Abstract
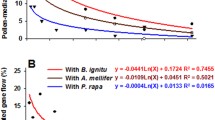
Weed control can be most effectively achieved through the use of herbicide-resistance transgene. A preliminary study bar-transgenic Zoysia japonica posed no serious risk on the unintended escape of the transgene from its cultivation site. The present follow-up investigation of the dispersion of pollen and its short-distance escape outside of the bar-transgenic Zoysia japonica habitats were ascertained in terms of environmental factors affecting anthesis and pollen viability. In a 24-h day cycle, zoysiagrass pollen was released predominantly between 08:00 and 10:00, and the pollen was most viable during the same time interval. Optimal temperature and humidity for pollen viability was 15–20 °C and 80–90%, respectively. The pollen germinated in 120 min after anthesis, but under cloudy conditions germination time doubled. No differences in pollen viability/longevity between the transgenic and non-transgenic plants were observed. The pollen-mediated gene flow of transgenic Zoysia japonica to wild-type non-transgenic zoysiagrass species was monitored by measuring the cross-over rate of the bar gene in the context of three different models. At distances within 5 m, the rate of gene flow ranged from 3 to 5.7% according to the models used. The greater the distance from the transgenic plant site, the lower the gene flow rate. The furthest transgene detected was 38 m away and exhibited a 0.25% gene flow rate. The radial model yielded a 3.7% escape rate within a 3 m radius and was wind direction dependent. The distance- and direction-dependent gene flow events were influenced by wind direction and velocity during flowering season.







Similar content being viewed by others
References
Bae TW, Vanjildorj E, Song SY, Nishiguchi S, Yang SS, Song IJ, Chandrasekhar T, Kang TW, Kim JL, Koh YJ, Park SY, Lee J, Lee YE, Ryu KH, Riu KZ, Song PS, Lee HY (2008) Environmental risk assessment of genetically engineered herbicide-tolerant Zoysia japonica. J Environ Qual 37:207–218 (Corrigendum 38: 2480)
Bhattacharya K, Datta BK (1992) Anthesis and pollen release of some plants of West Bengal. India Grana 31:67–71
Choi DK, Yang KM, Choi JS (2008) Flowering periods, genetic characteristics, and cross-pollination rate of Zoysia spp. in natural open-pollination. Kor Turfgrass Sci 22:13–24
Fei S, Nelson E (2003) Estimation of pollen viability, shedding pattern, and longevity of creeping bentgrass on artificial media. Crop Sci 43:2177–2181
Ganesan G, Han YJ, Bae TW, Hwang OJ, Chandrasekhar T, Shin AY, Goh CH, Nishiguchi S, Song IJ, Lee HY, Song PS (2012) Overexpression of phytochrome A and its hyperactive mutant improves shade tolerance and turf quality in creeping bentgrass and zoysiagrass. Planta 236:1135–1150
Green JM, Owen MDK (2011) Herbicide-resistant crops: utilities and limitations for herbicide-resistant weed management. J Agric Food Chem 59:5819–5829
Green JM (2012) The benefits of herbicide-resistant crops. Pest Manag Sci 68:1323–1331
Henry C, Morgan D, Weekes R, Roger D, Caroline B (2003) Farm scale evaluations of GM crops: monitoring gene flow from GM crops to non-GM equivalent crops in the vicinity (contract reference EPG 1/5/138) part I: forage maize. Corpus ID: 110884721
Honda M, Kono M (1963) Morphological and anatomical studies of the lawn grasses with special reference to the Japanese lawn grass, Zoysia japonica Steud. Tech Bull Fac Horticulture Chiba Univ 11:1–22
Huang Z, Zhu J, Mu X, Lin J (2004) Pollen dispersion, pollen viability and pistil receptivity in Leymus chinensis. Ann Bot 93:295–301
Kang HG, Bae TW, Jeong OC, Sun HJ, Lim PO, Lee HY (2009) Evaluation of viability, shedding pattern, and longevity of pollen from genetically modified (GM) herbicid-tolerant and wild-type zoysiagrass (Zoysia japonica Steud.). J Plant Biol 52:630–634
Koubouris GC, Metzidakis IT, Vasilakakis MD (2009) Impact of temperature on olive (Olea europaea L.) pollen performance in relation to relative humidity and genotype. J Exp Bot 67:209–214
Luna S, Figueroa JM, Baltazar MB, Gomez LR, Townsend R, Schoper JB (2001) Maize pollen longevity and distance isolation requirements for effective pollen control. Crop Sci 41:1551–1557
