Abstract
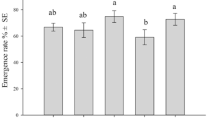
Aphid herbivory decreases primary production in natural ecosystems and reduces crop yields. The mechanism for how aphids reduce yield is poorly understood as some studies suggest aphid feeding directly impedes photosynthesis, whereas other studies suggest a change in allocation of resources from growth to defense compounds reduces yield. To determine the mechanisms underlying reduced plant growth by aphids, Nicotiana attenuata plants, native tobacco, were infested with Myzus persicae ssp. nicotianae, tobacco-adapted green peach aphids, at low and high densities, and plant performance including fitness was assessed. To test the direct defense capacity of salicylic acid (SA) on aphid performance, we fed aphids an artificial diet with varying levels of SA and measured their survivorship and fecundity. There was no detectable effect of aphid herbivory on net photosynthesis, yet herbivory reduced plant growth, final biomass (43 % at high aphid density), and seed set (18 % at high aphid density) at both low and high aphid infestation levels. High-density aphid attack during the rosette and flowering stage caused an increase in SA levels, but caused only a transient decrease in jasmonic acid concentration at low aphid density. SA concentrations similar to those found in infested flowering plants decreased aphid fecundity, suggesting that SA was an effective chemical defense response against aphids. These results suggest that as aphid densities increased the proximal cause of reduced growth and yield was not reduced photosynthesis, but instead resources may have been mobilized for defense via the SA pathway, decreasing the availability of resources for building plant biomass.


Similar content being viewed by others
References
Akbar MDS, Sharma HC, Jayalakshmi SK, Sreeramulu K (2012) Interaction of plant cell signaling molecules, salicylic acid and jasmonic acid, with the mitochondria of Helicoverpa armigera. J Bioenerg Biomembr 44:233–241. doi:10.1007/s10863-012-9405-8
Baron RL, Guthrie FE (1960) A quantitative and qualitative study of sugars found in tobacco as affected by the green peach aphid, Myzus persicae, and its honeydew. Ann Entomol Soc Am 53:220–228
Bezemer TM, Jones TH (1998) Plant-insect herbivore interactions in elevated atmospheric CO2: quantitative analyses and guild effects. Oikos 82:212–222. doi:10.2307/3546961
Blande JD, Korjus M, Holopainen JK (2010) Foliar methyl salicylate emissions indicate prolonged aphid infestation on silver birch and black alder. Tree Physiol 30:404–416. doi:10.1093/treephys/tpp124
Burd JD, Burton RL (1992) Characterization of plant-damage caused by Russian wheat aphid (Homoptera : Aphididae). J Econ Entomol 85:2017–2022
Burd JD, Elliott NC (1996) Changes in chlorophyll a fluorescence induction kinetics in cereals infested with Russian wheat aphid (Homoptera: Aphididae). J Econ Entomol 89:1332–1337
Cipollini D (2002) Does competition magnify the fitness costs of induced responses in Arabidopsis thaliana? A manipulative approach. Oecologia 131:514–520. doi:10.1007/s00442-002-0909-5
Cottrell TE, Wood BW, Ni X (2009) Chlorotic feeding injury by the black pecan aphid (Hemiptera: Aphididae) to pecan foliage promotes aphid settling and nymphal development. Environ Entomol 38:411–416. doi:10.1603/022.038.0214
De Vos M, Van Oosten VR, Van Poecke RM, Van Pelt JA, Pozo MJ, Mueller MJ, Buchala AJ, Metraux JP, Van Loon LC, Dicke M, Pieterse CM (2005) Signal signature and transcriptome changes of Arabidopsis during pathogen and insect attack. Mol Plant-Microbe Interact 18:923–937. doi:10.1094/mpmi-18-0923
Eastop VF, Blackman RL (2005) Some new synonyms in Aphididae (Hemiptera: Sternorrhyncha). Zootaxa 1089:1–36
