows the intragroup comparisons of target variables. Table 3 lists the results of the intergroup t test correlations between TAV and MAA in relation to spacer material. Mean artifacting behavior increased with spacer size. When magnesium was compared with titanium, there were significant differences in both MAA and TAV. When magnesium was compared with carbon, the differences were not significant. In fact, magnesium produces an artifacting behavior very similar to that of CFRP.
Discussion
Spinal surgeons have not stopped searching for the optimum spacer material that combines high biocompatibility with artifact-free MRI imaging behavior in the implant environment. This study conducted to determine whether cylindrical spacers made of the biomaterial magnesium are suitable as spinal implants by comparing their MRI artifacting with that of identically dimensioned spacers made of a titanium alloy and a carbon fiber-reinforced polymer.
In radiological spinal diagnostics, MRI is highly effective for clarifying postfusion questions regarding osseus and soft-tissue structures in relation to implant position. A comparative in vitro study shows that MRI has a higher sensitivity than CT in detecting osseus changes in the implant’s direct surroundings [19]. Moreover, MRI is well suited to demonstrate myelopathies, inflammatory and infectious processes, and any neurodegenerative changes. The MRI imaging behavior of spinal implants is obviously well documented in the literature [8, 10–12, 15, 17, 18, 20]. However, the aims of the published studies differed in that most focused on determining sequence-related artifact size. In a phantom study by Rudisch et al. [11], the relevance of metallic artifacts and implant-related characteristics, such as implant material and position, was demonstrated in addition to effects caused by the selected MRI sequence. In materials with a higher magnetizability like titanium alloys, implant shape additionally has an effect on the range of MRI artifacts [4].
The results of this comparative study showed that implant material and volume both affected the MRI artifacting behavior of our cylindrical test spacers. It was also noted that the smaller the implant size, the smaller was the range of susceptibility artifacts produced. The ratios calculated in Table 2 prove that the magnesium metal alloy exhibited behavior artifacting that was more like a non-metal.
Our results confirm previous findings that MRI artifacting caused by solid implants is influenced by implant material, volume, and shape [4]. Judging from its non-metal-like MRI artifacting behavior alone, magnesium would appear to be a more suitable biomaterial for spinal implants than titanium. Given its osseoconductive potential as a metal [7], implant alloys made with magnesium would combine the advantages to the two principal spacer materials currently used, but without their limitations, at least in terms of MRI artifacting. Hence, magnesium alloys may show promise as spinal implants.
References
Brantigan JW, Steffee AD (1993) A carbon fiber implant to aid interbody lumbar fusion. Two-year clinical results in the first 26 patients. Spine 18:2106–2107
Debatin JF, Nadel SN, Sostman HD, Spritzer CE, Evans AJ, Grist TM (1992) Magnetic resonance imaging-cardiac ejection fraction measurements. Phantom study comparing four different methods. Invest Radiol 27:198–204. doi:10.1097/00004424-199203000-00003
Ernstberger T, Heidrich G, Bruening T, Krefft S, Buchhorn G, Klinger HM (2006) The interobserver-validated relevance of intervertebral spacer materials in MRI artifacting. Eur Spine J 7:1–7
Ernstberger T, Heidrich G, Buchhorn G (2007) Post implantation MRI with cylindric and cubic intervertebral test implants: evaluation of implant shape, material and volume in MRI artifacting- an in vitro study. Spine J 7:353–359. doi:10.1016/j.spinee.2006.03.016
Henk CB, Brodner W, Grampp S, Breitenseher M, Thurnher M, Mostbeck GH, Imhof H (1999) The postoperative spine. Top Magn Reson Imaging 10:247–264. doi:10.1097/00002142-199908000-00006
Herold T, Caro WC, Heers G, Perlick L, Grifka J, Feuerbach S, Nitz W, Lenhart M (2004) Influence of sequence type on the extent of the susceptibility artifact in MRI-a shoulder specimen study after suture anchor repair. Rofo 176:1296–1301
