Abstract
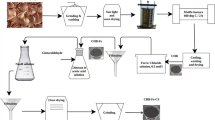
We investigated the adsorption potential of powdered branches from Ficus religiosa, an abundantly available plant, for the removal of Cu(II) from aqueous solution via column studies. Biomass was used as silica immobilized form and characterized using available techniques, including Fourier transformed infrared spectroscopy (FTIR) and scanning electron microscope (SEM). Breakthrough curve approach was used to explain removal capacity of biomass in a continuous flow mode, using different operating parameters like bed height (5-30 cm), inlet metal concentration (100-300mg/L) and pH (3-5) of the solution, at a fixed flow rate of 2mL/min. Biosorption of Cu(II) favored with increased service time (breakthrough and exhaust time) of the column with an increase in pH of inlet solution. Maximum biosorption capacity (17.5mg/g) for Cu(II) was achieved at 5 cm bed height, pH 5 and 300 mg/L influent Cu(II) concentration. Findings suggested that Ficus religiosa branch powder takes less service time and thus triggers fast removal of metal ions. Bed depth service time (BDST), Thomas and Yoon-Nelson models were effectively applied to the breakthrough data. The study indicated that the immobilized powdered branches could be used for the effective removal of Cu(II) ions in a continuous flow mode.
Similar content being viewed by others
References
C.M. Hasfalina, R. Z. Maryam, C.A. Luqman and M. Rashid, APCBEE Procedia, 3, 255 (2012).
A. Tor, N. Danaoglu, G. Arslan and Y. Cengeloglu, J. Hazard. Mater., 164, 271 (2009).
A.K. Christoforidis, S. Orfanidis, S.K. Papageorgiou, A.N. Lazaridou, E. P. Favvas and A.C. Mitropoulos, Chem. Eng. J., 277, 334 (2015).
V. S. Munagapati, V. Yarramuthi, S. K. Nadavala, S.R. Alla and K. Abburi, Chem. Eng. J., 157, 357 (2010).
Y. Nuhoglu and E. Oguz, Process. Biochem., 38, 1627 (2003).
E. Hassan, Chem. Int., 2, 89 (2016).
K.D. Ogundipe and A. Babarinde, Chem. Int., 3, 135 (2017).
M.A. Martín-Lara, G. Blázquez, M. Calero, A. I. Almendros and A. Ronda, Int. J. Miner. Proces., 148, 72 (2016).
G. Yan and T. Viraraghavan, Bioresour. Technol., 78, 243 (2001).
P. Goyal, M.M. Srivastava and S. Srivastava, J. Nucl. Agri. Biol., 36, 16 (2007).
B. Atkinson, F. Bux and H. Kasan, Water S. A., 24, 129 (1998).
D. Singh, B. Singh and R.K. Goel, J. Ethnopharmacol., 134, 565 (2011).
Z.M. Aslam, N. Ramzan, S. Naveed and N. Feroze, J. Chil. Chem. Soc., 55, 81 (2010).
P. Goyal and S. Srivastava, Arch. Environ. Prot., 34, 35 (2008).
B. Krishna and P. Venkateswarlu, Indian J. Chem. Technol., 18, 381 (2011).
S. Qaiser, A.R. Saleemi and M. Umar, J. Hazard. Mater., 166, 998 (2009).
K. S. Rao, S. Anand and P. Venkateswarlu, CLEAN, 39, 384 (2011).
A.-u.-R. Qureshi, U. Farooq, M. Athar, M. Salman and N. Rehmat, Des. Water Treat., 82, 201 (2017).
U. Farooq, M. Athar, M.A. Khan and J. A. Kozinski, Environ. Monit. Assess., 185, 845 (2013).
A.A. Abia and E.D. Asuquo, Tsing. Sci. Technol., 12, 485 (2007).
M. Lopez-Ramon, F. Stoeckli, C. Moreno-Castilla and F. Carrasco-Marin, Carbon, 37, 1215 (1999).
S. Tunali Akar, A. Gorgulu, T. Akar and S. Celik, Chem. Eng. J., 168, 125 (2011).
G. S. Bohart and E.Q. Adams, J. Amer. Chem. Soc., 42, 523 (1920).
H.C. Thomas, J. Am. Chem. Soc., 66, 1664 (1944).
Y. H. Yoon and J. H. Nelson, Am. Ind. Hyg. Assoc. J., 45, 509 (1984).
E. Heraldy, W.W. Lestari, D. Permatasari and D.D. Arimurti, J. Environ. Chem. Eng., 6, 1201 (2018).
R. Bodirlau and C.A. Teaca, Roman. J. Phys., 54, 93 (2009).
R. Sankar, R. Maheswari, S. Karthik, K. S. Shivashangari and V. Ravikumar, Mater. Sci. Eng. C., 44, 234 (2014).
T. Chuah, A. Jumasiah, I. Azni, S. Katayon and S.T. Choong, Desalination, 175, 305 (2005).
U. Farooq, J. A. Kozinski, M. A. Khan and M. Athar, Bioresour. Technol., 101, 5043 (2010).
S. Hydari, H. Sharififard, M. Nabavinia and M. reza Parvizi, Chem. Eng. J., 193, 276 (2012).
K. S. Bharathi and S. P.T. Ramesh, Appl. Water Sci., 3, 673 (2013).
H. Muhamad, H. Doan and A. Lohi, Chem. Eng. J., 158, 369 (2010).
M. Amin, A. Alazba and M. Shafiq, GLOBAL NEST JOURNAL, 19, 464 (2017).
M. Kapur and M. K. Mondal, Des. Water Treat., 57, 12192 (2016)
V. Mishra, C. Balomajumdar and V.K. Agarwal, J. Waste Manage., 2013, 1 (2013).
S. Bunluesin, M. Kruatrachue, P. Pokethitiyook, S. Upatham and G.R. Lanza, J. Biosci. Bioeng., 103, 509 (2007).
J. Lopez-Cervantes, D. I. Sanchez-Machado, R.G. Sanchez-Suarte and M. A. Correa-Murrieta, Adsorpt. Sci. Technol., 36, 215 (2018).
A. S.A. Aziz, L. A. Manaf, H. C. Man and N. S. Kumar, Environ. Sci. Poll. Res., 21, 7996 (2014).
M.A. Acheampong, K. Pakshirajan, A. P. Annachhatre and P. N. Lens, J. Ind. Eng. Chem., 19, 841 (2013).
X. Luo, Z. Deng, X. Lin and C. Zhang, J. Hazard. Mater., 187, 182 (2011).
E. Oguz and M. Ersoy, Chem. Eng. J., 164, 56 (2010).
W.-C. Tsai, M.D. G. de Luna, H. L. P. Bermillo-Arriesgado, C. M. Futalan, J. I. COlades and M.-W. Wan, Int. J. Polym. Sci., 2016, 1 (2016).
M. Khitous, S. Moussous, A. Selatnia and M. Kherat, Des. Water Treat., 57, 16559 (2016).
P.N. P.P. Sivakumar and P.N. Palanisamy, J. Sci. Ind. Res., 68, 894 (2009).
Z.Z. Chowdhury, S. M. Zain, R. A. Khan, R. F. Rafique and K. Khalid, BioResourc., 7, 2895 (2012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tariq, M., Farooq, U., Athar, M. et al. Biosorption of Cu(II) from aqueous solution onto immobilized Ficus religiosa branch powder in a fixed bed column: Breakthrough curves and mathematical modeling. Korean J. Chem. Eng. 36, 48–55 (2019). https://doi.org/10.1007/s11814-018-0189-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-018-0189-3