Abstract
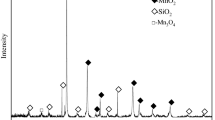
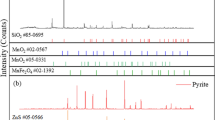
In this article, a novel efficient and environmental reductive roasting-acid leaching technique was proposed by a combined pretreatment of the microwave heating and reduction of alkali lignin. Factors influencing the leaching of manganese from manganese oxide ores were investigated, such as heating modes (microwave heating and conventional heating), microwave roasting parameters and acid leaching parameters. The results indicated that the leaching ratio of manganese by microwave heating was significantly higher than that by conventional heating under the same conditions. The reduction of manganese oxide ores by microwave heating was completed at a roasting temperature of 150°C and a roasting time of 5 min with the addition of 50% alkali lignin with the reduction sequence of MnO2 → Mn2O3 → Mn3O4 → MnO. A 97.43% leaching ratio of manganese was obtained from the roasted ore, which was leached by a 1.5 mol/L concentration of sulfuric acid at a 50°C leaching temperature, 50 rpm stirring speed and a 10:1 liquid-to-solid ratio for 5 min. Compared to the conventional reductive roasting-acid methods, the proposed technique was performed at a considerably lower temperature and shorter time, with higher reducing agent utilization ratio to processing the different manganese oxide ores.









Similar content being viewed by others
References
B. Das, S. Prakash, P.S.R. Reddy, and V.N. Misra, Resour. Conserv. Recycl. 50, 40–57 https://doi.org/10.1016/j.resconrec.2006.05.008 (2007).
P. Das, S. Upadhyay, S. Dubey, and K.K. Singh, J. Environ. Chem. Engin. 9, 105640 https://doi.org/10.1016/j.jece.2021.105640 (2021).
R. Zhang, X. Ma, X. Shen, Y. Zhai, T. Zhang, C. Ji, and J. Hong, J. Clean. Prod. 253, 119951 https://doi.org/10.1016/j.jclepro.2019.119951 (2020).
Od.S.H. Santos, Cd.F. Carvalho, GAd. Silva, and CGd. Santos, J. Environ. Manag. 147, 314–320 https://doi.org/10.1016/j.jenvman.2014.09.020 (2015).
Q.-Q. Lin, G.-H. Gu, H. Wang, R.-F. Zhu, Y.-C. Liu, and J.-G. Fu, Int. J. Miner. Metall. Mater. 23, 491–500 https://doi.org/10.1007/s12613-016-1260-x (2016).
J.D. Steenkamp, D. Chetty, A. Singh, S.A.C. Hockaday, and G.M. Denton, JOM 72, 3422–3435 https://doi.org/10.1007/s11837-020-04318-x (2020).
R. Elliott, K. Coley, S. Mostaghel, and M. Barati, JOM 70, 680–690 https://doi.org/10.1007/s11837-018-2769-4 (2018).
G. Chen, C. Jiang, and R. Liu, Sep. Purif. Technol. 277, 119472 https://doi.org/10.1016/j.seppur.2021.119472 (2021).
N. Toro, F. Rodríguez, A. Rojas, P. Robles, and Y. Ghorbani, Miner. Eng. 163, 106748 https://doi.org/10.1016/j.mineng.2020.106748 (2021).
V. Singh, T. Chakraborty, and S.K. Tripathy, Min. Proc. Ext. Met. Rev. 41(6), 417–438 https://doi.org/10.1080/08827508.2019.1634567 (2020).
A. Ganbari Arbat, E. Asghari Fesaghandis, A. Taghizadeh Tabrizi, and H. Aghajani, Trans. Indian Inst. Metals 73, 2355–2360 https://doi.org/10.1007/s12666-020-02036-1 (2020).
A. Shoghian-Alanaghi, A.J. Zamharir, H. Aghajani, and A.T. Tabrizi, Min. Metall. Explor. 39(4), 1753–1761 https://doi.org/10.1007/s42461-022-00642-9 (2022).
M. Petranikova, A.H. Tkaczyk, A. Bartl, A. Amato, V. Lapkovskis, and C. Tunsu, Waste Manag. 113, 521–544 https://doi.org/10.1016/j.wasman.2020.04.007 (2020).