Freeman JL, Garcia D, Kim D, Hopf A, Salt DE (2005) Constitutively elevated salicylic acid signals glutathione-mediated nickel tolerance in Thlaspi nickel hyperaccumulators. Plant Physiol 137:1082–1091. doi:10.1104/pp.104.055293
Gerloff ED, Ortman EE (1971) Physiological changes in barley induced by greenbug feeding stress. Crop Sci 11:174–176. doi:10.2135/cropsci1971.0011183X001100020003x
Giri AP, Wünsche H, Mitra S, Zavala JA, Muck A, Svatos A, Baldwin IT (2006) Molecular interactions between the specialist herbivore Manduca sexta (Lepidoptera, Sphingidae) and its natural host Nicotiana attenuata. VII. Changes in the plant’s proteome. Plant Physiol 142:1621–1641. doi:10.1104/pp.106.088781
Haile FJ (1999) Physiological and growth tolerance in wheat to Russian wheat aphid (Homoptera: Aphididae) injury. Environ Entomol 28:787–794
Halitschke R, Baldwin IT (2003) Antisense LOX expression increases herbivore performance by decreasing defense responses and inhibiting growth-related transcriptional reorganization in Nicotiana attenuata. Plant J 36:794–807. doi:10.1046/j.1365-313X.2003.01921.x
He Y, Fukushige H, Hildebrand DF, Gan S (2002) Evidence supporting a role of jasmonic acid in Arabidopsis leaf senescence. Plant Physiol 128:876–884. doi:10.1104/pp.010843
Heidel AJ, Baldwin IT (2004) Microarray analysis of salicylic acid- and jasmonic acid-signalling in responses of Nicotiana attenuata to attack by insects from multiple feeding guilds. Plant Cell Environ 27:1362–1373. doi:10.1111/j.1365-3040.2004.01228.x
Heil M, Hilpert A, Kaiser W, Linsenmair KE (2000) Reduced growth and seed set following chemical induction of pathogen defence: does systemic acquired resistance (SAR) incur allocation costs? J Ecol 88:645–654. doi:10.1046/j.1365-2745.2000.00479.x
Herms DA, Mattson WJ (1992) The dilemma of plants: to grow or defend. Q Rev Biol 67:283–335
Kennedy JS, Mittler TE (1953) A method of obtaining phloem sap via the mouth-parts of aphids. Nature 171:528–529. doi:10.1038/171528a0
Kerchev PI, Fenton B, Foyer CH, Hancock RD (2012) Plant responses to insect herbivory: interactions between photosynthesis, reactive oxygen species and hormonal signalling pathways. Plant Cell Environ 35:441–453. doi:10.1111/j.1365-3040.2011.02399.x
Kieckhefer RW (1980) Losses in yield in spring wheat in South Dakota caused by cereal aphids. J Econ Entomol 73:582–585
Kim JH, Jander G (2007) Myzus persicae (green peach aphid) feeding on Arabidopsis induces the formation of a deterrent indole glucosinolate. Plant J 49:1008–1019. doi:10.1111/j.1365-313X.2006.03019.x
Krügel T, Lim M, Gase K, Halitschke R, Baldwin IT (2002) Agrobacterium-mediated transformation of Nicotiana attenuata, a model ecological expression system. Chemoecology 12:177–183. doi:10.1007/pl00012666
Kuśnierczyk A, Winge PER, Jørstad TS, Troczyńska J, Rossiter JT, Bones AM (2008) Towards global understanding of plant defence against aphids—timing and dynamics of early Arabidopsis defence responses to cabbage aphid (Brevicoryne brassicae) attack. Plant Cell Environ 31:1097–1115. doi:10.1111/j.1365-3040.2008.01823.x
Macedo TB, Bastos CS, Higley LG, Ostlie KR, Madhavan S (2003) Photosynthetic responses of soybean to soybean aphid (Homoptera: Aphididae) injury. J Econ Entomol 96:188–193. doi:10.1603/0022-0493-96.1.188
Mewis I, Appel HM, Hom A, Raina R, Schultz JC (2005) Major signaling pathways modulate Arabidopsis glucosinolate accumulation and response to both phloem-feeding and chewing insects. Plant Physiol 138:1149–1162. doi:10.1104/pp.104.053389
Mittler TE (1958) Studies on the feeding and nutrition of Tuberolachnus salignus (Gmelin) (Homoptera, Aphididae). J Exp Biol 35:74–84
Mohase L, van der Westhuizen AJ (2002) Salicylic acid is involved in resistance responses in the Russian wheat aphid-wheat interaction. J Plant Physiol 159:585–590. doi:10.1078/0176-1617-0633