Park WS, Yeh WH, Lee SW, Han SS, Lee JS, Lim CK, Lee YH (2008) Electron microscopic study for the influence of soaking in hot water and prochloraz solution on spore and mycelium of Fusarium fujikuroi infected in rice seed. Res Plant Dis 14:176–181
Pfender W, Graw R, Bradley W, Carney M, Maxwell L (2007) Emission rates, survival, and modeled dispersal of viable pollen of creeping bentgrass. Crop Sci 47:2529–2539
Reichman JR, Watrud LS, Lee EH, Burdick CA, Bollman MA, Storm MJ, King GA, Mallory-Smith C (2006) Establishment of transgenic herbicide-resistant creeping bentgrass (Agrostis stolonifera L.) in nonagronomic habitats. Mol Ecol 15:4243–4255
Song IJ, Bae TW, Ganesan M, Kim JI, Lee HY, Song PS (2013) Transgenic herbicide-resistant turfgrasses. In: Price AJ, Kelton J (eds) Herbicides—current research and case studies in use. InTech, New York, pp 377–395
Song ZP, Lu BR, Zhu YG, Chen JK (2003) Gene flow from cultivated rice to the wild species Oryza rufipogon under experimental field conditions. New Phytol 157:657–665
Song ZP, Lu BR, Chen J (2004) Pollen flow of cultivated rice measured under experimental conditions. Biodivers Conserv 13:579–590
Stephens JC, Quinby JR (1934) Anthesis, pollination, and fertilization in sorghum. J Agric Res 49:123–136
Sun HJ, Kang HG, Bae TW, Cho TG, Kim J, Lim PO, Riu KZ, Lee HY (2010) Assessment of phosphinothricin acetyltransferase (PAT) degradation from transgenic zoysiagrass digested with simulated gastric fluid (SGF). J Plant Biol 53:113–120
Teare ID, Anwar MM, Canode CL (1969) Viability of Kentucky bluegrass pollen (Poa pratensis L. ‘Newport’) as influenced by collection time and temperature. Agron J 62:515–516
Toyama K, Bae CH, Kang JG, Lim YP, Adachi T, Riu KZ, Song PS, Lee HY (2003) Production of herbicide-tolerant zoysiagrass by Agrobacterium-mediated transformation. Mol Cells 16:19–27
Watrud LS, Lee EH, Fairbrother A, Burdick C, Reichman JR, Bollman M, Storm M, King G, de VanWater PK (2004) Evidence for landscape-level, pollen-mediated gene flow from genetically modified creeping bentgrass with CP4 EPSPS as a marker. Proc Natl Acad Sci 101(40):14533–14538
Wang ZY, Ge Y, Scott M, German S (2004) Viability and longevity of pollen from transgenic and non-transgenic tall fescue (Festuca arundinacea) (Poaceae) plants. Am J Bot 91:523–530
Yeam DY, Joo YK (1985) Optimum seed coat scarification and light treatment for the germination of zoysiagrass (Zoysia japonica Steud.) seed. Hort Environ Biotech 26:179–185
Yu TY, Yeam DY, Kim YJ (1975) Promoting of low temperature and dark germinating potentiality in red light treated seeds of Zoysia grass (Zoysia japonica Steud.). Hort Environ Biotech 16:225–232
Acknowledgments
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) of the Ministry of Education (2019R1A6A1A11052070), Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

11816_2021_667_MOESM1_ESM.jpg
Supplementary Fig. S1. Monitoring of long distance cross fertilization. Pool detection limit of the LL strip test (A) and PCR (B). Lane 1 and 2 are WT and JG21, respectively. Lane 3 to 7 mixed WT to JG21 at a certain ratio. Lane 3, 1:1; lane 4, 3:1; lane 5, 10:1; lane 6, 20:1; lane 7, 50:1. Arrow heads in Supplementary Fig. S1A and 1B indicate the PAT protein band and the targeted PCR product (810 bp), respectively. C, Each strip was tested with samples pooled from 9 sites and 107 strips were used for samples of total 963 sites. D, Each PCR was tested with samples pooled from 9 sites and 107 PCRs were used for samples of total 963 sites. The right agarose gel image shows the result of the first 50 out of 107 PCRs. The 0 lane is the result from the mixture of WT to JG21 (10:1). As a PCR control marker, an actin gene (900 bp) of zoysiagrass was amplified with 5′-GACATGGTAC TGGAATGGTC-3′ and 5′-AGGATTCAAA GGAGCTTCAG-3′ primer set (the left image)
Rights and permissions
About this article
Cite this article
Kang, HG., Chung, OC., Bae, TW. et al. Pollen-mediated flow of bar gene in transgenic herbicide-resistant turf grass Zoysia japonica. Plant Biotechnol Rep 15, 241–250 (2021). https://doi.org/10.1007/s11816-021-00667-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-021-00667-4