Abstract
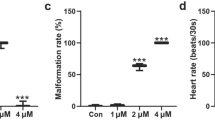
Genetically modified (GM) plants have been mainly advanced for the mass production of agricultural plants. However, there are concerns about GM crops which cause side effects on ecosystems and human beings. Zebrafish is a better in vivo model and possess the advantages; small size, large clutches, transparency, low cost, and physiological similarity to mammals. The objective of this study was to assess the safety of non-GM and GM grasses (Zoysia japonica) including JG21, JG21-MS1, JG21-MS2 in zebrafish embryos by examining the survival rates, heart-beating rate, yolk sac edema size, cell death, and morphology. The survival rates, heart-beating rates, and yolk sac edema sizes in non-GM and GM grass-treated zebrafish were similar change with the control group. And, all the groups did not show a significant difference within less than 1.0-fold in cell death by fluorescence levels. These results indicate that the zebrafish model is a useful and simple tool and can be used for risk assessment of GM plants.





Similar content being viewed by others
References
Antkiewicz DS, Burns CG, Carney SA, Peterson RE, Heideman W (2005) Heart malformation is an early response to TCDD in embryonic zebrafish. Toxicol Sci 84:368–377
Bae TW, Kim J, Song IJ, Song SY, Lim PO, Song PS, Lee HY (2009) Production of unbolting lines through gamma-ray irradiation mutagenesis in genetically modified herbicide-tolerant Zoysia japonica. Breed Sci 59:103–105
Braunbeck T, Lammer E (2006) Detailed review paper “Fish embryo toxicity assays”. UBA report under contract no. 20385422:298 German Federal Environment Agency, Berlin
Cellini F, Chesson A, Colquhoun I, Constable A, Davies HV, Engel KH, Gatehouse AMR, Kärenlampi S, Kok EJ, Leguay J-J, Lehesranta S, Noteborn HPJM, Pedersen J, Smith M (2004) Unintended effects and their detection in genetically modified crops. Food Chem Toxicol 42:1089–1125
Chassy B, Egnin M, Gao Y, Kleter GA, Nestel P, Newell-McGloughlin M, Phipps RH, Shillito R, Glenn K (2008) Nutritional and safety assessments of foods and feeds nutritionally improved through biotechnology. Compr Rev Food Sci F 7:50–113
Deng J, Yu L, Liu C, Yu K, Shi X, Yeung LWY, Lam PKS, Wu RSS, Zhou B (2009) Hexabromocyclododecane-induced developmental toxicity and apoptosis in zebrafish embryos. Aquat Toxicol 93:29–36
EFSA Gmo Panel Working Group on Animal Feeding Trials (2008) Safety and nutritional assessment of GM plants and derived food and feed: the role of animal feeding trials. Food Chem Toxicol 46:S2–S70
Eisen JS (1996) Zebrafish make a big splash. Cell 87:969–977
Embry MR, Belanger SE, Braunbeck TA, Galay-Burgos M, Halder M, Hinton DE, Léonard MA, Lillicrap A, Norberg-King T, Whale G (2010) The fish embryo toxicity test as an animal alternative method in hazard and risk assessment and scientific research. Aquat Toxicol 97:79–87
EMEA (European Medicines Agency) (2006) Guidance on the environmental risk assessment of medicinal products for human use. CPMP/SWP/4447/00. London, UK
Fishman MC (1999) Zebrafish genetics: the enigma of arrival. Proc Natl Acad Sci USA 96:10554–10556
Fraysse B, Mons R, Garric J (2006) Development of a zebrafish 4-day embryo-larval bioassay to assess toxicity of chemicals. Ecotoxicol Environ Saf 63:253–267
Gabriels IJ, Vergauwen L, De Boevre M, Van Dongen S, Blust R, De Saeger S, Eeckhout M, De Loose M, Knapen D (2019) Optimizing the use of zebrafish feeding trials for the safety evaluation of genetically modified crops. Int J Mol Sci 20:1472
Hallare AV, Köhler HR, Triebskrn R (2004) Developmental toxicity and stress protein responses in zebrafish embryos after exposure to diclofenac and its solvent, DMSO. Chemosphere 56:659–666
Hassoun E, Kariya C, Williams FE (2005) Dichloroacetate-induced developmental toxicity and production of reactive oxygen species in zebrafish embryos. J Biochem Mol Toxicol 19:52–58
Henn K, Braunbeck T (2011) Dechorionation as a tool to improve the fish embryo toxicity test (FET) with the zebrafish (Danio rerio). Comp Biochem Physiol C Toxicol Pharmacol 153:91–98
Hill AJ, Teraoka H, Heideman W, Peterson RE (2005) Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol Sci 86:6–19