Ng G, Seabolt S, Zhang C, Salimian S, Watkins TA, Lu H (2011) Genetic dissection of salicylic acid-mediated defense signaling networks in Arabidopsis. Genetics. doi:10.1534/genetics.111.132332
Rabbinge R, Drees EM, van der Graaf M, Verberne FCM, Wesselo A (1981) Damage effects of cereal aphids in wheat. Eur J Plant Pathol 87:217–232. doi:10.1007/bf02084437
Ramsey JS, Jander G (2008) Testing nicotine tolerance in aphids using an artificial diet experiment. J Vis Exp. doi:10.3791/701
Rayapuram C, Baldwin IT (2007) Increased SA in NPR1-silenced plants antagonizes JA and JA-dependent direct and indirect defenses in herbivore-attacked Nicotiana attenuata in nature. Plant J 52:700–715. doi:10.1111/j.1365-313X.2007.03267.x
Ryan JD, Johnson RC, Eikenbary RD, Dorschner KW (1987) Drought/greenbug interactions: photosynthesis of greenbug resistant and susceptible wheat. Crop Sci 27:283–288. doi:10.2135/cropsci1987.0011183X002700020033x
Thaler JS, Agrawal AA, Halitschke R (2010) Salicylate-mediated interactions between pathogens and herbivores. Ecology 91:1075–1082. doi:10.1890/08-2347.1
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley—powdery mildew interaction. Plant J 11:1187–1194. doi:10.1046/j.1365-313X.1997.11061187.x
Tjallingii WF, Esch TH (1993) Fine structure of aphid stylet routes in plant tissues in correlation with EPG signals. Physiol Entomol 18:317–328. doi:10.1111/j.1365-3032.1993.tb00604.x
Truernit E (2001) Plant physiology: the importance of sucrose transporters. Curr Biol 11:R169–R171. doi:10.1016/S0960-9822(01)00085-9
Vlot AC, Dempsey DMA, Klessig DF (2009) Salicylic acid, a multifaceted hormone to combat disease. Annu Rev Phytopathol 47:177–206. doi:10.1146/annurev.phyto.050908.135202
Voelckel C, Weisser WW, Baldwin IT (2004) An analysis of plant–aphid interactions by different microarray hybridization strategies. Mol Ecol 13:3187–3195. doi:10.1111/j.1365-294X.2004.02297.x
Wang L, Halitschke R, Kang JH, Berg A, Harnisch F, Baldwin IT (2007) Independently silencing two JAR family members impairs levels of trypsin proteinase inhibitors but not nicotine. Planta 226:159–167. doi:10.1007/s00425-007-0477-3
Weatherley PE, Peel AJ, Hill GP (1959) The physiology of the sieve tube. J Exp Bot 10:1–16. doi:10.1093/jxb/10.1.1
Welter SC (1989) Arthropod impact on plant gas exchange, Insect-plant interactions, vol 1. CRC Press, Boca Raton
Wood BW, Tedders WL (1986) Reduced net photosynthesis of leaves from mature pecan trees by three species of pecan aphid. J Entomol Sci 21:355–360
Zarate SI, Kempema LA, Walling LL (2007) Silverleaf whitefly induces salicylic acid defenses and suppresses effectual jasmonic acid defenses. Plant Physiol 143:866–875. doi:10.1104/pp.106.090035
Zvereva E, Lanta V, Kozlov M (2010) Effects of sap-feeding insect herbivores on growth and reproduction of woody plants: a meta-analysis of experimental studies. Oecologia 163:949–960. doi:10.1007/s00442-010-1633-1
Acknowledgments
We thank PJ Semtner of Virginia Tech University for providing us with M. persicae to begin a colony and IT Baldwin of the Max Planck Institute for Chemical Ecology in Jena, Germany, for providing us with N. attenuata genotypes. We also thank D Bilgin for DAB staining and providing images and thank lab members for constructive comments on an earlier draft. This research was supported in part by grants from the U.S. Department of Energy (No. DE-FG02-04ER63489) and by USDA grant #2002-02723.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Jarmo Holopainen.
Rights and permissions
About this article
Cite this article
Donovan, M.P., Nabity, P.D. & DeLucia, E.H. Salicylic acid-mediated reductions in yield in Nicotiana attenuata challenged by aphid herbivory. Arthropod-Plant Interactions 7, 45–52 (2013). https://doi.org/10.1007/s11829-012-9220-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-012-9220-5