Abstract
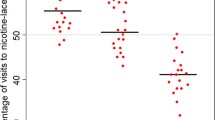
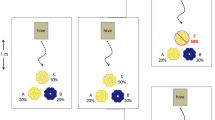
Floral nectar functions to attract insects, so the inclusion of toxic compounds calls for explanation. Recent work shows that honeybees prefer nectars with low concentrations of caffeine and nicotine, and that associative learning by honeybees is enhanced by caffeine, prompting speculation that pollination service could be enhanced. We directly tested caffeine’s effect on pollination service by allowing bumblebee colonies to feed on arrays of artificial flowers that offer nectar while also dispensing and receiving dye particles as pollen analogues. With caffeine levels signaled by flower color (blue, green, or yellow) in a factorial design, flowers offering nectar with 10−5 M caffeine received significantly more pollen analogue than did those with 10−4 M caffeine or with no caffeine. Effects of caffeine were unaffected by which colors were associated with which caffeine levels: Color alone had no significant effect, and there was no interaction between color and caffeine level. In cases where greater pollination service translates to increased fitness, we would expect stabilizing selection to maintain nectar caffeine at intermediate levels.



Similar content being viewed by others
References
Adler LS (2000) The ecological significance of toxic nectar. Oikos 91:409–420
Adler LS, Irwin RE (2012) Nectar alkaloids decrease pollination and female reproduction in a native plant. Oecologia 168:1033–1041
Avarguès-Weber A, Chittka L (2014) Observational conditioning in flower choice copying by bumblebees (Bombus terrestris): influence of observer distance and demonstrator movement. PLoS One 9:1–5
Baker HG, Baker I (1975) Studies of nectar constitution and pollinator-plant coevolution. In: Gilbert LE, Raven PH (eds) Coevolution of animals and plants. University of Texas Press, Austin, pp 100–140
Cartar RV (2004) Resource tracking by bumble bees: responses to plant-level differences in quality. Ecology 85:2764–2771
Castellanos MC, Wilson PS, Wolfe A, Keller SA, Thomson JD (2006) Anther evolution: pollen presentation strategies when pollinators differ in efficiency. Am Nat 167:288–296
Cembrowski A, Tan MG, Thomson JD, Frederickson M (2013) Ants and ant scent reduce bumblebee pollination of artificial flowers. Am Nat 183:133–139
Chittka L, Peng F (2013) Caffeine boosts bees’ memories. Science 339:1157–1159
Detzel A, Wink M (1993) Attraction, deterrence or intoxication of bees (Apis mellifera) by plant allelochemicals. Chemoecology 4:8–18
Dukas R (1995) Transfer and interference learning in bumble bees. Anim Behav 49:1481–1490
Gegear R, Manson JS, Thomson JD (2007) Ecological context influences pollinator deterrence by alkaloids in floral nectar. Ecol Lett 10:378–382
Hagler J, Buchmann SL (1993) Honey bee (Hymenoptera: Apidae) foraging responses to phenolic-rich nectars. J Kans Entomol Soc 66:223–230
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54:187–211
Kawaguchi LG, Ohashi K, Toquenaga Y (2007) Contrasting responses of bumble bees to feeding conspecifics on their familiar and unfamiliar flowers. Proc R Soc B 274:2661–2667
Makino TT, Sakai S (2007) Experience changes pollinator responses top floral display size: from size-based to reward-based foraging. Funct Ecol 21:854–863
Manson JS, Cook D, Gardner DR, Irwin RE (2013a) Dose-dependent effects of nectar alklaloids in a montane plant-pollinator community. J Ecol 101:1604–1612
Manson JS, Rasmann S, Halitsckhe R, Thomson JD, Agrawal AA (2013b) Cardenolides in nectar may be more a consequence of allocation to other plant parts: a phylogenetic study. Funct Ecol 26:1100–1110
Menzel R, Müller U (1996) Learning and memory in honeybees: from behaviour to neural substrates. Annu Rev Neurosci 19:379–404
R Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Rhoades DF, Bergdahl JC (1981) Adaptive significance of toxic nectar. Am Nat 117:798–803
Rodríguez-Gironés MA, Trillo A, Corcobado G (2013) Long term effects of aversive reinforcement on colour discrimination learning in free-flying bumblebees. PLoS One 8(8):e71551
Singaravelan N, Ne’eman G, Inbar M, Izhaki I (2005) Feeding responses of free-flying honeybees to secondary compounds mimicking floral nectars. J Chem Ecol 31:2791–2804
Thomson JD (1988) Effects of variation in inflorescence size and floral rewards on the visitation rates of traplining pollinators of Aralia hispida. Evolut Ecol 2:65–76
Thomson JD, Price MV, Waser NM, Stratton DA (1986) Comparative studies of pollen and fluorescent dye transport by bumble bees visiting Erythronium grandiflorum. Oecologia 69:561–566
Thomson JD, Ogilvie JE, Makino TT, Arisz A, Raju S, Rojas-Luengas V, Tan MG (2012) Estimating pollination success with novel artificial flowers: effects of nectar concentration. J Pollinat Ecol 9:108–114
Tiedeken EJ, Stout JC, Stevenson PC, Wright GA (2014) Bumblebees are not deterred by ecologically relevant concentrations of nectar toxins. J Exp Biol. doi:10.1242/jeb.097543 published online 13 February 2014
Wright GA, Baker DD, Palmer MJ, Stabler D, Mustard JA, Power EF, Borland AM, Stevenson PC (2013) Caffeine in floral nectar enhances a pollinator’s memory of reward. Science 339:1202–1204
Acknowledgments
We thank Biobest for supplying bees, Nicholas Hoban for help with 3D printing, David F. Andrews and Bart Harvey for statistical advice, Alice Zhu for laboratory assistance, and Jessamyn Manson, Lars Chittka, and an anonymous reviewer for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editors: Lars Chittka and Heikki Hokkanen.
Rights and permissions
About this article
Cite this article
Thomson, J.D., Draguleasa, M.A. & Tan, M.G. Flowers with caffeinated nectar receive more pollination. Arthropod-Plant Interactions 9, 1–7 (2015). https://doi.org/10.1007/s11829-014-9350-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-014-9350-z