Abstract
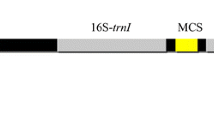
Endogenous tubulin promoter has been widely used for expressing foreign genes in green algae, but the efficiency and feasibility of endogenous tubulin promoter in the economically important Porphyra yezoensis (Rhodophyta) are unknown. In this study, the flanking sequences of beta-tubulin gene from P. yezoensis were amplified and two transient expression vectors were constructed to determine their transcription promoting feasibility for foreign gene gusA. The testing vector pATubGUS was constructed by inserting 5′-and 3′-flanking regions (Tub5′ and Tub3′) up-and down-stream of β-glucuronidase (GUS) gene (gusA), respectively, into pA, a derivative of pCAT®3-enhancer vector. The control construct, pAGUSTub3, contains only gusA and Tub3′. These constructs were electroporated into P. yezoensis protoplasts and the GUS activities were quantitatively analyzed by spectrometry. The results demonstrated that gusA gene was efficiently expressed in P. yezoensis protoplasts under the regulation of 5′-flanking sequence of the beta-tubulin gene. More interestingly, the pATubGUS produced stronger GUS activity in P. yezoensis protoplasts when compared to the result from pBI221, in which the gusA gene was directed by a constitutive CaMV 35S promoter. The data suggest that the integration of P. yezoensis protoplast and its endogenous beta-tubulin flanking sequences is a potential novel system for foreign gene expression.
Similar content being viewed by others
References
Bradford, M. M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analyt. Biochem., 72: 248–254.
Dai, J. X., Q. Q. Zhang, and Z. M. Bao, 1993. Genetic breeding and seedling raising experiments with Porphyra protoplasts. Aquaculture, 111:139–145.
Davies, J. P., D. P. Weeks, and A. R. Grossman, 1992. Expression of the arylsulfatase gene from the β2-tubulin promoter in Chlamydomonas reinhardtii. Nucleic Acids Res., 20: 2 959–2 965.
Gong, Q. H., F. Han, J. X. Dai, H. Q. Liu, H. S. Guan, et al., 2005. Rapid isolation and sequence analysis of the beta-tubulin gene from Porphyra yezoensis (Rhodophyta). J. Appl. Phycol., 17: 1–5.
Hallmann, A., A. Rappel, and M. Sumper, 1997. Gene replacement by homologous recombination in the multicellular green alga Volvox carteri. Proc. Natl. Acad. Sci. USA, 94: 7 469–7 474.
Jefferson, R. A., 1987. Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol. Bio. Rep., 5: 387–405.
Jiang, P., S. Qin, and C. K. Tseng, 2002. Expression of hepatitis B surface antigen gene (HbsAg) in Laminaria japonica (Laminariales, Phaeophyta). Chinese Sci. Bull., 47: 1 438–1 440.
Kuang, M., S. J. Wang, Y. Li, D. L. Shen, and C. K. Zeng, 1998. Transient expression of exogenous in P. yezoensis (Rhodophyta). Chin. J. Oceanol. Limnol., 16: 56–61.
Kubler, J. E., S. C. Minoch, and A. C. Mathieson, 1994. Transient expression of the GUS reporter gene in protoplasts of Porphyrus miniata (Rhodophyta). J. Marine Biotech., 1: 165–169.
Lioudmila, A., J. Zaslavskaia, and C. Lippmeier, 2000. Transformation of the diatom Phaeodactylum tricomutum (Bacillariophyceae) with a variety of selectable marker and reporter genes. J. Phycol., 36, 370–386.
Liu, H. Q., W. G. Yu, J. X. Dai, Q. H. Gong, K. F. Yang, et al., 2004. Construction of Porphyra yezoensis pure line from proprotoplasts and its 18S rDNA sequence determination. J. Ocean Univ. Chin., 3: 60–64.
Liu, H. Q., W. G. Yu, J. X. Dai, Q. H. Gong, K. F. Yang, et al., 2003. Increasing the transient expression of GUS gene in Porphyra yezoensis by 18S rDNA targeted homologous recombination. J. Appl. Phycol., 15: 371–377.
Mizukami, Y., M. Hado, H. Kito, M. Kunimoto, and N. Murase, 2004. Reporter gene introduction and transient expression in protoplasts of Porphyra yezoensis. J. Appl. Phycol., 16: 23–29.
Odell, J. T., F. Nagy, and N. H. Chua, 1985. Identification of DNA sequences required for activity of the cauliflower mosaic virus 35S promoter. Nature, 313: 810–812.
Rochaix, J. D., and J. Dillewijn, 1982. Transformation of the green alga Chlamydomonas reinhardii with yeast DNA. Nature, 296: 70–72.
Rosfjord, E., K. Lamb, and A. Rizzino, 1994. Cryptic promoter activity within the backbone of a plasmid commonly used to prepare promoter/reporter gene constructs. In Vitro Cell Dev. Biol. Anim., 30A: 477–481.
Russell, W. M., and T. R. Klaenhammer, 2001. Identification and cloning of gusA, encoding a new β-glucuronidase from Lactobacillus gasseri ADH. Appl. Environ. Microbiol., 67: 1253–1261.
Sambrook, J., E. F. Fritsch, and T. Maniatis, 2001. Purification of plasmid DNA by precipitation with polyethylene glycol. In: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press, New York, 59–62.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gong, Q., Yu, W., Dai, J. et al. Efficient gusA transient expression in Porphyra yezoensis protoplasts mediated by endogenous beta-tubulin flanking sequences. J Ocean Univ. China 6, 21–25 (2007). https://doi.org/10.1007/s11802-007-0021-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11802-007-0021-x