Abstract
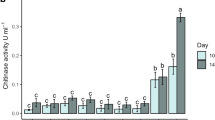
Twelve samples derived from different locations in south central area of China are treated by enrichment and spread-plate technique for initial screening. Seven chitinase-producing strains are isolated. The chitinase present in the culture supernatant of strain CS-01 possesses the maximum activity of 0.118 U/mL. Analysis of the morphological feature and the ITS rDNA sequence reveals that strain CS-01 belongs to Aspergillus fumigatus. Production of the chitinase is regulated by a inducible way and the maximum activity appears at 36 h in colloidal chitin culture. Purification of the chitinase is carried out by salting out, gel filtrate chromatography and anion exchange chromatography sequentially. Native-PAGE and SDS-PAGE indicate that the chitinase from A. fumigatus CS-01 is a monomer with the relative molecular mass estimated to be 4.50×104. Its maximum activity appears at pH 5 and 55 °C. The chitinase is stable at pH 4.0–7.5 and below 45 °C.
Similar content being viewed by others
References
RINAUDO M. Chitin and chitosan: Properties and applications [J]. Progress in Polymer Science, 2006, 31(7): 603–632.
MAJETI N V, RAVI K. A review of chitin and chitosan applications [J]. Reactive and Functional Polymers, 2000, 46(1): 1–27.
PRASHANTH K, THARANATHAN R. Chitin/chitosan: Modifications and their unlimited application potential—An overview [J]. Trends in Food Science & Technology, 2007, 18(3): 117–131.
HIRANO S. Chitin and chitosan as novel biotechnological materials [J]. Polymer International, 1999, 48(8): 732–734.
XING Rong-e, LIU Song, YU Hua-hua, GUO Zhan-yong, WANG Pi-bo, LI Cui-ping, LI Zhi-en, LI Peng-cheng. Salt assisted acid hydrolysis of chitosan to oligomers under microwave irradiation [J]. Carbohydrate Research, 2005, 340(13): 2150–2153.
HIEN N, NAGASAWA N, THAM L, YOSHII F, DANG V H, MITOMO H, MAKUUCHI K, KUME T. Growth-promotion of plants with depolymerized alginates by irradiation [J]. Radiation Physics and Chemistry, 2000, 59(1): 97–101.
ALLAN G, PEYRON M. Molecular weight manipulation of chitosan I: Kinetics of depolymerization by nitrous acid [J]. Carbohydrate Research, 1995, 277(2): 257–272.
PANTALEONE D, YALPANI M, SCOLLAR M. Unusual susceptibility of chitosan to enzymatic hydrolysis [J]. Carbohydrate Research, 1992, 237(1): 325–332.
BOLAR J P, NORELLI J L, WONG K W, HAYES C K, HARMAN G E, ALDWINCKLE H S. Expression of endochitinase from Trichoderma harzianum in transgenic apple increases resistance to apple scab and reduces vigor [J]. Phytopatholigy, 2000, 90(1): 72–77.
ZIMAND G, ELAD Y. Efect of Trichoderma harizianum on Bollrylis cinerea pathogenicity [J]. Phytopatholigy, 1996, 86(5): 945–956.
TRACHUK L, REVINA L, SHEMYAKINA T. Chitinases of bacillus lichenfoumis B-6839: Isolation and properties [J]. Canadian Journal of Micrrobiology, 1996, 42(4): 307–315.
XIAO Xiang, ZHOU Ying, WANG Feng-ping. Isolation and identification of a high-efficient chitin-degrading marine bacterium CB101 and studies on its chitinase system[J]. Acta Oceanologica Sinica, 2003, 25(1): 138–142. (in Chinese)
SHUTTON D, FOTHERQILL A, RINALDI M. Guide to clinically significant fungi [M]. Baltimore: Williams & Wilkins, 1998.
XIA Jin-lan, PENG An-an, HE Huan, YANG Yu, LIU Xue-duan, QIU Guan-zhou. A new strain acidithiobacillus albertensis BY-05 for bioleaching of metal sulfides ores [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(1): 168–175.
INNIS M A, GELFAND D H, SNINSKY J J, WHITE T J. PCR Protocols: A guide to methods and application [M]. San Diego, CA: Academic Press, 1990: 315–322.
RIVAS L A, PARRO V, MORENO-PAZ M. The Bacillus subtilis 168 csn gene encodes a chitosanase with similar properties to a streptomyces enzyme [J]. Microbiology, 2000, 146(1): 2929–2936.
WANG S L, CHEN Y H, WANG C L, YEN Y H, CHERN M K. Purification and characterization of a serine protease extracellularly produced by Aspergillus fumigatus in a shrimp and crab shell powder medium [J]. Enzyme and Microbial Technology, 2005, 36(5): 660–665.
ESCOTT G M, HEAM V M, ADAMS D J. Inducible chitinolytic system of aspergillus fumigatus [J]. Microbiology, 1998, 144(6): 1575–1581.
MARION M M. A rapid and sensitive method for the quantitation of protein utilizing the principle of protein-dye bindiag [J]. Analytical Biochemistry, 1976, 72: 248–254.
UEDA H, ARAI M. Purification and some properties of chitinases from Aeromonas sp. No.10S-24 [J]. Bioscience, Biotechnology, and Biochemistry, 1992, 56: 460–464.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Projects(50621063, 50674101) supported by the National Natural Science Foundation of China
Rights and permissions
About this article
Cite this article
Xia, Jl., Xiong, J., Xu, T. et al. Purification and characterization of extracellular chitinase from a novel strain Aspergillus fumigatus CS-01. J. Cent. South Univ. Technol. 16, 552–557 (2009). https://doi.org/10.1007/s11771-009-0092-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-009-0092-5