Lambotte A (1932) L’utilisation du magnesium comme materiel perdu dans l’ostheosynthèse. Bull Mem Coc Nat Chir 28:1325–1334
Malik AS, Boyko O, Atkar N, Young WF (2001) A comparative study of MR imaging profile of titanium pedicle screws. Acta Radiol 42:291–293. doi:10.1080/028418501127346846
Ortiz O, Pait TG, McAllister P, Sauter K (1996) Postoperative magnetic resonance imaging with titanium implants of the thoracic and lumbar spine. Neurosurgery 38:741–745. doi:10.1097/00006123-199604000-00022
Petersilge CA, Lewin JS, Duerk JL, Yoo JU, Ghaneyem AJ (1996) Optimizing imaging parameters for MR evaluation of the spine with titanium pedicle screws. Am J Roentenol 166:1213–1218
Rudisch A, Kremser C, Peer S, Kathrein A, Judmaier W, Daniaux H (1998) Metallic artifacts in magnetic resonance imaging of patients with spinal fusion. A comparison of implant materials and implant sequences. Spine 23:692–699. doi:10.1097/00007632-199803150-00009
Rupp R, Ebraheim NA, Savolaine ER, Jackson WT (1993) Magnetic resonance imaging evaluation of the spine with metal implants. General safety and superior imaging with titanium. Spine 18:379–385
Schreiner U, Schwarz M, Scheller G, Schroeder-Boersch H, Jani L (2000) Knöchernes Einwachsverhalten von Probekörpern aus kohlefaserverstärktem Kunststoff. Z Orthop Ihre Grenzgeb 138:540–543. doi:10.1055/s-2000-9598
Staiger MP, Pietak AM, Huadmai J, Dias G (2006) Magnesium and its alloys as orthopaedic biomaterials: a review. Biomaterials 27:1728–1734. doi:10.1016/j.biomaterials.2005.10.003
Thomsen M, Schneider U, Breusch SJ, Hansmann J, Freund M (2001) Artefacts and ferromagnetism dependent on different metal alloys in magnetic resonance imaging. An experimental study. Orthopade 30:540–544. doi:10.1007/s001320170063
Troitskii VV, Tsitrin DN (1944) The resorbing metallic alloy ‘Ostheosinthzit’ as material for fastening broken bone. Khirurgiia 8:41–44
Vaccaro AR, Chesnut RM, Scuderi G, Healy JF, Massie JB, Garfin SR (1994) Metallic spinal artifacts in magnetic resonance imaging. Spine 19:1237–1242
Van Goethem JW, Parizel PM, Jinkins JR (2002) Review article: MRI of the postoperative lumbar spine. Neuroradiology 44:723–739. doi:10.1007/s00234-002-0790-2
Walde TA, Weiland DE, Leung SB, Sychterz CJ, Ho S, Engh CA, Potter HG (2005) Comparison of CT, MRI and radiographs in assessing pelvic osteolysis: a cadaveric study. Clin Orthop Relat Res 437:138–144. doi:10.1097/01.blo.0000164028.14504.46
Wang JC, Sandhu HS, Yu MD, Minchew JT, Delamarter RB (1997) MR parameters for imaging titanium spinal instrumentation. J Spinal Disord 10:27–32. doi:10.1097/00002517-199702000-00004
Znamenskii MS (1945) Metallic ostheosynthesis by means of an apparatus made of resorbing metal. Khirurgiia 12:60–63
Acknowledgments
The authors thank Dr. Mark Riner of MedTech Composites GmbH, Switzerland, the Peter Brehm Company, Weisendorf, Germany, and the Material Science at the Technical University of Hannover, Germany for producing the respective test implants.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s11845-015-1266-9.
The above named article is to be retracted from the Irish Journal of Medical Science. As a result of poor communication, the manuscript was submitted to Neuroradiology whilst still under consideration by the Irish Journal of Medical Science, and subsequently published online by Neuroradiology on 26th May 2009. The article was published online within the Irish Journal of Medical Science on 20th August 2009; due to being the latter of the two article versions, the Editor-in-Chief of the Irish Journal of Medical Science has decided to retract the article. Readers wishing to access the article content can do so at Neuroradiology, August 2009, Volume 51, Issue 8, pp 525-529, DOI: 10.1007/s00234-009-0537-4
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Ernstberger, T., Buchhorn, G. & Heidrich, G. RETRACTED ARTICLE: Magnetic resonance imaging evaluation of intervertebral test spacers: an experimental comparison of magnesium versus titanium and carbon fiber reinforced polymers as biomaterials. Ir J Med Sci 179, 107–111 (2010). https://doi.org/10.1007/s11845-009-0394-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-009-0394-5