Jeong HR, Sun HJ, Kang JN, Kang HK, Lee HY (2020) Allergenicity and toxicity evaluation of the PAT protein expressed in herbicide-tolerant genetically modified Zoysia japonica. J Plant Biotechnol 47(4):316–323
Kim EA, Lee SH, Ko CI, Cha SH, Kang MC, Kang SM, Ko SC, Lee WW, Ko JY, Lee JH, Kang N, Oh JY, Ahn G, Jee YH, Jeon YJ (2014) Protective effect of fucoidan against AAPH-induced oxidative stress in zebrafish model. Carbohydr Polym 102:185–191
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203:253–310
Lema SC, Schultz IR, Scholz NL, Incardona JP, Swanson P (2007) Neural defects and cardiac arrhythmia in fish larvae following embryonic exposure to 2, 2′, 4, 4′-tetrabromodiphenyl ether (PBDE 47). Aquat Toxicol 82:296–307
Nagel R (2002) DarT: The embryo test with the zebrafish Danio reri–a general model in ecotoxicology and toxicology. Altex 19:38–48
Nasevicius A, Ekker SC (2001) The zebrafish as a novel system for functional genomics and therapeutic development applications. Curr Opin Mol Ther 3:224–228
OECD (The Organisation for Economic Co-Operation and Development) (1981) OCED guidelines for the testing of chemicals, section 4: health effects. Paris, France
Phipps RH, Beever DE (2000) New technology: issues relating to the use of genetically modified crops. J Anim Feed Sci 9:543–562
Rayan AM, Nigussie F, Abbott LC (2015) Safety evaluation of stacked genetically modified corn Event (MON89034×MON88017) using zebrafish as an animal model. Food Nutr Sci 6:1285
Richards FM, Alderton WK, Kimber GM, Liu Z, Strang I, Redfem WS, Valentin J-P, Winter MJ, Hutchinson TH (2008) Validation of the use of zebrafish larvae in visual safety assessment. J Pharmacol Toxicol Methods 58:50–58
Sanden M, Ornsrud R, Sissener NH, Jorgensen S, Gu J, Bakke AM, Hemre GI (2013) Cross-generational feeding of Bt (Bacillus thuringiensis)-maize to zebrafish (Danio rerio) showed no adverse effects on the parental or offspring generations. Br J Nutr 110:2222–2233
Shi X, Du Y, Lam PKS, Wu RSS, Zhou B (2008) Developmental toxicity and alteration of gene expression in zebrafish embryos exposed to PFOS. Toxicol Appl Pharmacol 230:23–32
Sissener NH, Sanden M, Krogdahl Å, Bakke A-M, Johannessen LE, Hemere G-L (2011) Genetically modified plants as fish feed ingredients. Can J Fish Aquat Sci 68:563–574
Snell C, Bernheim A, Bergé JB, Kuntz M, Pascal G, Paris A, Ricroch AE (2012) Assessment of the health impact of GM plant diets in long-term and multigenerational animal feeding trials: a literature review. Food Chem Toxicol 50:1134–1148
Sun L, Xin L, Peng Z, Jin R, Jin Y, Qian H, Fu Z (2014) Toxicity and enantiospecific differences of two β-blockers, propranolol and metoprolol, in the embryos and larvae of zebrafish (Danio rerio). Environ Toxicol 29:1367–1378
Toyama K, Bae CH, Kang JG, Lim YP, Adachi T, Riu KZ, Song PS, Lee HY (2003) Production of herbicide-tolerant zoysiagrass by Agrobacterium-mediated transformation. Mol Cells 16:19–27
Wang H, Che B, Duan A, Mao J, Dahlgren RA, Zhang M, Zhang H, Zeng A, Wang X (2014) Toxicity evaluation of β-diketone antibiotics on the development of embryo-larval zebrafish (Danio rerio). Environ Toxicol 29:1134–1146
Wijesinghe WAJP, Kim EA, Kang MC, Lee WW, Lee HS, Vairappan CS, Jeon YJ (2013) Assessment of anti-inflammatory effect of 5ß-hydroxypalisadin B isolated from red seaweed Laurencia snackeyi in zebrafish embryo in vivo model. Envion Toxicol 37:110–117
Acknowledgements
This research was supported by the 2019 scientific promotion program funded by Jeju National University. This research was also supported in part by grants from Bio-Green 21 Program of Rural Development Administration (PJ01368501) and from Basic Science Research Program through the National Research Foundation of Korea (NRF) of the Ministry of Education (2019R1A6A1A11052070), Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, HW., Kim, EA., Sun, HJ. et al. Risk assessment of genetically modified zoysiagrasses (Zoysia japonica) in a zebrafish model. Plant Biotechnol Rep 15, 251–258 (2021). https://doi.org/10.1007/s11816-021-00662-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-021-00662-9