A.P. Das, L.B. Sukla, N. Pradhan, and S. Nayak, Bioresour. Technol. 102, 7381–7387 https://doi.org/10.1016/j.biortech.2011.05.018 (2011).
Q. Zhao, L. Sun, G. Wang, C. Luo, Y. Shun, and K. Yan, Hydrometallurgy 189, 105113 https://doi.org/10.1016/j.hydromet.2019.105113 (2019).
O. Ostrovski, S.E. Olsen, M. Tangstad, and M. Yastreboff, Can. Metall. Q. 41, 309–318 (2002).
G. Akdogan, and R.H. Eric, Metall. Mater. Trans. B 26, 13–24 https://doi.org/10.1007/BF02648973 (1995).
R.K. Jana, B.D. Pandey, and Premchand, Hydrometallurgy 53, 45–56 https://doi.org/10.1016/S0304-386X(99)00031-6 (1999).
V. Nunna, S. Hapugoda, M.I. Pownceby, and G.J. Sparrow, Miner. Eng. 166, 106826 https://doi.org/10.1016/j.mineng.2021.106826 (2021).
W. Wei, Z. Shao, Y. Zhang, R. Qiao, and J. Gao, Appl. Therm. Eng. 157, 113751 https://doi.org/10.1016/j.applthermaleng.2019.113751 (2019).
Y. Sun, P. Gao, Y. Han, and D. Ren, Ind. Eng. Chem. Res. 52, 2323–2329 https://doi.org/10.1021/ie303233k (2013).
D.Q. Zhu, Y. Cui, K. Vining, S. Hapugoda, J. Douglas, J. Pan, and G.L. Zheng, Int. J. Miner. Process. 106–109, 1–7 https://doi.org/10.1016/j.minpro.2012.01.003 (2012).
G. Li, T. Shi, M. Rao, T. Jiang, and Y. Zhang, Miner. Eng. 32, 19–26 https://doi.org/10.1016/j.mineng.2012.03.012 (2012).
K.-O. Jang, V.R.M. Nunna, S. Hapugoda, A.V. Nguyen, and W.J. Bruckard, Miner. Eng. 60, 14–22 https://doi.org/10.1016/j.mineng.2014.01.021 (2014).
A. Mehdilo, and M. Irannajad, J. Ind. Eng. Chem. 33, 59–72 https://doi.org/10.1016/j.jiec.2015.09.018 (2016).
G. Li, M. Liu, M. Rao, T. Jiang, J. Zhuang, and Y. Zhang, J. Hazard. Mater. 280, 774–780 https://doi.org/10.1016/j.jhazmat.2014.09.005 (2014).
S. Agrawal, V. Rayapudi, and N. Dhawan, Miner. Eng. 132, 202–210 https://doi.org/10.1016/j.mineng.2018.12.012 (2019).
S. Agrawal, and N. Dhawan, Sustain. Mater. Technol. 27, e00246 https://doi.org/10.1016/j.susmat.2021.e00246 (2021).
Q. Ye, H. Zhu, L. Zhang, J. Ma, L. Zhou, P. Liu, J. Chen, G. Chen, and J. Peng, J. Alloys Compd. 613, 102–106 https://doi.org/10.1016/j.jallcom.2014.06.016 (2014).
Q. Ye, H. Zhu, L. Zhang, P. Liu, G. Chen, and J. Peng, RSC Adv. 4, 58164–58170 https://doi.org/10.1039/C4RA08010F).10.1039/C4RA08010F (2014).
Y. Sun, G. Fu, L. Jiang, and X. Cai, Min. Metall. Explor. 35, 215–220 https://doi.org/10.19150/mmp.8598 (2018).
S. Lin, R. Liu, and S. Guo, Renew. Energy 181, 714–724 https://doi.org/10.1016/j.renene.2021.09.055 (2022).
J. Ju, Y. Feng, H. Li, H. Yu, H. Wu, and S. Liu, Sustain. Chem. Pharm. 19, 100346 https://doi.org/10.1016/j.scp.2020.100346 (2021).
Y.-B. Zhang, Y. Zhao, Z.-X. You, D.-X. Duan, G.-H. Li, and T. Jiang, J Cent. South Univ. 22, 2515–2520 https://doi.org/10.1007/s11771-015-2780-7 (2015).
