Abstract
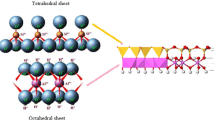
Swelling of montmorillonite (Mt) is an important factor for many industrial applications. In this study, crystalline swelling of alkali-metal- and alkaline-earth-metal-Mt has been studied through energy optimization and molecular dynamics simulations using the clay force field by Materials Studio 8.0. The delamination and exfoliation of Mt are primarily realized by crystalline swelling caused by the enhanced interlayer cation hydration. The initial position of the interlayer cations and water molecules is the dominated factor for the accuracy of the Mt simulations. Crystalline swelling can be carried out in alkali-metal-Mt and Mg-Mt but with difficulty in Ca-Mt, Sr-Mt and Ba-Mt. The crystalline swelling capacity values are in the order Na-Mt > K-Mt > Cs-Mt > Mg-Mt. This order of crystalline swelling of Mt in the same group can be attributed to the differences between the interlayer cation hydration strengths. In addition, the differences in the crystalline swelling between the alkali-metal-Mt and alkaline-earth-metal-Mt can be primarily attributed to the valence of the interlayer cations.




Similar content being viewed by others
References
A. Helmy, J. Colloid Interface Sci. 207, 128 (1998).
D.E. Smith, Langmuir 14, 5959 (1998).
F. Bergaya and G. Lagaly, Handbook of Clay Science, 2nd ed. (Amsterdam: Elsevier Science, 2013), p. 43.
S.M. Rao, T. Thyagaraj, and P. Raghuveer Rao, Geotech. Geol. Eng. 31, 1399 (2013).
E. Ferrage, B. Lanson, N. Malikova, A. Plançon, B.A. Sakharov, and V.A. Drits, Chem. Mater. 17, 3499 (2005).
E. Ferrage, B. Lanson, B.A. Sakharov, and V.A. Drits, Am. Mineral. 90, 1358 (2005).
S. Morodome and K. Kawamura, Clays Clay Miner. 59, 165 (2011).
L.L. Schramm and J.C.T. Kwak, Colloids Surf. 4, 43 (1982).
K. Jasmund and G. Lagaly, Tonminerale und Tone, 1st ed. (Darmstadt: Steinkopff Verlag, 1993), p. 66.
N.T. Skipper, G. Sposito, and F.R.C. Chang, Clays Clay Miner. 43, 294 (1995).
F.C. Chang, N.T. Skipper, and G. Sposito, Langmuir 11, 2074 (1997).
M.J. Avena, R. Cabrol, and C.P.D.E. Pauli, Clays Clay Miner. 38, 356 (1990).
W.K. Mekhamer and F.F. Assaad, J. Appl. Polym. Sci. 73, 659 (1998).
P. Na and F. Zhang, Acta Phys. Chim. Sin. 22, 1137 (2006).
L. Zhang, X. Lu, X. Liu, J. Zhou, and H. Zhou, J. Phys. Chem. C 118, 29811 (2014).
S.L. Teich-McGoldrick, J.A. Greathouse, C.F. Jove-Colon, and R.T. Cygan, J. Phys. Chem. C 119, 20880 (2015).
V. Marry, P. Turq, T. Cartailler, and D. Levesque, J. Chem. Phys. 117, 3454 (2002).
A. Delville and P. Laszlo, New J. Chem. 13, 481 (1989).
F.R.C. Chang, N.T. Skipper, and G. Sposito, Langmuir 11, 2734 (1995).
G. Lagaly and I. Dékány, Developments in clay science.Colloid Clay Science, 2nd ed., ed. F. Bergaya and G. Lagaly (Amsterdam: Elsevier Inc., 2013), p. 255.
R.F. Giese, C.J. Van Oss, J. Norris, and P.M. Costanzo, Report No. 86, Company, Mémoire, January, (1989).
M.E. Schrader and S. Yariv, J. Colloid Interface Sci. 136, 85 (1990).
L.J. Michot, F. Villieras, M. Francois, J. Yvon, R. Le Dred, and J.M. Cases, Langmuir 10, 3765 (1994).
H. Malandrini, F. Clauss, S. Partyka, and J. Douillard, J. Colloid Interface Sci. 194, 183 (1997).
L. Sun, J.T. Tanskanen, J.T. Hirvi, S. Kasa, T. Schatz, and T.A. Pakkanen, Chem. Phys. 455, 23 (2015).
J.D. Russell and V.C. Farmer, Clay Miner. 5, 443 (1964).
T. Journal, W. Kirmske, A. Press, W.L. Hase, J.W. Simons, E. Richardson, J.W. Rabalais, J.M. Mcdonakl, V. Scherer, P. Mcglynn, S.P. Waich, W. Goddard, G. Herzberg, V. Nostrand, G. Verhaegen, L. Russell, F.S. Rowland, H. Laufer, M. Bass, C.F. Curtis, R.B. Bird, C. Chan, J.T. Bryant, L.D. Spicer, Y.N. Lin, S.P. Pavlou, G.W. Taylor, K. Krohn, N.J. Parks, J.W. Root, A. Hosaka, P.J. Robinson, K. Holbrook, T.H. Richardson, D.L. Bunker, Q.C. Program, F.B. Growcock, and L. Johnson, Society 82, 26 (1978).
G. Sposito, R. Prost, and J.P. Gaultier, Clays Clay Miner. 31, 9 (1983).
C.T. Johnston, G. Sposito, and C. Erickson, Clays Clay Miner. 40, 722 (1992).
J.L. Bishop, C.M. Pieters, and J.O. Edwards, Clays Clay Miner. 42, 702 (1994).
S.F. Xu, W.Z. Johnston, C.T. Parker, and P. Agnew, Clays and Clay Miner. 48, 120 (2000).
E. Rinnert, C. Carteret, B. Humbert, G. Fragneto-Cusani, J.D.F. Ramsay, A. Delville, J.L. Robert, I. Bihannic, M. Pelletier, and L.J. Michot, J. Phys. Chem. B 109, 23745 (2005).
J.N. Israelachvili, Intermolecular and Surface Forces, 3rd ed. (Amsterdam: Elsevier inc, 2011), p. 71.
N. Malikova, A. Cadènea, E. Dubois, V. Marry, S. Durand-Vidal, P. Turq, J. Breu, S. Longeville, and J.M. Zanotti, J. Phys. Chem. C 111, 17603 (2007).
Acknowledgements
The financial support of this work from the National Natural Science Foundation of China under the Project Nos. 51474167, 51674183 and 51474011 is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, H., Song, S., Dong, X. et al. Molecular Dynamics Study of Crystalline Swelling of Montmorillonite as Affected by Interlayer Cation Hydration. JOM 70, 479–484 (2018). https://doi.org/10.1007/s11837-017-2666-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-017-2666-2