S. Yuan, W. Zhou, Y. Han, and Y. Li, Powder Technol. 361, 529–539 https://doi.org/10.1016/j.powtec.2019.11.082 (2020).
Y. Cao, Y. Sun, P. Gao, Y. Han, and Y. Li, Int. J. Min. Sci. Technol. 31, 1075–1083 https://doi.org/10.1016/j.ijmst.2021.09.008 (2021).
K. Li, J. Chen, G. Chen, J. Peng, R. Ruan, and C. Srinivasakannan, Bioresour. Technol. 286, 121381 https://doi.org/10.1016/j.biortech.2019.121381 (2019).
K. Li, G. Chen, J. Chen, J. Peng, R. Ruan, and C. Srinivasakannan, Bioresour. Technol. 291, 121838 https://doi.org/10.1016/j.biortech.2019.121838 (2019).
X. Wang, L. Mei, X. Xing, L. Liao, G. Lv, Z. Li, and L. Wu, Appl. Catal. B 160–161, 211–216 https://doi.org/10.1016/j.apcatb.2014.05.009 (2014).
F. Wu, Z. Cao, S. Wang, and H. Zhong, J. Alloys Compd. 722, 651–661 https://doi.org/10.1016/j.jallcom.2017.06.142 (2017).
F. Wu, J. Deng, B. Mi, Z. Xiao, J. Kuang, H. Liu, M. Liang, B. Liu, and P. Yu, Powder Technol. 356, 170–176 https://doi.org/10.1016/j.powtec.2019.08.020 (2019).
J. Geng, W.-L. Wang, Y.-X. Yu, J.-M. Chang, L.-P. Cai, and S.Q. Shi, Bioresour. Technol. 227, 1–6 https://doi.org/10.1016/j.biortech.2016.11.036 (2017).
D. Chen, K. Cen, X. Cao, F. Chen, J. Zhang, and J. Zhou, Renew. Sustain. Energy Rev. 136, 110444 https://doi.org/10.1016/j.rser.2020.110444 (2021).
D. Chen, Y. Wang, Y. Liu, K. Cen, X. Cao, Z. Ma, and Y. Li, Fuel 252, 1–9 https://doi.org/10.1016/j.fuel.2019.04.086 (2019).
Z. Abubakar, A.A. Salema, and F.N. Ani, Bioresour. Technol. 128, 578–585 https://doi.org/10.1016/j.biortech.2012.10.084 (2013).
B.T. Pérez-Martínez, M.A. Aboudzadeh, U.S. Schubert, J.R. Leiza, and R. Tomovska, Chem. Eng. J. 399, 125761 https://doi.org/10.1016/j.cej.2020.125761 (2020).
J. Huang, G. Xu, Y. Liang, G. Hu, and P. Chang, Fuel 266, 117022 https://doi.org/10.1016/j.fuel.2020.117022 (2020).
J. Li, J. Dai, G. Liu, H. Zhang, Z. Gao, J. Fu, Y. He, and Y. Huang, Biomass Bioenergy 94, 228–244 https://doi.org/10.1016/j.biombioe.2016.09.010 (2016).
Acknowledgements
This work was supported by Hunan Province Science and Technology Talent Support Project (grant number 2022TJ-N15); Key Scientific research project of Education Department of Hunan Province (grant number 20A245); General Project of Natural Science Foundation of Hunan Province (grant number 2021JJ30410, 2022JJ30348).
Author information
Authors and Affiliations
Contributions
JH: Conceptualization, Methodology, Formal analysis, Writing—review & editing, Funding acquisition; LC: Investigation, Data curation, Validation, Writing—original draft; JZ: Investigation, Data curation, Formal analysis; YZ: Investigation, Data curation; JZ: Conceptualization, Data curation; LC: Conceptualization, Data curation; WZ: Investigation, Data curation; HT: Investigation, Formal analysis; JY: Investigation, Data curation; FW*: Resources, Funding acquisition, Methodology, Supervision, Validation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, J., Chen, L., Zhang, J. et al. A Process for Extraction of Manganese from Manganese Oxide Ores by a Novel and Efficient Roasting-Acid Leaching Technique. JOM 75, 3511–3520 (2023). https://doi.org/10.1007/s11837-023-05896-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-023-05896-2